Abstract
Surface defect engineering has shown to be an effective way to prepare cheap and efficient photoanode. In this study, oxygen vacancies are introduced to the hematite photoanode by a stepped atmosphere calcination with varied duration. The property of oxygen vacancies is verified by XRD, TEM, XPS and EPR analysis. With this facile treatment, the solar water splitting performance has been investigated. The enhanced photocurrent is about 1.96 mA cm−2 at 1.23 VRHE for the photoanode after 1.5 h calcination, which can keep constant even after 10 h illumination. Further study reveals that the maximum IPCE value of the photoanode reaches 56.5%. The creation of oxygen vacancies can lower the corresponding band gap for about 0.1 eV, which enhances the light absorption performance at the wavelength range from 750 to 620 nm. This photoanode displays the shortest charge transfer duration about 0.919 ms and the highest DC photocurrent about 597.13 × 10–8 mA cm−2 based on IMPS measurement. The electronic property and interfacial resistance of the photoanode are also characterized by Mott–Schottky and EIS measurement. The corresponding detailed mechanism is discussed in this paper.
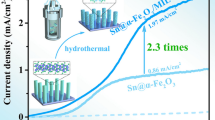
Graphical abstract

Schematic diagram and summary of the mechanism of oxygen vacancies introduced to the surface of α-Fe2O3 photoanode during solar water splitting.









Similar content being viewed by others
References
Walter MG, Warren EL, McKone JR et al (2010) Solar water splitting cells. Chem Rev 110:6446–6473
Yang J, Wang D, Han H, Li C (2013) Roles of cocatalysts in photocatalysis and photoelectrocatalysis. Acc Chem Res 46:1900–1909
Suntivich J, May KJ, Gasteiger HA, Goodenough JB, Shao-Horn Y (2011) A Perovskite oxide optimized for oxygen evolution catalysis from molecular orbital principles. Science 334:1383–1385
Nguyen M-T, Piccinin S, Seriani N, Gebauer R (2015) Photo-oxidation of water on defective Hematite(0001). ACS Catal 5:715–721
Sivula K, Le Formal F, Grätzel M (2011) Solar water splitting: progress using Hematite (α-Fe2O3) photoelectrodes. Chemsuschem 4:432–449
Chen Z, Jaramillo TF, Deutsch TG et al (2010) Accelerating materials development for photoelectrochemical hydrogen production: standards for methods, definitions, and reporting protocols. J Mater Res 25:3–16
Tamirat AG, Rick J, Dubale AA, Su WN, Hwang BJ (2016) Using hematite for photoelectrochemical water splitting: a review of current progress and challenges. Nanoscale Horizons 1:243–267
Sivula K, Zboril R, Le Formal F et al (2010) Photoelectrochemical water splitting with mesoporous hematite prepared by a solution-based colloidal approach. J Am Chem Soc 132:7436–7444
Shen S, Lindley SA, Chen X, Zhang JZ (2016) Hematite heterostructures for photoelectrochemical water splitting: rational materials design and charge carrier dynamics. Energy Environ Sci 9:2744–2775
Mishra M, Chun D-M (2015) α-Fe2O3 as a photocatalytic material: a review. Appl Catal A 498:126–141
Tilley SD, Cornuz M, Sivula K, Grätzel M (2010) Light-Induced water splitting with hematite: improved nanostructure and iridium oxide catalysis. Angew Chem Int Ed 49:6405–6408
Klahr B, Gimenez S, Fabregat-Santiago F, Hamann T, Bisquert J (2012) Water oxidation at hematite photoelectrodes: the role of surface states. J Am Chem Soc 134:4294–4302
Kment S, Riboni F, Pausova S et al (2017) Photoanodes based on TiO2 and alpha-Fe2O3 for solar water splitting - superior role of 1D nanoarchitectures and of combined heterostructures. Chem Soc Rev 46:3716–3769
Zhong DK, Cornuz M, Sivula K, Graetzel M, Gamelin DR (2011) Photo-assisted electrodeposition of cobalt-phosphate (Co-Pi) catalyst on hematite photoanodes for solar water oxidation. Energy Environ Sci 4:1759–1764
Yu X, Liu J, Yin W et al (2019) Ultrathin NiMn-layered double hydroxide nanosheets coupled with α-Fe2O3 nanorod arrays for photoelectrochemical water splitting. Appl Surf Sci 492:264–271
Zandi O, Klahr BM, Hamann TW (2013) Highly photoactive Ti-doped alpha-Fe2O3 thin film electrodes: resurrection of the dead layer. Energy Environ Sci 6:634–642
