Abstract
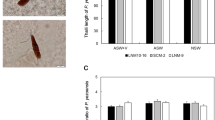
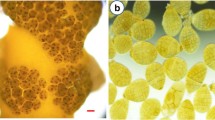
This work aimed to improve the techniques for cultivating the species Gelidium floridanum, testing different sources of calcium. Tetraspores and explants were grown in the laboratory for 20 days with seawater enriched with 50% von Stosch solution and oyster or mussel shell powder (252 mg L-1 and 336 mg L-1), or calcium chloride (147 mg L-1 and 295 mg L-1). Tetraspores or explants cultivated with no calcium salts or shell powder were used as control. Tetraspore germination rate and morphology, and germling morphology, average length, and growth rate were evaluated. Besides, the morphology, upright axes formation and growth rate of explants were also evaluated. There was no difference in tetraspore germination among treatments and control. Treatments with shell powder impaired tetraspore viability, indicated by a greenish color, cellular disorganization, and delay in germ tube development. Germlings cultivated with CaCl2 show better initial development but with no improvement compared to control. Concerning explants, those cultivated with mussel shell powder showed the highest growth rate (3.92 and 4.28% day-1, according to concentration) in relation to other treatments. In particular, those cultivated with 252 mg L-1 mussel shell powder had a significant formation of upright axes (10.15 ± 0.85 upright axes) compared to the control (5.18 ± 0.42 upright axes). Based on these results, we recommend using mussel shell powder at a concentration of 252 mg L-1 to optimize explant cultivation.






Similar content being viewed by others
Data Availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Aken ME, Griffin NJ, Robertson BL (1993) Cultivation of agarophyte Gelidium pristoides in Algoa Bay. South Africa Hydrobiologia 268:169–178
Altamirano M, Flores-Moya F, Kuhlenkamp R, Figueroa FL (2003) Stage-dependent sensitivity to ultraviolet radiation in zygotes of the brown alga Fucus serratus. Zygote 11:101–106
Armisén R (1991) Agar and agarose biotechnological applications. Hydrobiologia 221:157–166
Bouvie F (2018) Effects of extracellular calcium on the Gelidium floridanum germination. Dissertation. Universidade Federal de Santa Catarina, Brazil. (in Portuguese). P 64.
Bouzon ZL, Ouriques LO, Oliveira EC (2005) Ultrastructure of tetraspore germination in the agar-producing seaweed Gelidium floridanum (Gelidiales, Rhodophyta). Phycologia 44:409–415
Bouzon ZL, Ouriques LC, Oliveira EC (2006) Spore adhesion and cell wall formation in Gelidium floridanum (Rhodophyta, Gelidiales). J Appl Phycol 18:287–294
Cai G, Cresti M (2010) Microtubule motors and pollen tube growth still an open question. Protoplasma 247:131–143
Cárdenas L, Lovy-Wheeler A, Kunkel JG, Hepler PK (2008) Pollen tube growth oscillations and intracellular calcium levels are reversibly modulated by actin polymerization. Plant Physiol 146:1611–1621
Derksen J, Rutten T, Amstel TV, Win A, Doris F, Steer M (1995) Regulation of pollen tube growth. Acta Bot Neerl 44:93–119
EPAGRI (2019) Annual Summary of Agriculture in Santa Catarina 2018-2019 (In Portuguese). Available at: https://docweb.epagri.sc.gov.br/website_cepa/publicacoes/Sintese_2018_19.pdf (last accessed 20 January 2022)
Fei XG, Huang LJ (1991) Artificial sporeling and field cultivation of Gelidium in China. Hydrobiologia 221:119–124
Filipin EP, Pereira DT, Ouriques LC, Bouzon ZL, Simioni C (2019) Participation of actin filaments, myosin and phosphatidylinositol 3-kinase in the formation and polarisation of tetraspore germ tube of Gelidium floridanum (Rhodophyta, Florideophyceae). Plant Biol 21:352–360
Fredriksen S, Rueness J (1989) Culture studies of Gelidium latifolium (Grev.) Born. et Thur. (Rhodophyta) from Norway. Growth and nitrogen storage in response to varying photon flux density, temperature and nitrogen availability. Bot Mar 32:539–546
Green JJ, Cervantes DC, Peters NT, Logan KO, Kropf DL (2013) Dynamic microtubules and endomembrane cycling contribute to polarity establishment and early development of Ectocarpus mitospores. Protoplasma 250:1035–1043
Hable WE, Hart PE (2010) Signaling mechanisms in the establishment of plant and fucoid algal polarity. Mol Reprod Dev 77:751–758
