Abstract
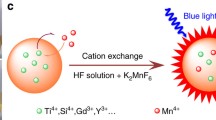
Using a green and convenient method to prepare fluoride red phosphor is an important challenge for industrialization applications. In this work, K2SiF6:Mn4+ red phosphor was successfully prepared through a brief one-step co-precipitation method without HF. A simplified and non-toxic ion exchange strategy for the formation of narrow-band red-emitting K2SiF6:Mn4+ phosphors was developed using different solvent (HAc, H3PO4, HNO3, HCl, H2O). The photoluminescence intensity of the prepared K2SiF6:Mn4+ red phosphor was the highest when the pH of the reaction solution was 1.0. Furthermore, using K2SiF6:Mn4+ as red component, the warm white light LED (light-emitting diode) with low correlated color temperature (CCT = 4095 K), high color rendering index (CRI, Ra = 87) was achieved, which proved that the phosphor synthesized by this method had great application potential. The preparation method provided in this article is safer and more environmentally friendly, and has important commercial and social value.









Similar content being viewed by others
References
Q. Zhang, X. Wang, X. Ding, Y. Wang, Inorg. Chem. 56, 6990 (2017)
L. Huang, Y. Liu, J. Yu, Y. Zhu, F. Pan, T. Xuan, M.G. Brik, C. Wang, J. Wang, A.C.S. Appl, Mater. Interfaces 10, 18082 (2018)
J. Hou, W. Jiang, Y. Fang, F. Huang, J. Mater. Chem. C 1, 5892 (2013)
Z. Xia, A. Meijerink, Chem. Soc. Rev. 46, 275 (2017)
C.C. Lin, R.S. Liu, J. Phys. Chem. Lett. 2, 1268 (2011)
H. Ming, S. Liu, L. Liu, J. Peng, J. Fu, F. Du, X. Ye, A.C.S. Appl, Mater. Interfaces 10, 19783 (2018)
S. Ye, F. Xiao, Y.X. Pan, Y.Y. Ma, Q.Y. Zhang, Mater. Sci. Eng. R 71, 1 (2010)
R.J. Xie, N. Hirosaki, T. Takeda, Appl. Phys. Express 2, e022401 (2009)
T. Suehiro, R.J. Xie, N. Hirosaki, Ind. Eng. Chem. Res. 52, 7453 (2013)
T. Suehiro, R.J. Xie, N. Hirosaki, Ind. Eng. Chem. Res. 53, 2713 (2014)
H. Xu, F. Hong, G. Liu, X. Dong, W. Yu, J. Wang, Opt. Mater. 99, e109500 (2020)
Y. Jin, Y. Hu, L. Chen, X. Wang, Z. Mu, G. Ju, T. Wang, Mater. Lett. 126, 75–77 (2014)
A. Katelnikovas, J. Plewa, S. Sakirzanovas, D. Dutczak, D. Enseling, F. Baur, H. Winkler, A. Kareiva, T. Jüstel, J. Mater. Chem. 22, 22126 (2012)
Q. Zhou, Y. Zhou, Y. Liu, L. Luo, Z. Wang, J. Peng, J. Yan, M. Wu, J. Mater. Chem. C 3, 3055 (2015)
C. Liao, R. Cao, Z. Ma, Y. Li, G. Dong, K.N. Sharafudeen, J. Qiu, J. Am. Ceram. Soc. 96, 3552 (2013)
Y. Jin, M.H. Fang, M. Grinberg, S. Mahlik, T. Lesniewski, M.G. Brik, G.Y. Luo, J.G. Lin, R.S. Liu, A.C.S. Appl, Mater. Interfaces. 8, 11194 (2016)
M. Zhu, Y. Pan, Y. Huang, H. Lian, J. Lin, J. Mater. Chem. C 6, 491 (2018)
E. Song, J. Wang, J. Shi, T. Deng, S. Ye, M. Peng, J. Wang, L. Wondraczek, Q. Zhang, A.C.S. Appl, Mater. Interfaces 9, 8805 (2017)
S. Adachi, T. Takahashi, J. Appl. Phys. 106, e013516 (2009)
L. Xi, Y. Pan, M. Zhu, H. Lian, J. Lin, Dalton Trans. 46, 13835 (2017)
W.L. Wu, M.H. Fang, W. Zhou, T. Lesniewski, S. Mahlik, M. Grinberg, M.G. Brik, H.S. Sheu, B.M. Cheng, J. Wang, R.S. Liu, Chem. Mater. 29, 935 (2017)
T. Takahashi, S. Adachi, Electrochem. Solid-State Lett. 12, J69 (2009)
J. Liao, L. Nie, L. Zhong, Q. Gu, Q. Wang, Luminescence 31, 802 (2016)
I. Jang, J. Kim, H. Kim, W.H. Kim, S.W. Jeon, J.P. Kim, Colloids Surf. A Physicochem. Eng. Asp. 520, 850 (2017)
L. Huang, Y. Zhu, X. Zhang, R. Zou, F. Pan, J. Wang, M. Wu, Chem. Mater. 28, 1495 (2016)
Z. Hou, X. Tang, X. Luo, T. Zhou, L. Zhang, R.J. Xie, J. Mater. Chem. C 6, 2741 (2018)
H. Bode, H. Jenssen, F. Bandte, Angew. Chem. 65, 304 (1953)
M.H. Du, J. Mater. Chem. C 2, 2475 (2014)
L.Y. Wang, E.H. Song, Y.Y. Zhou, T.T. Deng, S. Ye, Q.Y. Zhang, J. Mater. Chem. C 6, 8670 (2018)
Y. Zhu, L. Huang, R. Zou, J. Zhang, J. Yu, M. Wu, J. Wang, Q. Su, J. Chem. Mater. C 4, 5690 (2016)
X. Pan, D. Hou, M. Zhou, H. Lai, H. Ming, X. Ye, ECS J. Solid State Sci. Technol. 7, R3006 (2018)
X. Jiang, Y. Pan, S. Huang, X. Chen, J. Wang, G. Liu, J. Mater. Chem. C 2, 2301 (2014)
J. Zhou, Z. Xia, M. Bettinelli, Q. Liu, RSC Adv. 6, 2046 (2016)
S.S. Wang, W.T. Chen, Y. Li, J. Wang, H.S. Sheu, R.S. Liu, J. Am. Chem. Soc. 135, 12504 (2013)
G. Jiang, B. Yang, G. Zhao, Y. Liu, J. Zou, H. Sun, H. Ou, Y. Fang, J. Hou, Opt. Mater. 83, 93 (2018)
Acknowledgements
Jingshan Hou and Jingzhu Chen have equally contributed to this work. This work is financially supported by the National Natural Science Foundation of China (NSFC) (grant numbers: 51902203 and 51772184), Shanghai Rising-Star Program (21QC1401100), the Program of Shanghai Academic/Technology Research Leader (19XD1434700), the Shanghai Natural Science Foundation (grant number: 20ZR1455400) and the Ministry of Science and Technology of Taiwan (Contract Nos. MOST 109-2113-M-027-004, MOST 109-2622-M-027-001-CC2, MOST 110-2113-M-027-012, and MOST 110-2622-M-027-001).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hou, J., Chen, J., Hu, M. et al. Green route synthesis of K2SiF6:Mn4+ red phosphor through a brief one-step co-precipitation method for warm white light LEDs. J Mater Sci: Mater Electron 33, 2204–2212 (2022). https://doi.org/10.1007/s10854-021-07428-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10854-021-07428-7