Abstract
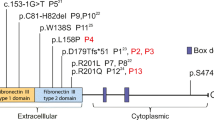
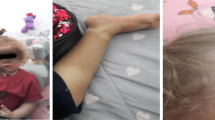
Autosomal recessive IL-1R-associated kinase 4 (IRAK-4) deficiency is a rare cause of recurrent pyogenic infections with limited inflammatory responses. We describe an adult female patient with severe lung disease who was phenotypically diagnosed as suffering from autosomal dominant Hyper IgE syndrome (AD HIES) because of recurrent skin infections with Staphylococcus aureus, recurrent pneumonia and elevated serum IgE levels. In contrast to findings in AD HIES patients, no abnormalities were found in the Th17 and circulating follicular helper T cell subsets. A panel-based sequencing approach led to the identification of a homozygous IRAK4 stop mutation (c.877C > T, p.Gln293*).


Similar content being viewed by others
References
Picard C, Puel A, Bonnet M, Ku CL, Bustamante J, Yang K, et al. Pyogenic bacterial infections in humans with IRAK-4 deficiency. Science. 2003;299(5615):2076–9.
Von Bernuth H, Picard C, Jin Z, Pankla R, Xiao H, Ku CL, et al. Pyogenic bacterial infections in humans with MyD88 deficiency. Science. 2008;321:691–6. doi:10.1126/science.1158298.
Picard C, von Bernuth H, Ghandil P, Chrabieh M, Levy O, Arkwright PD, et al. Clinical features and outcome of patients with IRAK-4 and MyD88 deficiency. Medicine (Baltimore). 2010;89(6):403–25. doi:10.1097/MD.0b013e3181fd8ec3.
Ku CL, von Bernuth H, Picard C, Zhang SY, Chang HH, Yang K, et al. Selective predisposition to bacterial infections in IRAK-4-deficient children: IRAK-4-dependent TLRs are otherwise redundant in protective immunity. J Exp Med. 2007;204(10):2407–22.
Maglione PJ, Simchoni N, Black S, Radigan L, Overbey JR, Bagiella E, et al. IRAK-4 and MyD88 deficiencies impair IgM responses against T-independent bacterial antigens. Blood. 2014;124(24):3561–71. doi:10.1182/blood-2014-07-587824.
Weller S, Bonnet M, Delagreverie H, Israel L, Chrabieh M, Maródi L, et al. IgM + IgD + CD27+ B cells are markedly reduced in IRAK-4-, MyD88-, and TIRAP- but not UNC-93B-deficient patients. Blood. 2012;120(25):4992–5001. doi:10.1182/blood-2012-07-440776.
Grimbacher B, Schäffer AA, Holland SM, Davis J, Gallin JI, Malech HL, et al. Genetic linkage of hyper-IgE syndrome to chromosome 4. Am J Hum Genet. 1999;65(3):735–44.
Grimbacher B, Holland SM, Gallin JI, Greenberg F, Hill SC, Malech HL, et al. Hyper-IgE syndrome with recurrent infections--an autosomal dominant multisystem disorder. N Engl J Med. 1999;340(9):692–702.
Minegishi Y, Saito M, Tsuchiya S, Tsuge I, Takada H, Hara T, et al. Dominant-negative mutations in the DNA-binding domain of STAT3 cause hyper-IgE syndrome. Nature. 2007;448(7157):1058–62.
Holland SM, DeLeo FR, Elloumi HZ, Hsu AP, Uzel G, Brodsky N, et al. STAT3 mutations in the hyper-IgE syndrome. N Engl J Med. 2007;357(16):1608–19.
Chandesris MO, Melki I, Natividad A, Puel A, Fieschi C, Yun L, et al. Autosomal dominant STAT3 deficiency and hyper-IgE syndrome: molecular, cellular, and clinical features from a French national survey. Medicine (Baltimore). 2012;91(4):e1–19. doi:10.1097/MD.0b013e31825f95b9.
Woellner C, Gertz EM, Schäffer AA, Lagos M, Perro M, Glocker EO, et al. Mutations in STAT3 and diagnostic guidelines for hyper-IgE syndrome. J Allergy Clin Immunol. 2010;125(2):424–32. doi:10.1016/j.jaci.2009.10.059.
Ma CS, Deenick EK. Human T follicular helper (TFH) cells and disease. Immunol Cell Biol. 2014;92(1):64–71. doi:10.1038/icb.2013.55.
