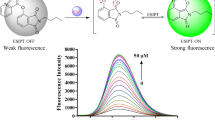
Abstract
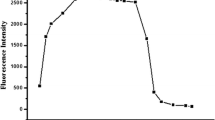
Novel fluorescent probes were constructed for the convenient and rapid analysis of Cu2+ ions, taking advantages of the the triphenylamine backbone as chromophore and acylhydrazone group as the Cu2+ recognition site. Especially, probe T2 could act as a dual-channel probe towards Cu2+ through both fluorescent and colorimetric method. Through the fluorescent method, the detection limit of probe T2 was calculated to be as low as 90 nmol/L and there was a good linear relationship between the intensity change and the concentration of Cu2+ ions. By virtue of the two-phase liquid-liquid extraction method, probe T2 could be successfully applied in practical extraction and separation of Cu2+. Furthermore, by applying a “turn-off-turn-on” circle, compound T2 could act as a sensitive probe towards S2− anions through the indirect approach and the detection limit of complex T2-Cu2+ for S2− anion was found to be 110 nmol/L.








Similar content being viewed by others
Data Availability
Not applicable.
Code Availability
Not applicable.
References
Wang L, Si W, Ye Y, Wang S, Hou F, Hou X, Cai H, Dou S, Liang J (2021) Cu-ion-implanted and polymeric carbon nitridedecorated TiO2 nanotube array for unassisted photoelectrochemical water splitting. ACS Appl Mater Interfaces 13:44184
Zhu Q, Cheng M, Zhang B, Jin K, Chen S, Ren Z, Yu Y (2019) Realizing a rechargeable high-performance Cu–Zn Battery by adjusting the solubility of Cu2+. Adv Funct Mater 29:1905979
Liu Y, Nie N, Tang H, Zhang C, Chen K, Wang W, Liu J (2021) Effective antibacterial activity of degradable copper-doped phosphate-based glass nanozymes. ACS Appl Mater Interfaces 13:11631
Wasi S, Tabrez S, Ahmad M (2013) Toxicological effects of major environmental pollutants: an overview. Environ Monit Assess 185:2585
Mogren CL, Trumble JT (2010) The impacts of metals and metal-loids on insect behaviour. Entomol Exp Appl 135:1
Khalaj M, Kamali M, Khodaparast Z, Jahanshahi A (2018) Copper based nanomaterials for environmental decontamination-an overview on technical and toxicological aspects. Ecotoxicol Environ Saf 148:813
Bolisetty S, Peydayesh M, Mezzenga R (2019) Sustainable technologies for water purifcation from heavy metals: review and analysis. Chem Soc Rev 48:463
Lee S, Chung CY, Liu P, Craciun L, Nishikawa Y, Bruemmer KJ, Hamachi I, Saijo K, Miller EW, Chang CJ (2020) Activity-based sensing with a metal-directed acyl imidazole strategy reveals cell type-dependent pools of labile brain copper. J Am Chem Soc 142:14993
Zha B, Fang S, Chen H, Guo H, Yang F An effective dual sensor for Cu2+ and Zn2+ with long-wavelength fuorescence in aqueous media based on biphenylacrylonitrile Schif-base. (2022) Spectrochim. Acta A Mol Biomol Spectrosc. 269:120765
Chen Y, Pan H, Wang F, Zhao Y, Yin H, Chen Y, Zhang J, Jiang J (2019) An ultrafast BODIPY single molecular sensor for multianalytes (acid/base/Cu2+/Bi3+) with different sensing mechanism. Dyes Pigm 165:279
Zeng L, Miller EW, Pralle A, Isacoff EY, Chang CJ (2006) A selective turn-on fluorescent sensor for imaging copper in living cells. J Am Chem Soc 128:10
Xie H, Gu Y, Liu L, Dai J (2020) A H-aggregating fuorescent probe for recognizing both mercury and copper ions based on a dicarboxyl-pyridyl bifunctionalized difuoroboron dipyrromethene. New J Chem 44:19713
Baslak C, Kursunlu AN (2018) A naked-eye fuorescent sensor for copper (II) ions based on a naphthalene conjugate bodipy dye. Photochem Photobiol Sci 17:1091
