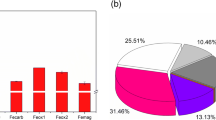
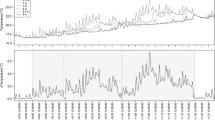
Abstract
In the western subarctic Pacific (WSP), iron (Fe) can enhance the biological carbon pump by stimulating phytoplankton photosynthesis. However, little is known about how Fe and light availability controls the phytoplankton photophysiology in the WSP near the Kuril Islands, where water mixing is sometimes enhanced, even in summer. Here, we conducted on-deck Fe-manipulated incubation experiments at two stations where surface mixed layer depths were distinct but showed similar initial macronutrient and dissolved Fe concentrations and phytoplankton community composition even experienced light availability for phytoplankton in the water column could be different. An Fe addition to water samples from the deeper surface-mixed layer enhanced the phytoplankton biomass and the photosynthetic competence in photosystem II (Fv/Fm), suggesting an Fe-light co-limitation, which was supported by the results from high functional absorption cross section of photosystem II (σPSII) and low light saturation index (Ek). At the shallower mixed layer station, Fe amendment did not stimulate phytoplankton biomass and photosynthesis, indicating that Fe was sufficient for the phytoplankton. Although the centric diatom Chaetoceros species were predominant at both stations throughout incubation, the pennate diatoms Pseudo-nitzschia spp. and Cylindrotheca closterium significantly increased at the deeper and shallower-mixed layer stations, respectively, after Fe enrichment. The elongation of the pennate diatom Neodenticula seminae chains was observed under low Fe availability caused by a chelator. This could be related to the fact that N. seminae is abundant in sediment trap samples from the WSP under Fe-limited conditions and contributed to the high biological pump efficiency.









Similar content being viewed by others
Data availability
The data that supports the findings of this study are available in the supplementary material of this article. The original data from our published articles, which was cited and analyzed during this study, are available on request from the corresponding authors.
References
Alderkamp AC, Van Dijken GL, Lowry KE, Lewis KM, Joy-Warren HL, Van De Poll W, Laan P, Gerringa L, Delmont TO, Jenkins BD, Arrigo KR (2019) Effects of iron and light availability on phytoplankton photosynthetic properties in the Ross Sea. Mar Eco Prog Ser 621:33–50. https://doi.org/10.3354/meps13000
Behrenfeld MJ, Halsey KH, Boss E, Karp-Boss L, Milligan AJ, Peers G (2021) Thoughts on the evolution and ecological niche of diatoms. Ecol Monogr 91: e01457. https://doi.org/10.1002/ecm.1457Behrenfeld MJ, Milligan AJ (2013) Photophysiological expressions of iron stress in phytoplankton. Ann Rev Mar Sci 5: 217–246. https://doi.org/10.1146/annurev-marine-121211-172356
Bouman HA, Platt T, Doblin M, Figueiras FG, Gudmundsson K, Gudfinnsson HG, Huang B, Hickman A, Hiscock M, Jackson T, Lutz VA, Mélin F, Rey F, Pepin P, Segura V, Tilstone GH, Van Dongen-Vogels V, Sathyendranath S (2018) Photosynthesis-irradiance parameters of marine phytoplankton: synthesis of a global data set. Earth Syst Sci Data 10:251–266. https://doi.org/10.5194/essd-10-251-2018
Boyd PW, Jickells T, Law CS, Blain S, Boyle EA, Buesseler KO, Coale KH, Cullen JJ, de Baar HJW, Follows M, Harvey M, Lancelot C, Levasseur M, Owens NPJ, Pollard R, Rivkin RB, Sarmiento J, Schoemann V, Smetacek V, Takeda S, Tsuda A, Turner S, Watson AJ (2007) Mesoscale iron enrichment experiments 1993–2005: synthesis and future directions. Science 315:612–617. https://doi.org/10.1126/science.1131669
