Abstract
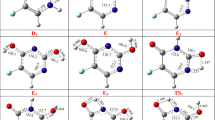
Variable temperature NMR spectroscopic measurements on (S)-warfarin [open-form: 3-(1′-phenyl-3′-oxobut-1′-yl)-4-hydroxycoumarin] in CDCl3, CD3OD and d6-DMSO generally showed tautomeric compositions in the order trans (2S,4S) coumarin hemiketal > cis (2R,4S) coumarin hemiketal > open (S) coumarin enol in slow dynamic equilibrium over temperature ranges rising modestly from ambient. A computational (DFT-M06-2X) examination of the lower energy tautomers including coumarin and chromone open and cyclic forms (gas phase, chloroform or DMSO fields) was consistent with the general solution compositions. The crystal and molecular structures for model compounds of the major solution tautomers are reported: (2S,4S)-warfarin methyl ketal [orthorhombic, P212121], (2R,4S)-warfarin methyl ketal [orthorhombic, P212121], (rac)-warfarin-4-methyl ether [monoclinic, P21/n], and the open chromone (S)-warfarin-2-methyl ether [monoclinic, P21, Z = 8]. A combination of direct integration and line-fitting methods were used to determine solution (S)-warfarin tautomer compositions. As temperatures were increased, the concentrations of the open coumarin form increased at the expense of the cyclic hemiketals. Equilibrium constants were used to determine the standard free-energy differences for the two open-cyclic equilibria (trans hemiketal \(\rightleftharpoons\) open, open \(\rightleftharpoons\) cis hemiketal, respectively) in three solvents: CDCl3 [+ 3.7(4), − 2.8(6) kJ/mol], CD3OD [+ 7.6(16), − 4.7(9) k/mol], d6-DMSO [+ 3.5(7), − 1.1(2) kJ/mol]. Standard enthalpy and entropy differences were also determined from van’t Hoff analysis. Rates of the respective reactions were estimated from line-widths for the cyclic hemiketals and solution equilibrium compositions for each species. Eyring analysis gave ΔG‡, ΔH‡, and ΔS‡, respectively, for the forward and reverse reactions of coumarin trans hemiketal \(\rightleftharpoons\) open-form and for the open-form \(\rightleftharpoons\) cis hemiketal. Negative entropic contributions to the observed transition state energies were consistent with solvent or solute ordering in the prototropic reactions. Open-form NMR signals were broader than could be accounted for by the open-cyclic equilibria alone, increasingly so in polar and protic solvents and with rising temperatures. While a conformational equilibrium may operate, an increasingly faster intermediate dynamic equilibrium between open coumarin-chromone tautomers may be a more likely explanation.
Graphical Abstract
Structures of methylated warfarin tautomers and computational models enabled assignment of overlapping warfarin tautomeric NMR spectra and through variable temperature analysis, provided the thermodynamics of the tautomeric equilibria in three solvents.














Similar content being viewed by others
Data Availability
Crystallographic details for structures 2 (CCDC #2286286), 3 (CCDC #2286287), 4 (CCDC #2286288) and 5 (CCDC #2286289) have been deposited with the Cambridge Crystallographic Data Centre (https://www.ccdc.cam.ac.uk) and are available on request.
References
Majerus PW, Broze GJ, Miletich JP, Tollefsen DM (1996) Anticoagulant, thrombolytic, and antiplatelet drugs. In: Hardman JG, Limbird LE (eds) Goodman and Gilman’s the pharmacological basis of therapeutics, 9th edn. McGraw-Hill, New York, pp 1346–1350
Essential Medicines List (EML) (2021) World Health Organization, 22nd list
Petitpas I, Bhattacharya AA, Twine S, East E, Curry S (2001) Crystal structure analysis of warfarin binding to human serum albumin: anatomy of drug site I. J Biol Chem 276:22804–22809
Wu S, Chen X, Da J-Y, Stafford DW, Pedersen LG, Tie J-K (2018) Warfarin and vitamin K epoxide reductase: a molecular accounting for observed inhibition. Blood 132(6):647–657