Shen Y, Zheng W, Yao Y, Wang D, Lv G, Li C (2020) Phenoxazinebased near-infrared fuorescent probes for the specifc detection of copper (II) ions in living cells. Chem Asian J 15:2864
Mishra SK, Dehuri S, Bag B (2020) Efect of n-alkyl substitution on Cu(II)-selective chemosensing of rhodamine B derivatives. Org Biomol Chem 18:316
Chawla HM, Munjal P, Goel P (2015) Synthesis and evaluation of a new colorimetric and ratiometric fuorescence probe for copper ions. J Lumin 164:138
Jung HS, Kwon PS, Lee JW, Kim JI, Hong CS, Kim JW, Yan S, Lee JY, Lee JH, Joo T, Kim JS (2009) Coumarin-derived Cu2+-selective fuorescence sensor: Synthesis, mechanisms, and applications in living cells. J Am Chem Soc 131:2008
He G, Zhao X, Zhang X, Fan H, Wu S, Li H, He C, Duan C (2010) A turn-on PET fuorescence sensor for imaging Cu2+ in living cells. New J Chem 34:1055
Wang X, Shao J, Hu Y (2019) An efficient chemodosimeter for Cu(II) ions based on hydrolysis of fuorescein and its utility in live cell imaging. Dyes Pigm 171
Xu T, Huang J, Fang M, Sui M, Zhu Y, Shentu Y, Li C, Zhu W (2020) A novel turn-on fuorescent probe based on naphthalimide for the tracking of lysosomal Cu2+ in living cells. New J Chem 44:21167
Singh P, Mittal LS, Kumar S, Bhargava G, Kumar S (2014) Perylene diimide appended with 8-hydroxyquinoline for ratiometric detection of Cu2+ ions and metal displacement driven turn on Cyanide sensing. J Fluoresc 24:909
Kowser Z, Rayhan U, Akther T, Redshaw C, Yamato T (2021) A brief review on novel pyrene based fuorometric and colorimetric chemosensors for the detection of Cu2+. Mater Chem Front 5:2173
Mu Y, Zhang C, Gao Z, Zhang X, Lu Q, Yao J, Xing S (2020) A highly selective colorimetric, absorption and fluorescence probe for Al3+ detection based on a new Schiff base compound. Synth Met 262:116334
Chowdhury S, Rooj B, Dutta A, Mandal U (2018) Review on recent advances in metal ions sensing using different fluorescent probes. J Fluoresc 28:999
Abdurahman A, Wang L, Zhang Z, Feng Y, Zhao Y, Zhang M (2020) Novel triazole-based AIE materials: Dual-functional, highly sensitive and selective fluorescence probe. Dyes Pigm 174:108050
Chen H, Yang P, Li Y, Zhang L, Ding F, He X, Shen J (2020) Insight into triphenylamine and coumarin serving as copper (II) sensors with OFF strategy and for bio-imaging in living cells. Spectrochim Acta Part A 224:117384
Mishra A, Ma C-Q, Baüerle P (2009) Functional oligothiophenes: molecular design for multidimensional nanoarchitectures and their applications. Chem Rev 109:1141
Liu Z, Yang Z, Chen S, Liu Y, Sheng L, Tian Z, Huang D, Xu H (2020) A smart reaction-based fluorescence probe for ratio detection of hydrazine and its application in living cells. Microchem J 156:104809
Samanta S, Huang M, Lin F, Das P, Chen B, Yan W, Chen J-J, Ji K, Liu L, Qu J, Yang Z (2020) Solo Smart fluorogenic probe for potential Cancer diagnosis and tracking in vivo tumorous lymphatic systems via distinct Emission signals. Anal Chem 92:1541
Cheng X, Li S, Jia H, Zhong A, Zhong C, Feng J, Qin J, Li Z (2012) Fluorescent and colorimetric probes for Mercury(II): tunable structures of Electron donor and π-Conjugated Bridge. Chem Eur J 18:1691
Cheng X, He P, Zhong Z, Liang G (2016) Reaction-based probe for hydrogen sulfite: dual-channel and good ratiometric response. Luminescence 31:1372
Fan L, Pan Y, Li W, Xu Y, Duan Y, Li R, Lv Y, Chen H, Yuan Z (2021) A near-infrared fluorescent probe with large Stokes shift for visualizing and monitoring mitochondrial viscosity in live cells and inflammatory tissues. Anal Chim Acta 1149:338203