Browning T, Achterberg E, Rapp I, Engel A, Bertrand EM, Tagliabue A, Moore CM (2017) Nutrient co-limitation at the boundary of an oceanic gyre. Nature 551:242–246. https://doi.org/10.1038/nature24063
Camoying MG, Thoms S, Geuer JK, Koch BP, Bischof K, Trimborn S (2022) In contrast to diatoms, cryptophytes are susceptible to iron limitation, but not ocean acidification. Physiol Plant 174:e13614. https://doi.org/10.1111/ppl.13614
de Baar HJW, Boyd PW, Coale KH, Landry MR, Tsuda A, Assmy P, Bakker DCE, Bozec Y, Barber RT, Brzezinski MA, Buesseler KO, Boyé M, Croot PL, Gervais F, Gorbunov MY, Harrison PJ, Hiscock WT, Laan P, Law LC, CS, Levasseur M, Marchetti A, Millero FJ, Nishioka J, Nojiri Y, van Oijen T, Riebesell U, Rijkenberg MJA, Saito H, Takeda S, Timmermans KR, Veldhuis MJW, Waite AM, Wong CS, (2005) Synthesis of iron fertilization experiments: from the iron age in the age of enlightenment. J Geophys Res C Ocean 110:1–24. https://doi.org/10.1029/2004JC002601
D’Haene SE, Sobotka R, Bučinská L, Dekker JP, Komenda J (2015) Interaction of the PsbH subuni twith a chlorophyll bound to histidine 114 of CP47 is responsible for the red 77 K fluorescence of Photosystem II. Biochim Biophys Acta 1847:1327–1334. https://doi.org/10.1016/j.bbabio.2015.07.003
Endo H, Yoshimura T, Kataoka T, Suzuki K (2013) Effects of CO2 and iron availability on phytoplankton and eubacterial community compositions in the northwest subarctic Pacific. J Exp Mar Bio Ecol 439:160–175. https://doi.org/10.1016/j.jembe.2012.11.003
Fujiki T, Matsumoto K, Mino Y, Sasaoka K, Wakita M, Kawakami H, Honda MC, Watanabe S, Saino T (2014) Seasonal cycle of phytoplankton community structure and photophysiological state in the western subarctic gyre of the North Pacific. Limnol Oceanogr 59:887–900. https://doi.org/10.4319/lo.2014.59.3.0887
Geider RJ, LaRoche J (1994) The role of iron in phytoplankton photosynthesis, and the potential for iron-limitation of primary productivity in the sea. Photosynth Res 39:275–301
Goto Y, Yasuda I, Nagasawa M (2016) Turbulence estimation using fast-response thermistors attached to a free-fail vertical microstructure profiler. J Atmos Ocean Technol 33:2065–2078. https://doi.org/10.1175/JTECH-D-15-0220.1
Goto Y, Yasuda I, Nagasawa M (2018) Comparison of turbulence intensity from CTD-attached and free-fall microstructure profilers. J Atmos Ocean Technol 35:147–162. https://doi.org/10.1175/JTECH-D-17-0069.1
Goto Y, Yasuda I, Nagasawa M, Kouketsu S, Nakano T (2021) Estimation of basin-scale turbulence distribution in the North Pacific Ocean using CTD-attached thermistor measurements. Sci Rep 11:1–13. https://doi.org/10.1038/s41598-020-80029-2
Greene RM, Geider RJ, Kolber Z, Falkowski PG (1992) Iron-induced changes in light harvesting and photochemical energy conversion processes in eukaryotic marine algae. Plant Physiol 100:565–575. https://doi.org/10.1104/pp.100.2.565
Hama T, Miyazaki T, Ogawa Y, Iwakuma T, Takahashi M, Otsuki A, Ichimura S (1983) Measurement of photosynthetic production of a marine phytoplankton population using a stable 13C isotope. Mar Biol 73:31–36
Harrison PJ, Boyd PW, Varela DE, Takeda S, Shiomoto A, Odate T (1999) Comparison of factors controlling phytoplankton productivity in the NE and NW subarctic Pacific gyres. Prog Oceanogr 43:205–234. https://doi.org/10.1016/S0079-6611(99)00015-4
Hattori-Saito A, Nishioka J, Ono T, McKay RML, Suzuki K (2010) Iron deficiency in micro-sized diatoms in the Oyashio region of the Western subarctic Pacific during spring. J Oceanogr 66:105–115. https://doi.org/10.1007/s10872-010-0009-9
