Abstract
Purpose
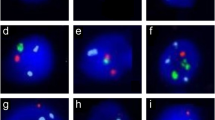
To evaluate the frequency of sperm nuclei disomy for chromosomes 8, X, and Y in patients with severe non-obstructive oligozoospermia and to assess possible correlations between sperm nuclei aneuploidy and semen parameters or a particular clinical phenotype.
Materials and methods
The sperm aneuploidy rate for chromosomes X, Y, and 8 were assessed in 16 infertile men with severe non-obstructive oligozoospermia and 7 healthy men with normal semen parameters. The frequency of sperm aneuploidy was compared between several patients groups according to their clinical and biological factors.
Results
The total rate of chromosomally abnormal spermatozoa was significantly higher in patients with severe oligozoospermia compared to control group (P < 0.001). A significant relationship was found between the age of patients, sperm concentration, and morphology and the mean rate of sex chromosomes disomy. In addition to the low sperm count (<5 × 106/ml), an elevated FSH level and an exposed to an elevated temperature are two major predictive factors leading to the production of higher numbers of chromosomally abnormal gametes.
Conclusion
Patients with severe oligozoospermia, who are potential candidates for assisted reproduction technology, presented a high level of sex numerical chromosome abnormalities, and consequently are at high risk of chromosome abnormalities in their offspring.



Similar content being viewed by others
References
Egozcue J, Templado C, Vidal F, Navarro J, Morer-Fargas F, Marina S. Meiotic studies in a series of 1100 infertile and sterile males. Hum Genet. 1983;65:185–8.
Egozcue J, Sarrate Z, Codina-Pascual M, Egozcue S, Oliver- Bonet M, Blanco J, et al. Meiotic abnormalities in infertile males. Cytogenet Genome Res. 2005;111:337–42.
Miharu N, Best RG, Young SR. Numerical chromosome abnormalities in spermatozoa of fertile and infertile men detected by fluorescence in situ hybridization. Hum Genet. 1994;93:502–6.
Guttenbach M, Martinez-Exposito MJ, Michelmann HW, Engel W, Schmid M. Incidence of diploid and disomic sperm nuclei in 45 infertile men. Hum Reprod. 1997;12:468–73.
Pang MG, Hoegerman SF, Cuticchia AJ, Moon SY, Doncel GF, Acosta AA, et al. Detection of aneuploidy for chromosomes 4, 6, 7, 8, 9, 10, 11, 12, 13, 17, 18, 21, X and Y by fluorescence in-situ hybridization in spermatozoa from nine patients with oligoasthenoteratozoospermia undergoing intracytoplasmatic sperm injection. Hum Reprod. 1999;14:1266–73.
Vegetti W, VanAssche E, Frias A, Verheyen G, Bianchi MM, Bonduelle M, et al. Correlation between semen parameters and sperm aneuploidy rates investigated by fluorescence in-situ hybridization in infertile men. Hum Reprod. 2000;15:351–65.
McInnes B, Rademaker A, Greene CA, Ko E, Barclay L, Martin RH. Abnormalities for chromosomes 13 and 21 detected in spermatozoa from infertile men. Hum Reprod. 1998;13:2787–90.
Bernadini L, Martini E, Geraedts JPM, Hopman AHN, Lanteri S, Conte N, et al. Comparison of gonosomal aneuploidy in spermatozoa of normal fertile men and those with severe male factor detected by in-situ hybridization. Mol Hum Reprod. 1997;3:431–8.
Nishikawa N, Murakami I, Ikuta K, Suzumori K. Sex chromosomal analysis of spermatozoa from infertile men using fluorescence in situ hybridization. J Assist Reprod Genet. 2000;17:97–102.
Martin RH, Rademaker AW, Greene C, Ko E, Hoang T, Barclay L, et al. A comparison of the frequency of sperm chromosome abnormalities in men with mild, moderate, and severe oligozoospermia. Biol Reprod. 2003;69:535–9.
Schmid TE, Kamischke A, Bollwein H, Nieschlag E, Brinkworth MH. Genetic damage in oligozoospermic patients detected by FISH, iRSM, SCSA and the Comet assay. Hum Reprod. 2003;18:1474–86.
Mehdi M, Smatti B, Saad A, Guerin JF, Benchaib M. Analysis by fluorescence in situ hybridization (FISH) of the relationship between gonosomic aneuploidy and the results of assisted reproduction in men with severe oligozoospermia. Andrologia. 2006;38:137–41.
