Abstract
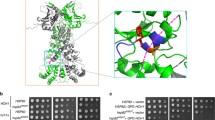
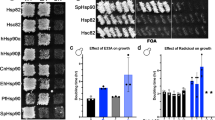
A single nucleotide polymorphism (SNP) that causes a missense mutation of highly conserved Gln488 to His of the α isoform of the 90-kDa heat shock protein (Hsp90α) molecular chaperone is observed in Caucasians. The mutated Hsp90α severely reduced the growth of yeast cells. To investigate this molecular mechanism, we examined the domain–domain interactions of human Hsp90α by using bacterial 2-hybrid system. Hsp90α was expressed as a full-length form, N-terminal domain (residues 1–400), or middle (residues 401–617) plus C-terminal (residues 618–732) domains (MC domain/amino acids 401–732). The Gln488His substitution in MC domain did not affect the intra-molecular interaction with N-terminal domain, whereas the dimeric interaction-mediated by the inter-molecular interaction between MC domains was decreased to 32%. Gln488Ala caused a similar change, whereas Gln488Thr, which exceptionally occurs in mitochondrial Hsp90 paralog, fully maintained the dimeric interaction. Therefore, the SNP causing Gln488His mutation could abrogate the Hsp90 function due to reduced dimerization.





Similar content being viewed by others
Abbreviations
- SNP:
-
Single nucleotide polymorphism
- Hsp:
-
Heat shock protein
- Hsp90:
-
90-kDa Heat shock protein
- hHsp90α and hHsp90β:
-
α and β Isoforms, respectively, of human Hsp90
- Grp94:
-
94-kDa Glucose-regulated protein/endoplasmic reticulum paralog of Hsp90
- Trap1:
-
Mitochondrial paralog of Hsp90
- yeast Hsp82 and Hsc82:
-
82-kDa Heat shock protein and heat shock cognate protein of yeast, respectively
- HtpG:
-
Bacterial ortholog of eukaryotic Hsp90
- PAGE:
-
Polyacrylamide gel electrophoresis
- SDS:
-
Sodium dodecyl sulfate
References
Hickey E, Brandon SE, Smale G, Lloyd D, Weber LA (1989) Mol Cell Biol 9:2615–2626
Rebbe NF, Ware J, Bertina RM, Modrich P, Sttafford DW (1987) Gene 53:235–245
Borkovich KA, Farrelly FW, Finkelstein DB, Taulien J, Lindquist S (1989) Mol Cell Biol 9:3919–3930
Farrelly FW, Finkelstein DB (1984) J Biol Chem 259:5745–5751
Filho PAH, Cedenho AP, Lima SB, Ortiz V, Srougi M (2005) Int J Braz J Urol 31:236–244
Passarino G, Cavalleri GL, Stecconi R, Franceschi C, Altomare K, Dato S, Greco V, Sforza LLC, Underhill PA, Benedictis G (2003) Hum Mutat 21:554–555
MacLean MJ, Martinez M, Bot N, Picard D (2006) Biochem Biophys Res Commun 337:133–137
Nemoto T, Sato N, Iwanari H, Yamashita H, Takagi T (1997) J Biol Chem 272:26179–26187
Prodromou C, Roe SM, O’Brien R, Ladbury JE, Piper PW, Pearl LH (1997) Cell 90:65–75
Stebbins CE, Russo AA, Schneider C, Rosen N, Hartl FU, Pavletich NP (1997) Cell 89:239–250
Matsumoto S, Tanaka S, Nemoto TK, Ono T, Takagi T, Kobayakawa T, Imai J, Kimura Y, Yahara I, Ayuse T, Oi K, Mizuno A (2002) J Biol Chem 277:34959–34966
Nemoto T, Ohara-Nemoto Y, Ota M, Takagi T, Yokoyama K (1995) Eur J Biochem 233:1–8