Honda MC (2003) Biological pump in northwestern North Pacific. J Oceanogr 59:671–684. https://doi.org/10.1023/B:JOCE.0000009596.57705.0c
Hooker SB, Morrow JH, Matsuoka A (2013) Apparent optical properties of the Canadian Beaufort Sea—Part 2: the 1% and 1 cm perspective in deriving and validating AOP data products. Biogeosciences 10:4511–4527. https://doi.org/10.5194/bg-10-4511-2013
Hooker SB, Lind RN, Morrow JW, Kudela RM, Housekeeper HF, Suzuki K (2018) Advances in above- and in-water radiometry. Autonomous atmospheric and Oceanic Observing Systems, vol 2. NASA Goddard Space Flight Center, Greenbelt, Maryland, p 69
Hutchins DA, Franck VM, Brzezinski MA, Bruland KW (1999) Inducing phytoplankton iron limitation in iron-replete coastal waters with a strong chelating ligand. Limnol Oceanogr 44:1009–1018. https://doi.org/10.4319/lo.1999.44.4.1009
Kaneko H, Yasuda I, Komatsu K, Itoh S (2013) Observations of vertical turbulent nitrate flux across the Kuroshio. Geophys Res Lett 40:3123–3127. https://doi.org/10.1002/grl.50613
Kaneko H, Itoh S, Kouketsu S, Okunishi T, Hododa S, Suga T (2015) Evolution and modulation of a poleward-propagating anticyclonic eddy along the Japan and Kuril-Kamchatka Trench. J Geophys Res Ocean 120:4418–4440. https://doi.org/10.1088/1751-8113/44/8/085201
Katsuki K, Takahashi K (2005) Diatoms as paleoenvironmental proxies for seasonal productivity, sea-ice and surface circulation in the Bering Sea during the late quaternary. Deep Res II 52:2110–2130. https://doi.org/10.1016/j.dsr2.2005.07.001
Katsuki K, Takahashi K, Okada M (2003) Diatom assemblage and productivity changes during the last 340,000 years in the subarctic Pacific. J Oceanogr 59:695–707. https://doi.org/10.1023/B:JOCE.0000009598.93075.78
Kawakami H, Yang YL, Honda MC, Kusakabe M (2004) Particulate organic carbon fluxes estimated from 234Th deficiency in winters and springs in the northwestern North Pacific. Geochem J 38:581–592. https://doi.org/10.2343/geochemj.38.581
Kawakami H, Honda MC, Matsumoto K, Wakita M, Kitamura M, Fujiki T, Watanabe S (2015) POC fluxes estimated from 234Th in late spring-early summer in the western subarctic North Pacific. J Oceanogr 71:311–324. https://doi.org/10.1007/s10872-015-0290-8
Kirk JT (2010) Light and photosynthesis in aquatic ecosystems, 3rd edn. Cambridge University Press, Cambridge
Kolber ZS, Prášil O, Falkowski PG (1998) Measurements of variable chlorophyll fluorescence using fast repetition rate techniques: defining methodology and experimental protocols. Biochim Biophys Acta 1367:88–106. https://doi.org/10.1016/S0005-2728(98)00135-2
Kondo Y, Takeda S, Nishioka J, Sato M, Saito H, Suzuki K, Furuya K (2013) Growth stimulation and inhibition of natural phytoplankton communities by model organic ligands in the western subarctic Pacific. J Oceanogr 69:97–115. https://doi.org/10.1007/s10872-012-0160-6
Konno S, Ohira R, Harada N, Jordan RW (2007) Six new taxa of subarctic Parmales (Chrysophyceae). J Nannoplankt Res 29:108–128
Kudo I, Noiri Y, Nishioka J, Taira Y, Kiyosawa H, Tsuda A (2006) Phytoplankton community response to Fe and temperature gradients in the NE (SERIES) and NW (SEEDS) subarctic Pacific Ocean. Deep-Sea Res II 53:2201–2213. https://doi.org/10.1016/j.dsr2.2006.05.033
MacIntyre HL, Kana TM, Anning T, Geider RJ (2002) Photoacclimation of photosynthesis irradiance response curves and photosynthetic pigments in microalgae and cyanobacteria. J Phycol 38:17–38. https://doi.org/10.1046/j.1529-8817.2002.00094.x