Williams PA, Cosme J, Ward A, Angove HC, Vinković DM, Jhoti H (2003) Crystal structure of human cytochrome P450 2C9 with bound warfarin. Nature 424:464–468
Porter WR (2010) Warfarin: history, tautomerism and activity. J Comput Aided Mol Des 24:553–573
Malde AK, Stroet M, Caron B, Visscher KM, Mark AE (2018) Predicting the prevalence of alternative warfarin tautomers in solution. J Chem Theory Comput 14:4405–4415
Nicholls IA, Karlsson BCG, Rosengren AM, Henschel H (2010) Warfarin: an environment-dependent switchable molecular probe. J Mol Recognit 23:604–608
Stella VJ, Mooney KG, Pipkin JD (1984) Dissolution and ionization of warfarin. J Pharm Sci 73:946–948
Vasquez JM, Vu A, Schultz JS, Vullev VI (2009) Fluorescence enhancement of warfarin induced by interaction with beta-cyclodextrin. Biotechnol Prog 25:906–914
Valente EJ, Ruggiero G, Miller CW, Zubkowski JD, Eggleston DS (1997) Diastereomeric discrimination: structural aspects. In: Materials synthesis and characterization, Chapter 2, pp 19–60. Plenum Publishing Co., New York
Bravic G, Gaultier J, Hauw C (1973) Crystal structure of an antivitamin K, warfarin. Compt Rendus Acad Sci Paris Ser C 277(22):1215–1218
Valente EJ, Trager WF, Jensen LH (1975) Crystal and molecular structure and absolute configuration of (-)(S)-warfarin. Acta Crystallogr A B31:954–960
Pisklak M, Maciejewska M, Herold F, Wawer I (2003) Solid state structure of coumarin anticoagulants: warfarin and sintrom. 13C CPMAS NMR and GIAO DFT calculations. J Mol Struct 649:169–176
Valente EJ, Santarsiero BD, Schomaker V (1979) Conformation of dihydropyran rings: the structures of two 3,4-dihydro-2H,5H-pyrano[3,2-c][1]benzopyran-5-ones. J Org Chem 44:798–802
Valente EJ, Hodgson DJ (1980) 2-Methyl-2-hydroxy-4-cyclohexyl-3,4-dihydro-2H,5H-pyrano[3,2 c][1]benzopyran 5 one. Acta Crystallogr A B35:3099–3101
Valente EJ, Schomaker V (1984) A Structure containing diastereomers, (2S,2R)-trans and (2R,4R)-cis 2-hydroxy 2,4 dimethyl 3,4 dihydro-2H,5H-pyrano[3,2 c][1]benzopyran 5 one. Acta Crystallogr A C40:1068–1070
Valente EJ, Eggleston DS, Schomaker V (1986) Structures of five trans-2-hydroxy and methoxy-2-methyl-3,4-dihydro-4-aryl-2H,5H -pyrano[3,2 c][1]benzopyran-5-ones. Acta Crystallogr A C42:1809–1813
Valente EJ, Eggleston DS, Schomaker V (1987) Structures of four trans-2-hydroxy- and methoxy-2-methyl-3,4-dihydro-4-alkyl-2H,5H -pyrano[3,2-c][1]benzopyran-5-ones. Acta Crystallogr A C43:533–536
Castleberry BS, Ruggiero G, Valente EJ, Eggleston DS (1988) Structures of cis-4-(2-methoxyphenyl)- and trans-4-(3-methoxy-4-hydroxyphenyl)-2-hydroxy-2-methyl-3,4- dihydro-2H,5H -pyrano[3,2 c][1]benzopyran 5 one. Acta Crystallogr A C44:1281–1283
Ruggiero G, Valente EJ, Eggleston DS (1989) Structures of (±) cis-2-hydroxy-4-(2-phenylethyl) and cis-4-phenyl-2-methyl-3,4-dihydro-2H,5H -pyrano[3,2 c][1]benzopyran 5 one. Acta Crystallogr A C45:1369–1372
Savell VH Jr, Valente EJ, Eggleston DS (1989) Structures of (±) trans-4-(3-nitrophenyl)- and (+)-trans-4 (4-fluorophenyl)-2-hydroxy-2-methyl-3,4-dihydro-2H,5H -pyrano[3,2 c][1]benzopyran 5 one. Acta Crystallogr A C45:1908–1911
Ruggiero G, Platt OL Jr, Valente EJ, Eggleston DS (1989) Structures of (±)-trans-4-(2,3-dimethoxyphenyl) and (+) trans 4-(4-methylphenyl)-2-hydroxy-2-methyl-3,4-dihydro-2H,5H -pyrano[3,2 c][1]benzopyran-5-one. Acta Crystallogr A C45:1740–1743
Valente EJ, Eggleston DS (1991) Dihydropyran Ring Conformations I: Structures of 2-methoxy and hydroxy-2,4-dimethyl-3,4-dihydro-2H,5H -pyrano[3,2 c][1]benzopyran 5 ones. Acta Crystallogr A B47:498–501
Giannini DA, Chan KK, Roberts JD (1974) Carbon-13 nuclear magnetic resonance spectroscopy. Structure of the anticoagulant warfarin and related compounds in solution. Proc Natl Acad Sci USA 71:4221–4223
Valente EJ, Lingafelter EC, Porter WR, Trager WF (1977) The structure of warfarin in solution. J Med Chem 20:1489–1493
Obaseki AO, Porter WP (1987) Structure of analogs of warfarin in solution. Arch Pharm Chem Life Sci 5:110–121