Chen G, Li W, Zhou T, Peng Q, Zhai D, Li H, Yuan W, Zhang Y, Tang B (2015) Conjugation-Induced Rigidity in twisting molecules: filling the gap between aggregation-caused Quenching and Aggregation-Induced Emission. Adv Mater 27:4496
Xu L, Xiong F, Kang M, Huang Y, Wu K (2022) Triphenylamine indanedione as an AIE-based molecular sensor with one-step facile synthesis towards viscosity detection of liquids. Analyst 147:4132
He J, Xie Z, Xue S, Liu Y, Shi W, Chen X (2021) Synthesis of salicylhydrazone probe with high selectivity and rapid detection Cu2+ and its application in logic gate and adsorption. Chin J Org Chem 41:2839
Williams AT, R, Winfield SA, Miller JN (1983) Relative fluorescence quantum yields using a computer controlled luminescence spectrometer. Analyst 108:1067
Liu L, Dong X, Xiao Y, Lian W, Liu Z (2011) Two-photon excited fluorescent chemosensor for homogeneous determination of copper(II) in aqueous media and complicated biological matrix. Analyst 136:2139
Ressalan S, Iyer CSP (2005) Absorption and fluorescence spectroscopy of 3-hydroxy-3-phenyl- 1-o-carboxyphenyltriazene and its copper (II), nickel (II) and zinc (II) complexes: a novel fluorescence sensor. J Lumin 111:121
Barra M, Bohne C, Scaiano JC (1990) Effect of cyclodextrin complexation on the photochemistry of xanthone. Absolute measurement of the kinetics for triplet-state exit. J Am Chem Soc 112:8075
Delley B (2000) From molecules to solids with the DMol3 approach. J Chem Phys 113:7756
Wen Y, Xie Z, Shi T, Chu Y, Zhou R, Tao Y, Liang H, Qiu H, Zhao Y (2022) Synthesis of Triazole Functionalized Triphenylamine Cu2+ fluorescent probe and its application in detection and HeLa cells. Chin J Org Chem 42:1463
Lou X, Mu H, Gong R, Fu E, Qin J, Li Z (2011) Displacement method to develop highly sensitive and selective dual chemosensor towards sulfide anion. Analyst 136:684
Zhang L, Lou X, Yu Y, Qin J, Li Z (2011) A new disubstituted polyacetylene bearing pyridine moieties: convenient synthesis and sensitive chemosensor towards sulfide anion with high selectivity. Macromolecules 44:5186
Funding
This work was financially supported by the National Key Research and Development Plan (2019YFE0107200) and the Xiangyang Science and Technology Project (No. 2020YL03).
Author information
Authors and Affiliations
Contributions
XC: supervision, investigation designed and directed the study, planned and carried out the experiments, wrote the manuscript. Dr. RZ: contributed to the design and synthesis of the organic compounds. JS: contributed to the DFT calculation. KX: contributed to the DFT calculation. WL: contributed to the the fluorescence spectrum test and analysis. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics Approval
For this type of study, the ethical approval was not required, because this study does not involve animal manipulation.
Consent to Participate
All authors gave their consent to participate in the research.
Consent for Publication
All authors gave their consent to participate in the publication of the research.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Cheng, X., Zhang, R., Sun, J. et al. Acylhydrazone Functionalized Triphenylamine-Based Fluorescent Probe for Cu2+: Tunable Structures of Conjugated Bridge and Its Practical Application. J Fluoresc (2023). https://doi.org/10.1007/s10895-023-03492-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10895-023-03492-w