Maldonado MT, Price NM (1999) Utilization of Fe bound to strong organic ligands by phytoplankton communities in the subarctic Pacific Ocean. Deep Res II 46:2447–2473. https://doi.org/10.1016/S0967-0645(99)00071-5
Maldonado MT, Boyd PW, Harrison PJ, Price NM (1999) Co-limitation of phytoplankton growth by light and Fe during winter in the NE subarctic Pacific Ocean. Deep Res Part II 46:2475–2485. https://doi.org/10.1016/S0967-0645(99)00072-7
Maranón E, Cermeno P, Latasa M, Tadonléké RD (2012) Temperature, resources, and phytoplankton size structure in the ocean. Limnol Oceanogr 57:1266–1278. https://doi.org/10.4319/lo.2012.57.5.1266
Maranón E, Cermeno P, Latasa M, Tadonléké RD (2015) Resource supply alone explains the variability of marine phytoplankton size structure. Limnol Oceanogr 60:1848–1854. https://doi.org/10.1002/lno.10138
McKay RML, Geider RJ, LaRoche J (1997) Physiological and biochemical response of the photosynthetic apparatus of two marine diatoms to Fe stress. Plant Physiol 114:615–622. https://doi.org/10.1104/pp.114.2.615
Medvedeva LA, Nikulina TV (2014) Catalogue of Freshwater Algae of the southern part of the Russian Far East. Vladivostok Dalnauka, Vladivostok
Monterey G, Levitus S (1997) Seasonal variability of mixed layer depth for the World Ocean. NOAA Atlas NESDIS, vol 14. US Government Printing Office, Washington DC, p 102
Nishioka J, Obata H (2017) Dissolved iron distribution in the western and central subarctic Pacific: HNLC water formation and biogeochemical processes. Limnol Oceanogr 62:2004–2022. https://doi.org/10.1002/lno.10548
Nishioka J, Takeda S, Wong CS, Johnson WK (2001) Size-fractionated iron concentrations in the northeast Pacific Ocean: distribution of soluble and small colloidal iron. Mar Chem 74:157–179. https://doi.org/10.1016/S0304-4203(01)00013-5
Nishioka J, Ono T, Saito H, Nakatsuka T, Takeda S, Yoshimura T, Suzuki K, Kuma K, Nakabayashi S, Tsumune D, Mitsudera H, Johnson WK, Tsuda A (2007) Iron supply to the western subarctic Pacific importance of iron export from the Sea of Okhotsk. J Geophys Res Ocean. https://doi.org/10.1029/2006JC004055
Nishioka J, Nakatsuka T, Watanabe YW, Yasuda I, Kuma K, Ogawa H, Ebuchi N, Scherbinin A, Volkov YN, Shiraiwa T, Wakatsuchi M (2013) Intensive mixing along an island chain controls oceanic biogeochemical cycles. Global Biogeochem Cycles 27:920–929. https://doi.org/10.1002/gbc.20088
Nishioka J, Nakatsuka T, Ono K, Volkov YN, Scherbinin A, Shiraiwa T (2014) Quantitative evaluation of iron transport processes in the Sea of Okhotsk. Prog Oceanogr 126:180–193. https://doi.org/10.1016/j.pocean.2014.04.011
Nishioka J, Obata H, Ogawa H, Ono K, Yamashita Y, Lee K, Takeda S, Yasuda I (2020) Subpolar marginal seas fuel the North Pacific through the intermediate water at the termination of the global ocean circulation. Proc Natl Acad Sci U S A 117:12665–12673. https://doi.org/10.1073/pnas.2000658117
Nishioka J, Hirawake T, Nomura D, Yamashita Y, Ono K, Murayama A, Shcherbinin A, Volkov YN, Mitsudera H, Ebuchi N, Wakatsuchi M, Yasuda I (2021) Iron and nutrient dynamics along the East Kamchatka current western bering Sea Basin and Gulf of Anadyr. Prog Oceanogr 198:102662. https://doi.org/10.1016/j.pocean.2021.102662
Nosaka Y, Isada T, Kudo I, Saito H, Hattori H, Tsuda A, Suzuki K (2014) Light utilization efficiency of phytoplankton in the Western subarctic gyre of the North Pacific during summer. J Oceanogr 70:91–103. https://doi.org/10.1007/s10872-013-0217-1