Luo ZB, Li CC, Zhang D, Wang T, Gong JL (2016) Highly-oriented Fe2O3/ZnFe2O4 nanocolumnar heterojunction with improved charge separation for photoelectrochemical water oxidation. Chem Commun 52:9013–9015
Youn DH, Park YB, Kim JY, Magesh G, Jang YJ, Lee JS (2015) One-pot synthesis of NiFe layered double hydroxide/reduced graphene oxide composite as an efficient electrocatalyst for electrochemical and photoelectrochemical water oxidation. J Power Sour 294:437–443
Tilley SD, Cornuz M, Sivula K, Gratzel M (2010) Light-induced water splitting with hematite: improved nanostructure and iridium oxide catalysis. Angew Chem-Int Edit 49:6405–6408
Mohapatra SK, John SE, Banerjee S, Misra M (2009) Water photooxidation by smooth and ultrathin α-Fe2O3 nanotube arrays. Chem Mater 21:3048–3055
Ling Y, Wang G, Wheeler DA, Zhang JZ, Li Y (2011) Sn-Doped hematite nanostructures for photoelectrochemical water splitting. Nano Lett 11:2119–2125
Zhang P, Wang T, Chang X, Zhang L, Gong J (2016) Synergistic cocatalytic effect of carbon nanodots and Co3O4 nanoclusters for the photoelectrochemical water oxidation on hematite. Angew Chem 55:5851–5855
Wang L, Jo M-J, Katagiri R et al (2018) Antioxidant effects of citrus pomace extracts processed by super-heated steam. LWT 90:331–338
Luo Z, Li C, Liu S, Wang T, Gong J (2017) Gradient doping of phosphorus in Fe2O3 nanoarray photoanodes for enhanced charge separation. Chem Sci 8:91–100
Hao L, Huang H, Zhang Y, Ma T (2021) Oxygen vacant semiconductor photocatalysts. Adv Func Mater 31:2100919
Zhuang G, Chen Y, Zhuang Z, Yu Y, Yu J (2020) Oxygen vacancies in metal oxides: recent progress towards advanced catalyst design. Sci China Mater 63:2089–2118
Xiao YM, Wu JH, Cheng CX et al (2012) Low temperature fabrication of high performance and transparent Pt counter electrodes for use in flexible dye-sensitized solar cells. Chin Sci Bull 57:2329–2334
Li M, Deng JJ, Pu AW et al (2014) Hydrogen-treated hematite nanostructures with low onset potential for highly efficient solar water oxidation. J Mater Chem A 2:6727–6733
Feng C, Fu S, Wang W, Zhang Y, Bi Y (2019) High-crystalline and high-aspect-ratio hematite nanotube photoanode for efficient solar water splitting. Appl Catal B 257:117900
Zhu CQ, Li CL, Zheng MJ, Delaunay JJ (2015) Plasma-induced oxygen vacancies in ultrathin hematite nanoflakes promoting photoelectrochemical water oxidation. ACS Appl Mater Interfaces 7:22355–22363
Wang H, Zhang W, Li X et al (2018) Highly enhanced visible light photocatalysis and in situ FT-IR studies on Bi metal@defective BiOCl hierarchical microspheres. Appl Catal B 225:218–227
Yu H, Li J, Zhang Y et al (2019) Three-in-one oxygen vacancies: whole visible-spectrum absorption, efficient charge separation, and surface site activation for Robust CO2 photoreduction. Angew Chem Int Ed Engl 58:3880–3884
Wang ZL, Mao X, Chen P et al (2019) Understanding the roles of oxygen vacancies in Hematite-based photoelectrochemical processes. Angew Chem-Int Edit 58:1030–1034
van de Krol R (2012) in van de Krol R, Grätzel M (eds) Photoelectrochemical hydrogen production. Springer US, Boston, MA
Qiu P, Yang H, Yang L, Wang Q, Ge L (2018) Solar water splitting with nanostructured hematite: the role of annealing-temperature. Electrochim Acta 266:431–440
Kim HS, Cook JB, Lin H et al (2017) Oxygen vacancies enhance pseudocapacitive charge storage properties of MoO3-x. Nat Mater 16:454–460
Liu Y, Wang W, Xu X, Marcel Veder J-P, Shao Z (2019) Recent advances in anion-doped metal oxides for catalytic applications. J Mater Chem A 7:7280–7300
Sarkar A, Khan GG (2019) The formation and detection techniques of oxygen vacancies in titanium oxide-based nanostructures. Nanoscale 11:3414–3444
Zhu Y, Liu X, Jin S et al (2019) Anionic defect engineering of transition metal oxides for oxygen reduction and evolution reactions. J Mater Chem A 7:5875–5897
Yamashita T, Hayes P (2008) Analysis of XPS spectra of Fe2+ and Fe3+ ions in oxide materials. Appl Surf Sci 254:2441–2449
