Abstract
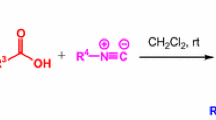
Benzene-1,2-, 1,3-, and 1,4-di-N-substituted carbamates (1–15) are synthesized as the constrained analogs of gauche, eclipsed, and anti conformations, respectively, for the glycerol backbones of triacylglycerol. Carbamates 1–15 are characterized as the pseudo substrate inhibitors of cholesterol esterase. Long chain carbamates are more potent inhibitors than short chain ones. Comparison of different geometries for benzene-di-substituted carbamates, such as benzene-1,2-di-N-octylcarbamate (3) (ortho-3), benzene-1,3-di-N-octylcarbamate (8) (meta-8), and benzene-1,4-di-N-octylcarbamates (13) (para-13), indicates that inhibitory potencies are as followed: meta-8 > para-13 > ortho-3. Therefore, we suggest that the preferable conformation for the C(sn-1)–O/C(sn-2)–O glycerol backbone in the enzyme–triacylgycerol complex is the eclipsed conformation. Meanwhile, kinetic data indicate that among ortho, meta, and para carbamates, meta carbamates most resemble the substrate cholesterol ester.





Similar content being viewed by others
Abbreviations
- ACS:
-
Acyl chain binding site
- CEase:
-
Cholesterol esterase
- LDL:
-
Low-density lipoprotein
- PNPB:
-
p-nitrophenyl butyrate
- TX:
-
Triton-X 100
References
Lopez-Candales A, Bosner MS, Spilburg CA, Lange LG (1993) Biochemistry 32:12085–12089
Wang C-S, Hartsuck JA (1993) Biochim Biophys Acta 1166:1–19
Hui DY (1996) Biochim Biophys Acta 1302:1–14
Wang X, Wang C-S, Tang J, Dyda F, Zhang XC (1997) Structure 5:1209–1218
Chen JC-H, Miercke LJW, Krucinski J, Starr JR, Saenz G, Wang X, Spilburg CA, Lange LG, Ellsworth JL, Stroud RM (1998) Biochemistry 37:5107–5117
Brodt-Eppley J, White P, Jenkins S, Hui D (1995) Biochim Biophys Acta 1272:69–72
Auer J, Eber B (1999) J Clin Basic Cardiol 2:203–208
Maron DJ, Fazio S, Linton MF (2000) Circulation 101:207–213
Pioruńska-Stolzmann M, Piroruńska-Mikołajczak A (2002) Pharm Res 43:359–362
Chiou S-Y, Lai G-W, Lin L-Y, Lin G (2006) Ind J Biochem Biophys 43:52–55
Hosie L, Sutton LD, Quinn DM (1987) J Biol Chem 262:260–264
Feaster SR, Lee K, Baker N, Hui DY, Quinn DM (1996) Biochemistry 35:16723–16734
Feaster SR, Quinn DM (1997) Meth Enzymol 286:231–252
Lin G, Shieh C-T, Ho H-C, Chouhwang J-Y, Lin W-Y, Lu C-P (1999) Biochemistry 38:9971–9981
Lin G, Lin Y-F, Hwang M-T, Lin Y-Z (2004a) Bioorg Med Chem Lett 14:751–755
Lin G, Yu G-Y (2005) Bioorg Med Chem Lett 15:2405–2408
Lin M-C, Hwang M-T, Chang H-G, Lin C-S, Lin G (2007a) J Biochem Mol Toxicol 21:348–353
Lin M-C, Hwang M-T, Chang H-G, Lin C-S, Lin G (2007b) Eur J Lipid Sci Technol 109:1104–1110
Pietsch M, Gütschow M (2002) J Biol Chem 277:24006–24013
Pietsch M, Gütschow M (2005) J Med Chem 48:8270–8288
Lin G, Lin Y-F, Hwang M-T, Lin Y-Z (2004b) Bioorg Med Chem Lett 14:751–755
Acknowledgement
We thank the National Science Council of Taiwan for financial support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chiou, SY., Lin, MC., Hwang, MT. et al. Benzene-di-N-Substituted Carbamates as Conformationally Constrained Substrate Analogs of Cholesterol Esterase. Protein J 27, 276–282 (2008). https://doi.org/10.1007/s10930-008-9135-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10930-008-9135-2