Nosaka Y, Yamashita Y, Suzuki K (2017) Dynamics and origin of transparent exopolymer particles in the Oyashio region of the western subarctic Pacific during the spring diatom bloom. Front Mar Sci 4:1–16. https://doi.org/10.3389/fmars.2017.00079
Obata H, Karatani H, Nakayama E (1993) Automated determination of iron in seawater by chelating resin concentration and chemiluminescence detection. Anal Chem 65:1524–1528. https://doi.org/10.1021/ac00059a007
Obata H, Karatani H, Matsui M, Nakayama E (1997) Fundamental studies for chemical speciation of iron in seawater with an improved analytical method. Mar Chem 56:97–106
Ono K, Ohshima KI, Kono T, Itoh M, Katsumata K, Volkov YN, Wakatsuchi M (2007) Water mass exchange and diapycnal mixing at Bussol’ strait revealed by water mass properties. J Oceanogr 63:281–291. https://doi.org/10.1007/s10872-007-0028-3
Onodera J, Takahashi K, Honda MC (2005) Pelagic and coastal diatom fluxes and the environmental changes in the northwestern North Pacific during December 1997-May 2000. Deep Res II 52:2218–2239. https://doi.org/10.1016/j.dsr2.2005.07.005
Osborn TR (1980) Estimates of the local rate of vertical diffusion from dissipation measurements. J Phys Oceanogr 10:83–89
Osborn TR, Cox CS (1972) Oceanic fine structure. Geophys Fluid Dyn 3:321–345
Platt T, Gallegos CL, Harrison WG (1980) Photoinhibition of photosynthesis in natural assemblages of marine phytoplankton. J Mar Res 38:687–701
Raven JA (1995) Scaling the seas. Plant Cell Environ 18:1090–1100. https://doi.org/10.1111/j.1365-3040.1995.tb00621.x
Round FE, Crawford RM, Mann DG (2007) The diatom: biology & morphology of the genera. Cambridge University Press, Cambridge
Sakshaug E, Bricaud A, Dandonneau Y, Falkowski PG, Kiefer DA, Legendre L, Morel A, Parslow J, Takahashi M (1997) Parameters of photosynthesis: definitions, theory and interpretation of results. J Plankton Res 19:1637–1670. https://doi.org/10.1093/plankt/19.11.1637
Sato H, Takeda S, Furuya K (2009) Responses of pico- and nanophytoplankton to artificial iron infusions observed during the second iron enrichment experiment in the western subarctic Pacific (SEEDS II). Deep-Sea Res II 56:2745–2754. https://doi.org/10.1016/j.dsr2.2009.06.002
Schaeffer BA, Hein KJC, MK, (2012) Phytoplankton community composition in nearshore coastal waters of Louisiana. Mar Pollut Bull 64:1705–1712. https://doi.org/10.1016/j.marpolbul.2012.03.017
Shaked Y, Twining BS, Tagliabue A, Maldonado MT (2021) Probing the bioavailability of dissolved iron to marine eukaryotic phytoplankton using in situ single cell iron quotas. Glob Biogeochem Cycles 35:e2021GB006979. https://doi.org/10.1029/2021GB006979
Sicko-Goad LM, Schelske CL, Stoermer EF (1984) Estimation of intracellular carbon and silica content of diatoms from natural assemblages using morphometric techniques. Limnol Oceanogr 29:1170–1178. https://doi.org/10.4319/lo.1984.29.6.1170
Strzepek RF, Hunter KA, Frew RD, Harrison PJ, Boyd PW (2012) Iron-light interactions differ in Southern Ocean phytoplankton. Limnol Oceanogr 57:1182–1200. https://doi.org/10.4319/lo.2012.57.4.1182
Strzepek RF, Boyd PW, Sunda WG (2019) Photosynthetic adaptation to low iron, light, and temperature in Southern Ocean phytoplankton. Proc Natl Acad Sci U S A 116:4388–4393. https://doi.org/10.1073/pnas.1810886116
Suggett DJ, Moore CM, Hickman AE, Geider RJ (2009) Interpretation of fast repetition rate (FRR) fluorescence: signatures of phytoplankton community structure versus physiological state. Mar Ecol Prog Ser 376:1–19. https://doi.org/10.3354/meps07830
