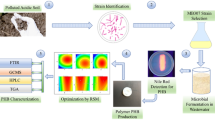
Abstract
The polymers/enzymes produced by the microorganisms isolated from the various thermal sources have stability at different levels and their usage areas can be improved according to this characteristic. In this study, prokaryotic diversity of water sample taken from Çitgöl thermal spring in Kütahya-Simav, has been investigated by cultivation techniques. In the result of this study, Geobacillus thermoparaffinivorans, Geobacillus kaustophilus, Geobacillus sp., Geobacillus thermoleovorans and Thermus thermophilus were isolated from water sample. Then, the scanning studies were performed to choose an effective PHB producer with thermal stability. Designing of experiments, analyzing of experimental results, creating the mathematical models and finding optimization conditions were performed using Box-Behnken experimental design method. PHB yield of G. kaustophilus C4-2 increased to 8.66% PHB under the determined conditions which were found as 35 °C, 3.75 days and 10 g/L sucrose. The validity and practicability of this statistical optimization strategy was confirmed relation between predicted and experimental values. The characterization of obtained PHB was performed through analyses including FTIR, H-NMR, TG/DTA and viscosity. According to the result of TG/DTA, PHB was thermal resistant and could remain 14.3% without decomposition at 600 °C.







Similar content being viewed by others
References
Woese CR, Kandler O, Wheelis ML (1990) Towards a natural system of organisms: proposal for the domains Archaea, Bacteria, and Eucarya. Proc Natl Acad Sci USA 87(12):4576–4579
Olsen GJ, Woese CR, Overbeek R (1994) The winds of (evolutionary) change: breathing new life into microbiology. J Bacteriol 176(1):1–6
Wemheuer B, Taub R, Akyol P, Wemheuer F, Daniel R (2013) Microbial diversity and biochemical potential encoded by thermal spring metagenomes derived from the Kamchatka peninsula. Archaea. https://doi.org/10.1155/2013/136714
Cai L, Ye L, Tong AH, Lok S, Zhang T (2013) Biased diversity metrics revealed by bacterial 16S pyrotags derived from different primer sets. PLoS ONE 8:e53649. https://doi.org/10.1371/journal. pone.0053649
Amin A, Ahmed I, Salam N, Kim BY, Singh D, Zhi XY, Xiao M, Li WJ (2016) Diversity and distribution of thermophilic bacteria in hot springs of Pakistan. Microb Ecol. https://doi.org/10.1007/s00248-017-0930-1
Wiegel J, Ljungdahl LG (1985) The importance of thermophilic bacteria in biotechnology. Crit Rev Biotechnol 3:39–108
Holst O, Manelius A, Krahe M, Miirkl H, Rawen N, Sharp R (1997) Thermophiles and fermentation technology. Comp Biochem Physiol 118A:415–422
Steinbuchel A (2001) Perspectives for biotechnological production and utilization of biopolymers: metabolic engineering of polyhydroxyalkanoate biosynthesis pathways as a successful example. Macromol Biosci 1:1–24
Pantazaki AA, Papaneophytou CP, Pritsa AG, Liakopoulou-Kyriakides M, Kyriakidis DA (2009) Production of polyhydroxyalkanoates from whey by Thermus thermophilus HB8. Process Biochem 44:847–853
Prabisha TP, Sindhu R, Binod P, Sankar V, Raghu KG, Pandey A (2015) Production and characterization of PHB from a novel isolate Comamonas sp. from a dairy effluent sample and its application in cell culture. Biochem Eng J 101:150–159
Pandian SR, Deepa V, Kalishwaralal K, Rameshkumar N, Jeyaraj M, Gurunathan S (2010) Optimization and fed-batch production of PHB utilizing dairy waste and sea water as nutrient sources by Bacillus megaterium SRKP-3. Bioresour technol 101(2):705–711
Sheua DS, Chenb WM, Yanga JY, Changa RC (2009) Thermophilic bacterium Caldimonas taiwanensis produces poly(3-hydroxybutyrate-co-3-hydroxyvalerate) from starch and valerate as carbon sources. Enzyme Microb Technol 44:289–294
Güngörmedi G, Demirbilek M, Mutlu MB, Denkbas EB, Çabuk A (2014) Polyhydroxybutyrate and hydroxyvalerate production by Bacillus megaterium strain A1 isolated from hydrocarbon-contaminated soil. J Appl Polym Sci. https://doi.org/10.1002/APP.40530
Lemoigne M (1926) Produits de deshydration et depolymerisation de l’acide β–oxybytyrique. Bull Soc Chim Biol 8:770–782