Zhang Y, Yuan S-Y, Zou Y, Li T-T, Liu H, Wang J-J (2022) Enhanced charge separation and conductivity of hematite enabled by versatile NiSe2 nanoparticles for improved photoelectrochemical water oxidation. Appl Mater Today 28:101552
Peter LM, Ponomarev EA, Franco G, Shaw NJ (1999) Aspects of the photoelectrochemistry of nanocrystalline systems. Electrochim Acta 45:549–560
Ding C, Wang Z, Shi J et al (2016) Substrate-electrode interface engineering by an electron-transport layer in hematite photoanode. ACS Appl Mater Interfaces 8:7086–7091
Pu AW, Deng JJ, Hao YY, Sun XH, Zhong J (2014) Thickness effect of hematite nanostructures prepared by hydrothermal method for solar water splitting. Appl Surf Sci 320:213–217
Kennedy JH, Flatband KWF Jr (1978) Potentials and donor densities of polycrystalline α‐Fe2O3 determined from Mott‐Schottky plots. J Electrochem Soc 125:723–726
Sharma P, Jang JW, Lee JS (2019) Key strategies to advance the photoelectrochemical water splitting performance of alpha-Fe2O3 photoanode. ChemCatChem 11:157–179
Ma YM, Pendlebury SR, Reynal A, Le Formal F, Durrant JR (2014) Dynamics of photogenerated holes in undoped BiVO4 photoanodes for solar water oxidation. Chem Sci 5:2964–2973
Liao CH, Lee PM, Lin CH et al (2019) Experiment and theoretical calculation of the surface space charge region effect on photocurrent generation of SnO2 bilayer photodiode devices. Adv Electr Mater 5:6
Lan H, Xia Y, Feng K, Wei A, Kang Z, Zhong J (2019) Co-doped carbon layer to lower the onset potential of hematite for solar water oxidation. Appl Catal B 258:117962
Khataee A, Azevedo J, Dias P et al (2019) Integrated design of hematite and dye-sensitized solar cell for unbiased solar charging of an organic-inorganic redox flow battery. Nano Energy 62:832–843
Franking R, Li LS, Lukowski MA et al (2013) Facile post-growth doping of nanostructured hematite photoanodes for enhanced photoelectrochemical water oxidation. Energy Environ Sci 6:500–512
Wachs IE, Roberts CA (2010) Monitoring surface metal oxide catalytic active sites with Raman spectroscopy. Chem Soc Rev 39:5002–5017
Cesar I, Sivula K, Kay A, Zboril R, Grätzel M (2009) Influence of feature size, film thickness, and silicon doping on the performance of nanostructured hematite photoanodes for solar water splitting. The J Phys Chem C 113:772–782
Hedenstedt K, Bäckström J, Ahlberg E (2017) In-situ raman spectroscopy of α- and γ-FeOOH during cathodic load. J Electrochem Soc 164:H621–H627
Fernández-Climent R, Giménez S, García-Tecedor M (2020) The role of oxygen vacancies in water splitting photoanodes. Sustain Energy Fuels 4:5916–5926
Hu J, Zhao X, Chen W, Chen Z (2018) Enhanced charge transport and increased active sites on alpha-Fe2O3 (110) nanorod surface containing oxygen vacancies for improved solar water oxidation performance. ACS Omega 3:14973–14980
Ding J, Dai Z, Qin F, Zhao H, Zhao S, Chen R (2017) Z-scheme BiO1-xBr/Bi2O2CO3 photocatalyst with rich oxygen vacancy as electron mediator for highly efficient degradation of antibiotics. Appl Catal B 205:281–291
Geng Z, Kong X, Chen W et al (2018) Oxygen vacancies in ZnO nanosheets enhance CO electrochemical reduction to CO. Angew Chem Int Ed 57:6054–6059
Bao J, Zhang X, Fan B et al (2015) Ultrathin spinel-structured nanosheets rich in oxygen deficiencies for enhanced electrocatalytic water oxidation. Angew Chem Int Ed Engl 54:7399–7404
Acknowledgements
This work was financially supported by the National Natural Science Foundation of China [Grant No. 52071335] and the funding of China University of Petroleum (Beijing) [Grant No. 2462015YQ0602]. We are also grateful to the reviewers for the helpful comments. We also thank Prof. Steven Baldelli (University of Houston) for many valuable suggestions.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest to declare that are relevant to this article.
Additional information
Handling Editor: Dale Huber.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Xu, Y., Zhang, H., Gong, D. et al. Solar water splitting with nanostructured hematite: the role of oxygen vacancy. J Mater Sci 57, 19716–19729 (2022). https://doi.org/10.1007/s10853-022-07885-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10853-022-07885-3