Sugie K, Kuma K, Fujita S, Ushizaka S, Suzuki K, Ikeda T (2011) Importance of intracellular Fe pools on growth of marine diatoms by using unialgal cultures and on the Oyashio region phytoplankton community during spring. J Oceanogr 67:183–196. https://doi.org/10.1007/s10872-011-0017-4
Sugie K, Nishioka J, Kuma K, Volkov YN, Nakatsuk T (2013) Availability of particulate Fe to phytoplankton in the Sea of Okhotsk. Mar Chem 152:20–31. https://doi.org/10.1016/j.marchem.2013.03.005
Sunda WG, Huntsman SA (1995) Iron uptake and growth limitation in oceanic and coastal phytoplankton. Mar Chem 50:189–206
Sunda WG, Huntsman SA (1997) Interrelated influence of iron, light and cell size on marine phytoplankton growth. Nature 390:389–392. https://doi.org/10.1038/37093
Suzuki K, Minami C, Liu H, Saino T (2002) Temporal and spatial patterns of chemotaxonomic algal pigments in the subarctic Pacific and the Bering Sea during the early summer of 1999. Deep Res II 49:5685–5704. https://doi.org/10.1016/S0967-0645(02)00218-7
Suzuki K, Hinuma A, Saito H, Kiyosawa H, Liu H, Saino T, Tsuda A (2005) Responses of phytoplankton and heterotrophic bacteria in the northwest subarctic Pacific to in situ iron fertilization as estimated by HPLC pigment analysis and flow cytometry. Prog Oceanogr 64:167–187. https://doi.org/10.1016/j.pocean.2005.02.007
Suzuki K, Saito H, Isada T, Hattori-Saito A, Kiyosawa H, Nishioka J, McKay RML, Kuwata A, Tsuda A (2009) Community structure and photosynthetic physiology of phytoplankton in the northwest subarctic Pacific during an in situ iron fertilization experiment (SEEDS-II). Deep Res II 56:2733–2744. https://doi.org/10.1016/j.dsr2.2009.06.001
Suzuki K, Hattori-Saito A, Sekiguchi Y, Nishioka J, Shigemitsu M, Isada T, Liu H, McKay RML (2014) Spatial variability in iron nutritional status of large diatoms in the Sea of Okhotsk with special reference to the Amur River discharge. Biogeosciences 11:2503–2517. https://doi.org/10.5194/bg-11-2503-2014
Taguchi S (1976) Relationship between photosynthesis and cell size of marine diatoms. J Phycol 12:185–189. https://doi.org/10.1111/j.1529-8817.1976.tb00499.x
Takahashi T, Sutherland SC, Sweeney C, Poisson A, Metzl N, Tilbrook B, Bates N, Wanninkhof R, Feely RA, Sabine C, Olafsson J, Nojiri Y (2002) Global sea–air CO2 flux based on climatological surface ocean pCO2, and seasonal biological and temperature effects. Deep Res II 49:1601–1622. https://doi.org/10.1016/S0967-0645(02)00003-6
Takeda S, Obata H (1995) Response of equatorial Pacific phytoplankton to subnanomolar Fe enrichment. Mar Chem 50:219–227. https://doi.org/10.1016/0304-4203(95)00037-R
Timmermans KR, Davey MS, van der Wagt B, Snoek J, Geider RJ, Veldhuis MJW, Gerringa LJA, de Baar HJW (2001) Co-limitation by iron and light of Chaetoceros brevis, C. dichaeta and C. calcitrans (Bacillariophyceae). Mar Ecol Prog Ser 217:287–297. https://doi.org/10.3354/meps217287
Tomas CR (1997) Identifying marine phytoplankton. Academic Press, London
Tréguer P, Bowler C, Moriceau B, Dutkiewicz S, Gehlen M, Aumont O, Bittner L, Dugdale R, Finkel Z, Iudicone D, Jahn O, Guidi L, Lasbleiz M, Leblanc K, Levy M, Pondaven P (2018) Influence of diatom diversity on the ocean biological carbon pump. Nat Geosci 11:27–37. https://doi.org/10.1038/s41561-017-0028-x
Tsuda A, Takeda S, Saito H, Nishioka J, Nojiri Y, Kudo I, Kiyosawa H, Shiomoto A, Imai K, Ono T, Shimamoto A, Tsumune D, Yoshimura T, Aono T, Hinuma A, Kinugasa M, Suzuki K, Sohrin Y, Noiri Y, Tani H, Deguchi Y, Tsurushima N, Ogawa H, Fukami K, Kuma K, Saino T (2003) A mesoscale iron enrichment in the western subarctic Pacific induces a large centric diatom bloom. Science 300:958–961. https://doi.org/10.1126/science.1082000