World Health Organization. Laboratory manual for the examination of human semen and semen-cervical mucus interaction. New York: Cambridge University Press; 1992.
David G. Editorial: sperm banks in France. Arch Fr Pediatr. 1975;5:401–4.
Ohashi Y, Miharu N, Honda H, Samura O, Ohama K. High frequency of XY disomy in spermatozoa of severe oligozoospermic men. Hum Reprod. 2001;16:703–8.
Faure AK, Aknin-Seifer I, Frérot G, Pelletier R, De Robertis C, Cans C, et al. Predictive factors for an increased risk of sperm aneuploidies in oligo-astheno-teratozoospermic males. Int J Androl. 2007;30:153–62.
Kinakin B, Rademaker A, Martin R. Paternal age effect of YY aneuploidy in human sperm, as assessed by fluorescence in situ hybridization. Cytogenet Cell Genet. 1997;78:116–9.
Schmid TE, Brinkworth MH, Hill F, Sloter E, Kamischke A, Marchetti F, et al. Detection of structural and numerical chromosomal abnormalities by ACM-FISH analysis in sperm of oligozoospermic infertility patients. Hum Reprod. 2004;19:1395–400.
Shi Q. Martin RH:Aneuploidy in human spermatozoa: FISH analysis in men with constitutional chromosomal abnormalities, and in infertile men. Reproduction. 2001;121:655–66.
Morel F, Mercier S, Roux C, Elmrini T, Clavequin MC, Bresson JL. Interindividual variations in the disomy frequencies of human spermatozoa and their correlation with nuclear maturity as evaluated by aniline blue staining. Fertil Steril. 1998;69:1122–7.
Rives N, SaintClair A, Mazurier S, Sibert L, Simeon N, Joly G, et al. Relationship between clinical phenotype, semen parameters and aneuploidy frequency in sperm nuclei of 50 infertile males. Hum Genet. 1999;105:266–72.
Calogero AE, De Palma A, Grazioso C, Barone N, Romeo R, Rappazzo G, et al. Aneuploidy rate in spermatozoa of selected men with abnormal semen parameters. Hum Reprod. 2001;16:1172–9.
Damri LE, Vutyavanich T, Fishel S. Comparison of sex chromosome aneuploidy in spermatozoa of fertile men and those requiring ICSI treatment detected by fluorescence in situ hybridization. J Obstet Gynaecol Res. 2000;26:181–8.
Schultz H, Mennicke K, Schlieker H, AlHasani S, BalsPratsch M, Diedrich K, et al. Comparative study of disomy and diploidy rates in spermatozoa of fertile and infertile men: a donor-adapted protocol for multi-colour fluorescence in situ hybridization (FISH). Int J Androl. 2000;23:300–8.
Machev N, Gosset P, Viville S. Chromosome abnormalities in sperm from infertile men with normal somatic karyotypes: teratozoospermia. Cytogenet Genome Res. 2005;111:352–7.
Sun F, Ko E, Martin RH. Is there a relationship between sperm chromosome abnormalities and sperm morphology? Reprod Biol Endocrinol. 2006;4:1–5.
Vendrell JM, Garcia F, Veiga A, Calderon G, Egozcue S, Egozcue J, et al. Meiotic abnormalities and spermatogenic parameters in severe oligoasthenozoospermia. Hum Reprod. 1999;14:375–8.
Mieusset R, Bujan L. Testicular heating and its possible contributions to male infertility: a review. Int J Androl. 1995;18:169–84.
Robbins WA, Elashoff DA, Xun L, Jia J, Li N, Wu G, et al. Effect of lifestyle exposures on sperm aneuploidy. Cytogenet Genome Res. 2005;111:371–7.
Author information
Authors and Affiliations
Corresponding author
Additional information
Soumaya Mougou-Zerelli and Sonia Brahem equally contribute to the work.
Capsule
Patients with severe oligozoospermia presented a high level of sex numerical chromosome abnormalities, and consequently are at high risk of chromosome abnormalities in their offspring.
Rights and permissions
About this article
Cite this article
Mougou-Zerelli, S., Brahem, S., Kammoun, M. et al. Detection of aneuploidy rate for chromosomes X, Y and 8 by fluorescence in-situ hybridization in spermatozoa from patients with severe non-obstructive oligozoospermia. J Assist Reprod Genet 28, 971–977 (2011). https://doi.org/10.1007/s10815-011-9621-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-011-9621-x