Morbach H, Eichhorn EM, Liese JG, Girschick HJ. Reference values for B cell subpopulations from infancy to adulthood. Clin Exp Immunol. 2010;162(2):271–9. doi:10.1111/j.1365-2249.2010.04206.x.
Medvedev AE, Lentschat A, Kuhns DB, Blanco JC, Salkowski C, Zhang S, et al. Distinct mutations in IRAK-4 confer hyporesponsiveness to lipopolysaccharide and interleukin-1 in a patient with recurrent bacterial infections. J Exp Med. 2003;198(4):521–31.
Kurosaki T, Kometani K, Ise W. Memory B cells. Nat Rev Immunol. 2015. doi:10.1038/nri3802.
de Beaucoudrey L, Puel A, Filipe-Santos O, Cobat A, Ghandil P, Chrabieh M, et al. Mutations in STAT3 and IL12RB1 impair the development of human IL-17-producing T cells. J Exp Med. 2008;205(7):1543–50. doi:10.1084/jem.20080321.
Moens LN, Falk-Sörqvist E, Asplund AC, Bernatowska E, Smith CI, Nilsson M. Diagnostics of primary immunodeficiency diseases: a sequencing capture approach. PLoS One. 2014;9(12), e114901. doi:10.1371/journal.pone.0114901.
Nijman IJ, van Montfrans JM, Hoogstraat M, Boes ML, van de Corput L, Renner ED, et al. Targeted next-generation sequencing: a novel diagnostic tool for primary immunodeficiencies. J Allergy Clin Immunol. 2014;133(2):529–34. doi:10.1016/j.jaci.2013.08.032.
Stoddard JL, Niemela JE, Fleisher TA, Targeted RSD, NGS. A cost-effective approach to molecular diagnosis of PIDs. Front Immunol. 2014;5:531. doi:10.3389/fimmu.2014.00531.
Tewfik MA, Bossé Y, Lemire M, Hudson TJ, Vallée-Smejda S, Al-Shemari H, et al. Polymorphisms in interleukin-1 receptor-associated kinase 4 are associated with total serum IgE. Allergy. 2009;64(5):746–53. doi:10.1111/j.1398-9995.2008.01889.x.
Zhang Y, Lin X, Desrosiers M, Zhang W, Meng N, Zhao L, et al. Association pattern of interleukin-1 receptor-associated kinase-4 gene polymorphisms with allergic rhinitis in a Han Chinese population. PLoS One. 2011;6(6), e21769. doi:10.1371/journal.pone.0021769.
Avery DT, Ma CS, Bryant VL, Santner-Nanan B, Nanan R, Wong M, et al. STAT3 is required for IL-21-induced secretion of IgE from human naive B cells. Blood. 2008;112(5):1784–93. doi:10.1182/blood-2008-02-142745.
Acknowledgments
This work was supported by a GOA grant from the Research Council of the Catholic University of Leuven, Belgium. HS receives research support from the Research Foundation Flanders (FWO). RS and IM are supported by a KOF mandate of the KU Leuven, Belgium.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Author contributions
GF drafted the manuscript and conducted experiments. LM conducted experiments and aided in preparing the manuscript and the HIES work-up. GW conducted the TLR testing. RS, BB, HS, and LD participated in the clinical care of the patient. XB supervised the routine laboratory immunology work-up and TLR testing. AC performed the genetic analysis and established the genetic diagnosis. IM drafted and finalized the manuscript, characterized the immune deficiency and is coordinating clinical care for the patient. Each author has critically revised the final version of the manuscript and has read and approved the final manuscript.
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
The study was performed in accordance with the 1964 Helsinki declaration and its later amendments. Informed consent was obtained for genetic analysis and report of the case. The study was approved by the Ethics Committee of UZ Leuven.
Additional information
Xavier Bossuyt, Anniek Corveleyn and Isabelle Meyts contributed equally to this work.
Rights and permissions
About this article
Cite this article
Frans, G., Moens, L., Schrijvers, R. et al. PID in Disguise: Molecular Diagnosis of IRAK-4 Deficiency in an Adult Previously Misdiagnosed With Autosomal Dominant Hyper IgE Syndrome. J Clin Immunol 35, 739–744 (2015). https://doi.org/10.1007/s10875-015-0205-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10875-015-0205-x