Valente EJ, Trager WF (1978) The anomalous chiroptical properties of warfarin and phenprocoumon. J Med Chem 21:141–143
Rosengren AM, Karlsson BCG (2011) Spectroscopic evidence for the presence of the cyclic hemiketal form of warfarin in aqueous solution: Consequences for bioavailability. Biochem Biophys Res Commun 407:318–320
Obaseki AO, Porter WR, Trager WF (1982) 4-Hydroxycoumarin/2-hydroxychromone tautomerism: infrared spectra of 2–13C and 3-D labeled 4-hydroxycoumarin and its anion. J Heterocycl Chem 19:385–390
Townsend MG, Odam EM (1976) The identification of two products formed during the methylation of warfarin with diazomethane. Chem Ind 6:274–275
Valente EJ (1977) A structural study of coumarin anticoagulants and other derivatives of 4-hydroxycoumarin. University of Washington, Seattle, WA. Dissertation
Guasch L, Peach ML, Nicklaus MC (2015) Tautomerism of warfarin: combined chemoinformatics, quantum chemical, and NMR Investigation. J Org Chem 80:9900–9909
Martin YC (2009) Let’s not forget the tautomers. J Comput Aided Mol Des 23:693–704
Kim H, Yes C, Preston P, Chin J (2006) Substrate-directed stereoselectivity in vicinal diamine-catalyzed synthesis of warfarin. Org Lett 8:5239–5242
West BD, Preis S, Schroeder CH, Link KP (1961) Studies on the 4-hydroxycoumarins. XVII. The resolution and absolute configuration of warfarin. J Am Chem Soc 83:2676–2679
He M, Korzekwa KR, Jones JP, Rettie AE, Trager WF (1999) Structural forms of phenprocoumon and warfarin that are metabolized at the active site of CYP2C91. Arch Biochem Biophys 372:16–28
Bush E, Trager WF (1983) High yield synthesis of warfarin and its hydroxylated metabolites: new compounds. J Pharm Sci 72:830–831
CrysalisPro (2023) Software for collection and analysis for single-crystal X-ray data. Rigaku Corporation, version 2020
Clark RS, Reid JS (1995) The analytical calculation of absorption in multifaceted crystals. Acta Crystallogr A51:887–897
Sheldrick GM (2008) A short history of SHELX. Acta Crystallogr A64:112–122; program release of 2018
Johnson CK, Guerdon JF, Richard P, Whitlow S, Hall SR (1972) ORTEP. The X-RAY system of crystallographic programs. TR-192. 283p
Bruker Corporation, Billerica MA (2023) Topspin 4.3.0; Analytical tool “Teddy”
Dennington R, Keith TA, Millam JM (2016) GaussView, Version 6.1, Semichem Inc., Shawnee Mission, KS
Dnielyan E, Zunigs M, Hoang K, Magers DH, Osborne DA, Valente EJ (2023) Cyclic-open tautomerism in a warfarin analog. J Chem Crystallogr. https://doi.org/10.1007/s10870-023-00984-2
Hohenberg P, Kohn W (1964) Inhomogeneous electron gas. Phys Rev B 136:864–871
Kohn W, Sham LJ (1965) Self-consistent equations including exchange and correlation effects. Phys Rev A 140:1133–1138
Zhao Y, Truhlar DG (2006) A new local density functional for main-group thermochemistry, transition metal bonding, thermochemical kinetics, and noncovalent interactions. J Phys Chem 110:5121–5129
Dunning TH Jr (1989) Gaussian basis sets for use in correlated molecular calculations. I. The atoms boron through neon and hydrogen. J Chem Phys 90:1007–1023
Mennucci B, Cances C, Tomasi J (1997) Evaluation of solvent effects in isotropic and anisotropic dielectrics and in ionic solutions with a unified integral equation method: theoretical bases, computational implementation, and numerical applications. J Phys Chem B 101(49):10506–10517
Tomasi J, Mennucci B, Cammi R (2005) Quantum mechanical continuum solvation models. Chem Rev 105(8):2999–3094