Nogales B, Moore ER, Abrham WR, Timmis KN (1999) Identification of the metabolically active members of a bacterial community in a polychlorinated biphenylpolluted moorland soil. Environ Microbiol 1:199–212
Cifuentes A, Anton J, Benlloch S, Donnelly A, Herbert RA, Rodriguez-Valera F (2000) Prokaryotic diversity in Zostera noltii colonized marine sediments. Appl Environ Microbiol 66:1715–1719
Altschul SF, Gish W, Miller W, Myers EW, Lipman DJ (1990) Basic local alignment search tool. J Mol Biol 215:3389–3402
Pantazaki AA, Tambaka MG, Langlois V, Guerin P, Kyriakidis DA (2004) Polyhydroxyalkanoate (PHA) biosynthesis in Thermus thermophilus: purification and biochemical properties of PHA synthase. Mol Cell Biochem 254:173–183
Unpublished. https://www.ncbi.nlm.nih.gov/nucleotide/KT266806.1?report=genbank&log$=nuclalign&blast_rank=1&RID=SX13Z9FV014
Gaultier NE, Junqueira ACM, Uchida A, Purbojati RW, Houghton JNI, Chénard C, Wong A, Kolundžija S, Clare ME, Kushwaha KK, Panicker D, Putra A, Kee C, Premkrishnan BNV, Heinle CE, Lim SBY, Vettath VK, Drautz-Moses DI, Schuster SC (2018) Genome Sequence of Geobacillus thermoleovorans SGAir0734, isolated from Singapore air. Genome Announc 6(27):e00636–e00618. https://doi.org/10.1128/genomeA.00636-18
Zeigler DR (2005) Application of a recN sequence similarity analysis to the identification of species within the bacterial genus Geobacillus. Int J Syst Evol Microbiol 55:1171–1179
Kumar V, Thakur V, Ambika, Kumar S, Dharam S (2018) Bioplastic reservoir of diverse bacterial communities revealed along altitude gradient of Pangi-Chamba trans-Himalayan region. FEMS Microbiol Lett 365(14):1–9
Reddy CSK, Ghai R, Rashmi T, Kalia VC (2003) Polyhydroxyalkanoates: an overview. Bioresour Technol 87:137–146
Logan NA, De Vos P, Dinsdale A et al (2009) Genus Geobacillus Nazina et al. 2001. In: De Vos P, Garrity G, Jones D, Krieg NR, Ludwig W, Rainey FA, Schleifer K-H, Whitman WB (eds) Bergey’s manual of systematic bacteriology, vol 3, 2nd edn. Springer, New York, pp 144–160
Chung YJ, Cha HJ, Yeo JS, Yoo YJ (1997) Production of poly(3-hydroxybutyric-co-3-hydroxyvaleric) acid using propionic acid by pH regulation. J Ferment Bioeng 83(5):492–495
Sathiyanarayanan G, Saibaba G, Kiran GS, Selvin J (2013) A statistical approach for optimization of polyhydroxybutyrate production by marine Bacillus subtilis MSBN17.Int. J Biol Macromol 59:170–177
Giedraityte G, Kalediene L (2015) Purification and characterization of polyhyroxybutyrate produced from thermophilic Geobacillus sp. AY 946034 strain. Chemija 26:38–45
Lundgren DG, Alper R, Schnaitman C, Marchessault RH (1965) Characterization of poly-β-hydoxtbutyrate extracted from different bacteria. J Bacteriol 89:245–251
Olkhov AA, Iordanskii AL, Zaikov GE (2014) Morphology and mechanical parameters of biocomposite based on LDPE-PHB. J Balkan Tribol Assoc 20:101–110
Hassan MA, Bakhiet EK, Ali SG, Hussien RH (2016) Production and characterization of polyhydroxybutyrate (PHB) produced by Bacillus sp. isolated from Egypt. J Appl Pharm Sci 6(04):046–051
Hassan MA, Bakhiet EK, Hussein HR, Ali SG (2018) Statistical optimization studies for polyhydroxybutyrate (PHB) production by novel Bacillus subtilis using agricultural and industrial wastes, Int J Environ Sci Technol. https://doi.org/10.1007/s13762-018-1900-y
Acknowledgements
The study was supported by Eskisehir Osmangazi University Scientific Research Projects Committee (Project No. BAP 201319021). The authors would like to thank Dr. Okan Zafer Yeşilel and Dr. Yeliz Buruk Sahin at Eskisehir Osmangazi University for FTIR spectra of the biomaterial and statistical analysis, respectively. This study is based partly on the PhD thesis of S. Gedikli who is one of the co-authors. We state that there is no conflict of interests in this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gedikli, S., Çelik, P.A., Demirbilek, M. et al. Experimental Exploration of Thermostable Poly (β-Hydroxybutyrates) by Geobacillus kaustophilus Using Box-Behnken Design. J Polym Environ 27, 245–255 (2019). https://doi.org/10.1007/s10924-018-1335-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10924-018-1335-z