Hamester MRR, Balzer PS, Becker D (2012) Characterization of calcium carbonate obtained from oyster and mussel shells and incorporation in polypropylene. Mater Res 15:204–208
Hansen JE (1980) Physiological considerations in the mariculture of red algae. J World Mariculture Soc 14:1–4
McLachlan J (1973) Growth media-marine. In: Stein JR (ed) Handbook of Phycological methods. Culture Methods and Growth Measurements. Cambridge University Press, Cambridge, pp 25–55
Mairh O, Rao P (1978) Culture studies on Gelidium pusillum (Stackh.) Le Jolis. Bot Mar 21:169–174
Melo RA, Harger BWW, Neushul M (1991) Gelidium cultivation in the sea. Hydrobiologia 221:91–106
Otaíza RD, Cáceres JH, Rodriguez CY, Sanhueza AG (2019) Seeding of fragments of the agarophyte Gelidium lingulatum (Rhodophyta, Gelidiales) for the repopulation of lower levels of wave exposed, intertidal rocky shores. J Appl Phycol 31:2133–2143
Porse H, Rudolph B (2017) The seaweed hydrocolloid industry: 2016 updates, requirements, and outlook. J Appl Phycol 29:2187–2200
Santelices B (1976) Note on mass cultivation of some species of Gelidiales (Rhodophyta). Rev Biol Mar Oceanogr 16:27–33 (In Spanish)
Santelices B (1991) Production ecology of Gelidium. Hydrobiologia 221:31–44
Santos R, Melo RA (2018) Global shortage of technical agars: back to basics (resource management). J Appl Phycol 30:2463–2473
Simioni C, Schmidt ÉC, Rover T, Santos R, Filipin EP, Pereira DT, Costa GB, Oliveira ER, Chow F, Ramlov F, Ouriques L, Maraschin M, Bouzon ZL (2015) Effects of cadmium metal on young gametophytes of Gelidium floridanum: metabolic and morphological changes. Protoplasma 252:1347–1359
Veeragurunathan V, Vadodariya N, Chaudhary JP, Gogda A, Saminathan KR, Meena R (2018) Experimental cultivation of Gelidium pusillum in open sea along the south east Indian coast. Indian J Geo Mar Sci 47:336–345
Yarish C, Pereira R (2008) Mass production of marine macroalgae. In: Jørgensen SE, Fath BD (eds) Encyclopedia of ecology. Elsevier, Oxford, pp 2236–2247
Yokoya NS, Handro W (1997) Thallus regeneration and growth induced by plant growth regulators and light intensity in Grateloupia dichotoma (Rhodophyta). In: Kitamura T (ed) Proceedings of the I.T.I.T. International Symposium on New Technologies from Marine-Sphere, Kagawa, Japan, pp 83–86
Zhou W, Sui Z, Wang J, Hu Y, Kang KH, Hong HR, Niaz Z, Wei H, Du Q, Peng C, Mi P, Que Z (2016) Effects of sodium bicarbonate concentration on growth, photosynthesis, and carbonic anhydrase activity of macroalgae Gracilariopsis lemaneiformis, Gracilaria vermiculophylla, and Gracilaria chouae (Gracilariales, Rhodophyta). Photosynth Res 128:259–270
Acknowledgments
CPB thanks Fundação de Amparo à Pesquisa e Inovação do Estado de Santa Catarina (FAPESC) for the master's scholarship. LH thanks Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) for the Productivity Fellowship (Process number 311680/2020-8). This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior – Brasil (CAPES) – Finance Code 001. We also thanks Fernando Zwierzikowski da Silva for the technical support and Prof. Zenilda L. Bouzon for the Plant Cell Biology Laboratory infrastructure.
Funding
This work was funding by Coordenação de Aperfeiçoamento de Pessoal de Nível Superior – Brasil (CAPES) – Finance Code 001, Fundação de Amparo à Pesquisa e Inovação do Estado de Santa Catarina (FAPESC) for the master's scholarship of Camila Pereira Bruzinga and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) for the Productivity Fellowship (Process number 311680/2020-8) of Leila Hayashi.
Author information
Authors and Affiliations
Contributions
All authors whose names appear on the submission
1) made substantial contributions to the conception or design of the work; or the acquisition, analysis, or interpretation of data; or the creation of new software used in the work;
2) drafted the work or revised it critically for important intellectual content;
3) approved the version to be published; and
4) agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Bruzinga, C.P., Simioni, C. & Hayashi, L. Shell powder as source of calcium in the development of Gelidium floridanum (Rhodophyta, Gelidiales) tetraspores and explants. J Appl Phycol 34, 2579–2588 (2022). https://doi.org/10.1007/s10811-022-02796-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10811-022-02796-x