Twining BS, Baines SB (2013) The trace metal composition of marine phytoplankton. Ann Rev Mar Sci 5:191–215. https://doi.org/10.1146/annurev-marine-121211-172322
Wahl RJ, Teague WJ (1983) Estimation of Brunt-Väisälä frequency from temperature profiles. J Phys Oceanogr 13:2236–2240. https://doi.org/10.1175/1520-0485(1983)013%3c2236:EOBVFF%3e2.0.CO;2
Wells ML (1999) Manipulating iron availability in nearshore waters. Limnol Oceanogr 44:1002–1008
Yagi M, Yasuda I (2012) Deep intense vertical mixing in the Bussol’ Strait. Geophys Res Lett 39:1–5. https://doi.org/10.1029/2011GL050349
Yasuda I (2000) Cold-core anticyclonic eddies south of the Bussol’ Strait in the Northwestern subarctic Pacific. J Phys Oceanogr 30:1137–1157
Yasuda I, Fujio S, Yanagimoto D, Lee KJ, Sasaki Y, Zhai S, Tanaka M, Itoh S, Tanaka T, Hasegawa D, Goto Y, Sasano D (2021) Estimate of turbulent energy dissipation rate using free-fall and CTD-attached fast-response thermistors in weak ocean turbulence. J Oceanogr 77:17–28. https://doi.org/10.1007/s10872-020-00574-2
Yoshida K, Endo H, Lawrenz E, Isada T, Hooker SB, Prášil O, Suzuki K (2018) Community composition and photophysiology of phytoplankton assemblages in coastal Oyashio waters of the western North Pacific during early spring. Estuar Coast Sci 218(218):80–94. https://doi.org/10.1016/j.ecss.2018.06.018
Yoshida K, Nakamura S, Nishioka J, Hooker SB, Suzuki K (2020) Community composition and photosynthetic physiology of phytoplankton in the western subarctic Pacific near the Kuril Islands with special reference to iron availability. J Geophys Res Biogeosciences 125:e2019JG005525. https://doi.org/10.1029/2019JG005525
Yoshimura T, Nishioka J, Nakatsuka T (2010) Iron nutritional status of the phytoplankton assemblage in the Okhotsk Sea during summer. Deep Res I 57:1454–1464. https://doi.org/10.1016/j.dsr.2010.08.003
Acknowledgements
We thank the officers and crew of R/V Professor Multanovskiy. Thanks also extended to Dr. Y. N. Volkov and the late Mr. Scherbinin, the Far Eastern Regional Hydrometeorological Research Institute, for cooperation under the Japanese-Russian joint research program. We acknowledge Ms. Suzu Nakamura, Ms. Natsuko Araki, and Ms. Aiko Murayama for their shipboard and on-land assistance. This study was partly supported by the JSPS Grant‐in‐Aid for Scientific Research (JP17H00775, JP18H03352, JP21H05056, JP22H05205, JP21H04921, JP23H03516) funded to JN, KS, and IY, and the Joint Research Program of the Institute of Low Temperature Science, Hokkaido University, and JAXA 2nd Research Announcement on the Earth Observations, Japan Aerospace Exploration Agency (ER2GCF304). KY was partly supported by the Sasakawa Scientific Research Grant from The Japan Science Society (27‐752).
Funding
Japan Society for the Promotion of Science London, JP17H00775,Jun Nishioka, JP18H03352,Koji Suzuki, JP21H05056, Jun Nishioka, JP22H05205, Jun Nishioka, JP21H04921, Ichiro Yasuda, JP23H03516, Koji Suzuki, Japan Aerospace Exploration Agency, ER2GCF304,Koji Suzuki, Japan Foundation, 27‐752, Kazuhiro Yoshida.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflicts of interest
The authors declare that they have no conflicts of interest.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yoshida, K., Nishioka, J., Yasuda, I. et al. Different responses of phytoplankton to Fe manipulation in Fe-limited waters with contrasting surface mixed layer depths in the western subarctic Pacific. J Oceanogr 79, 483–497 (2023). https://doi.org/10.1007/s10872-023-00692-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10872-023-00692-7