Abstract
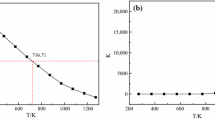
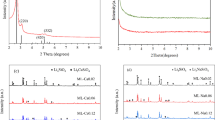
Solid absorption is regarded as an option to separate CO2 from high temperature gases. At present, suitable sorbents are limited to calcium or lithium containing oxides. Among the latter, lithium orthosilicate (Li4SiO4) might be preferred due to its high reactivity for CO2 absorption. Improved sorption properties are obtained by appropriate doping of Li4SiO4. Especially, doping of vacancies into the lattice of crystalline Li4SiO4 seems to be advantageous. This is concluded from sorption experiments of Al- and Fe-doped Li4SiO4 that are presented in this article. In addition, a comparison to eutectic doping by use of potassium is provided. For the investigation of practical applicability, cyclic absorption/desorption was performed on selected samples.
Similar content being viewed by others
References
B. BALASUBRAMANIAN, A. LOPEZ ORTIZ, S. KAYTAKOGLU and D. P. HARRISON, Chem. Eng. Sci. 54 (1999) 3543.
A. LOPEZ ORTIZ and D. P. HARRISON, Ind. Eng. Chem. Res. 40 (2001) 5102.
C. HAN and D. P. HARRISON, Chem. Eng. Sci. 49 (1994) 5875.
A. SILABAN, M. NARCIDA and D. P.HARRISON, Chem. Eng. Comm. 146 (1996) 149.
M. KATO, K. ESSAKI, S. YOSHIKAWA, K. NAKAGAWA and H. UEMOTO, in Proceedings of the 6th Int. Conf. on GHGT, Kyoto, October 2002, edited by J. Gale and Y. Kaya (Elsevier, 2003) Vol. II, p. 1579.
M. KATO, S. YOSHIKAWA and K. NAKAGAWA, J. Mater. Sci. Lett. 21 (2002) 485.
J. F. STEBBINS, Z. XU and D. VOLLATH, Solid State Ionics 78 (1995) L1.
K. JACKOWSKA and A. R. WEST, J. Mater. Sci. 18 (1983) 2380.
V. THANGADURAI and W. WEPPNER, Ionics 8 (2002) 281.
R. XIONG, J. IDA and Y. S. LIN, Chem. Eng. Sci. 58 (2003) 4377.
J.-I. IDA and Y. S. LIN, Environ. Sci. Technol. 37 (2003) 1999.
C. GAUER, Diploma Thesis, TU Bergakademie Freiberg, GER, 2004.
C. GAUER and W. HESCHEL, VGB PowerTech 85/4 (2005) 81.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gauer, C., Heschel, W. Doped lithium orthosilicate for absorption of carbon dioxide. J Mater Sci 41, 2405–2409 (2006). https://doi.org/10.1007/s10853-006-7070-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10853-006-7070-1