Karimova G, Ullmann A, Ladant D (2001) J Mol Microbiol Biotechnol 3:73–82
Tanaka E, Nemoto TK, Ono T (2001) Eur J Biochem 268:5270–5277
Maki RG, Old LJ, Srivastava PK (1990) Proc Natl Acad Sci USA 87:5658–5662
Bardwell JC, Craig EA (1987) Proc Natl Acad Sci USA 84:5177–5181
Felts SJ, Owen BA, Nguyen P, Trepel J, Donner DB, Toft DO (2000) J Biol Chem 275:3305–3312
Song HY, Dunbar JD, Zhang YX, Guo D, Toft DO (1996) J Biol Chem 270:3574–3581
Strausberg RL, Feingold EA, Grouse LH, Derge JG, Klausner RD, Collins FS, Wagner L, Shenmen CM, Schuler GD, Altschul SF, Zeeberg B, Buetow KH, Schaefer CF, Bhat NK, Hopkins RF, Jordan H, Moore T, Max SI, Wang J, Hsieh F, Diatchenko L, Marusina K, Farmer AA, Rubin GM, Hong L, Stapleton M, Soares MB, Bonaldo MF, Casavant TL, Scheetz TE, Brownstein MJ, Usdin TB, Toshiyuki S, Carninci P, Prange C, Raha SS, Loquellano NA, Peters GJ, Abramson RD, Mullahy SJ, Bosak SA, McEwan PJ, McKernan KJ, Malek JA, Gunaratne PH, Richards S, Worley KC, Hale S, Garcia AM, Gay LJ, Hulyk SW, Villalon DK, Muzny DM, Sodergren EJ, Lu X, Gibbs RA, Fahey J, Helton E, Ketteman M, Madan A, Rodrigues S, Sanchez A, Whiting M, Madan A, Young AC, Shevchenko Y, Bouffard GG, Blakesley RW, Touchman JW, Green ED, Dickson MC, Rodriguez AC, Grimwood J, Schmutz J, Myers RM, Butterfield YS, Krzywinski MI, Skalska U, Smailus DE, Schnerch A, Schein JE, Jones SJ, Marra MA (2002) Proc Natl Acad Sci USA 99:16899–16903
Koyasu S, Nishida E, Kadowaki T, Matsuzaki F, Iida K, Harada F, Kasuga M, Sakai H, Yahara I (1986) Proc Natl Acad Sci USA 83:8054–8058
Welch WJ, Feramisco JR (1982) J Biol Chem 257:14949–14959
Meyer P, Prodromou C, Hu B, Vaughan C, Roe SM, Panaretou B, Piper PW, Pearl LH (2003) Mol Cell 11:647–658
Prodromou C, Roe SM, Piper PW, Pearl LH (1997) Nat Struct Biol 4:477–482
Meyer P, Prodromou C, Liao C, Hu B, Roe SM, Vaughan CK, Vlasic I, Panaretou B, Piper PW, Pearl LH (2004) EMBO J 23:1402–1410
Shiau AK, Harris SF, Southworth DR, Agard DA (2006) Cell 127:329–340
Kobayakawa T, Yamada S, Mizuno A, Nemoto TK (2008) Cell Stress Chaperones 13:97–104
Yamada S, Ono T, Mizuno A, Nemoto TK (2003) Eur J Biochem 270:146–154
Moore SK, Kozak C, Robinson EA, Ullrich SJ, Appella E (1989) J Biol Chem 264:5343–5351
Lopatin DE, Combs A, Sweier DG, Fenno JC, Dhamija S (2000) Infect Immun 68:1980–1987
Acknowledgments
This work was supported in part by Grants-in Aid for Scientific Research from the Ministry of Education, Culture, Sports, Science, and Technology of Japan (to S.Y.) and from Nagasaki University (Y.O.-N.).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kobayakawa, T., Yamada, Si., Mizuno, A. et al. Single Nucleotide Polymorphism that Accompanies a Missense Mutation (Gln488His) Impedes the Dimerization of Hsp90. Protein J 28, 24–28 (2009). https://doi.org/10.1007/s10930-008-9160-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10930-008-9160-1