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Petersson GA, Nakatsuji H, Li X, Caricato M, Marenich AV, Bloino J, Janesko BG, Gomperts R, Mennucci B, Hratchian HP, Ortiz JV, Azmaylov AF, JSonnenberg JL, Williams-Young D, Ding J, Lipparini F, Egidi F, Goings J, Peng B, Petrone A, Henderson T, Ranasinghe D, Zakrzewski VG, Gao J, Rega N, Zheng G, Liang W, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T , Honda Y, Kitao O, Nakai H, Vreven T, Throssell K, Montgomery JA Jr, Peralta JE, Ogliaro F, Bearpark MJ, Heyd JJ, Brothers EN, Kudin KN, Staroverov VN, Keith TA, Kobayashi R, Normand J, Raghavachari K, Kendell AP, Burant JC, Iyengar SS, Tomasi J, Cossi M, Millam JM, Klene M, Adamo C, Cammi R, Ochterski JW, Martin RL, Morokuma K, Farkas O, Foresman JB, Fox DJ (2016). Gaussian 16, Revision C.01, Gaussian, Inc., Wallingford CT
Shahzadi S, Ali S, Asif I, Ashraf R, Jin G-X (2006) The mechanism and crystal structure of 2-methoxy-2-methyl-4-phenyl-3,4,4a, 0b-tetrahydro-2H,5H-pyrano[3,2-c]chromen-5-one. Acetal of warfarin acid. Turk J Chem 30(6):703–709
Sakpal SS et al (2021) The curious case of aqueous warfarin: structural isomers or distinct excited states? J Phys Chem B 125:287–2878
Nowak P, Olechowska P, Mitoraj M, Wozniakiewicz M, Kocielniak P (2015) Determination of acid dissociation constants of warfarin and hydroxywarfarins by capillary electrophoresis. J Pharm Biomed Anal 112:89–97
Castleberry B, Valente EJ (1990) Open-cyclic warfarin isomerism: 5-hydroxywarfarin. J Chem Crystallogr 20:583–593
Casalone G, Pilati T, Binello A (1998) Five 3,4-dihydro-2H,5H-pyrano[3,2-c][1]-benzopyran-5-one derivatives. Acta Crystallogr A C54:1042–1047
Wang Y, Zhang W, Xi X, Zhang G (2010) (4R)-Ethyl 4-(4-chlorophenyl)-2-hydroxy-5-oxo-2,3,4,5-tetrahydropyrano[3,2-c]-chromene-2-carboxylate. Corrigendum. Acta Crystallog E 66:o217
Valente EJ, Porter WR, Trager WF (1978) The conformation of selected 3-substituted 4-hydroxycoumarins in solution by NMR: warfarin and phenprocoumon. J Med Chem 21:231–234
Duong T-V, Carroll TS, Bejan DS, Valente EJ (2020) β-Chain hydrogen-bonding in 4-hydroxycoumarins. J Chem Crystallogr 50:387–399
Henschel H, Karlsson BCG, Rosengren AM, Nicholls IA (2010) The mechanistic basis for warfarin’s structural diversity and implications for its bioavailability. J Mol Struct 958:7–9
Mercury (2016) Cambridge Crystallographic Data Center, version 3.10
Talsi VP, Evdokimov SN (2017) New cases of prototropic tautomerism in substituted pyridines. J Chem Eng Bioanal Chem 1:38–48
Friebolin H (1993) Basic one-and two-dimensional NMR spectroscopy, 2nd edn. VCH Publishers, New York
Chibber SS, Sharma RP (1978) Tautomerism of 2-hydroxychromones and 4-hydroxycoumarins. Curr Sci 47:730–731
Beak P, Lee J-K (1974) Methylation of prototropic ambident nucleophiles. the proton as a formal directing group. J Org Chem 40:147–148
Manolov I, Maichle-Moesssmer C (2013) Synthesis and crystal structure of 4-hydroxy-3-[(3E)-3-(hydroxyimino)-1-(4-nitrophenyl)butyl]-2H-chromen-2-one. Bul Chem Commun 15:109–113
Manolov I, Maichle-Moesssmer C (2007) Crystal structure of 4-hydroxy-3-[1-phenyl-2-(4-methoxybenzoyl)ethyl]-2H-1-benzopyran-2-one. Anal Sci 23:x79–x80
Sheth AR, Brennessel WW, Young VC Jr, Muller FX, Grant DJW (2004) Solid-State properties of warfarin sodium 2-propanol solvate. J Pharm Sci 93:2669–2690
Author information
Authors and Affiliations
Contributions
National Science Foundation (MRI #0604188) for crystallographic equipment (EV), and University of Portland Butine and SURE funding for computational hardware and software (DO), and a research stipend (KH). DO, ED, and KH performed the computations, DO analyzed the computations and determined torsional, angle, and distance metrics. EJV performed the syntheses, spectroscopic and crystallographic experiments. We thank Dr. David H. Magers, Mississippi College, for discussions on computational strategies.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicting financial or other interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Osborne, D.A., Danielyan, E., Hoang, K. et al. Warfarin Tautomers in Solution: A Structural, Computational and Thermodynamic Study. J Chem Crystallogr 54, 64–83 (2024). https://doi.org/10.1007/s10870-023-00999-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10870-023-00999-9