Abstract
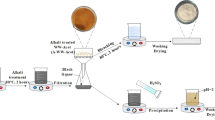
Carrageenans are economically important biopolymers used in the pharmaceutical, chemical and food industries. The present study was performed to optimise the extraction of carrageenan from Hypnea musciformis collected from Saint Martin Island, Bay of Bengal, Bangladesh, and characterisation of its chemical, rheological properties and antioxidant activities. Aqueous- and alkali-treated carrageenan from H. musciformis was extracted using conventional method and ultrasonic-assisted extraction (UAE) because this technique allows rapid extraction, which is important to avoid degradation of labile compounds. The resulting carrageenan was investigated in terms of yield, sulphate, 3,6-anhydrogalactose (AG), galactose (Gal) contents and structural properties using Fourier transform infrared (FT-IR) and 1H NMR spectroscopy. The carrageenan yield was higher using the novel UAE method and was comparable to that using the conventional technique. In vitro results confirmed a slight variation in sulphate, AG and Gal contents depending on the extraction method used. FT-IR and 1H NMR spectrum showed that carrageenan extracted using either the conventional method or UAE possesses typical analytical characteristics of κ-carrageenan. The rheological properties of carrageenan were evaluated using differential scanning calorimetery (DSC) and viscosity measurements and found to be similar to those of carrageenan extracted from same species of red alga. The antioxidant activity of carrageenan was measured using the 2,2-diphenyl-1-picrylhydrazyl (DPPH), OH radical scavenging, reducing power and phosphomolybdenum assays. The results indicated that carrageenan possesses antioxidant activity, the degree of which depends on its structural composition and the extraction method used.





Similar content being viewed by others
References
Al-Alawi AA, Al-Marhubi IM, Al-Belushi MSM, Soussi B (2011) Characterization of carrageenan extracted from Hypnea bryoides in Oman. Mar Biotechnol 13:893–899
Alves MGCF, Dore CMPG, Castro AJG, Nascimento MS, Cruz AKM, Soriano ME, Benevides MB, Leite EL (2012) Antioxidant, cytotoxic and hemolytic effects of sulfated galactans from edible red alga Hypnea musciformis. J Appl Phycol 24:1217–1227
Arman M, Qader SA (2012) Structural analysis of kappa-carrageenan isolated from Hypnea musciformis (red algae) and evaluation as an elicitor of plant defense mechanism. Carbohyd Res 88:1264–1271
Aziza M, Givernaud T, Chikhaoui-khay M, Bennasser L (2008) Seasonal variation of the growth, chemical composition and carrageenan extracted from Hypnea musciformis (Wulfen) Lamouroux harvested along the Atlantic coast of Morocco. Scient Res Essay 10:509–514
Beara IN, Lesjak MM, Jovin ED, Balong KJ, Anacov GT, Orcic DZ, Mimica-Dukic NM (2009) Plantin (Plantago L.) species as novel sources of flavonoid antioxidants. J Agric Food Chem 57:9268–9273
Bi F, Arman M, Mahmood-ul-Hussan IS (2006) Isolation and characterization of kappa carrageenan from Hypnea musciformis (red algae) of Karachi coast. J Saudi Chem Soc 10:501–50
Blois MS (1958) Antioxidant determinations by the use of a stable free radical. Nature 181:1199–1200
Bradford MM (1976) A rapid and sensitive for the quantitation of microgram quantitites of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Cacere PJ, Carlucci MJ, Damonte EB, Matsuhiro B, Zuniga EA (2000) Carrageenans from Chilean samples of Stenogramme interrupta (Phyllophoraceae): structural analysis and biological activity. Phytochem 53: 81-86
Campo VL, Kawano DF, Silva DB, Carvalho I (2009) Carrageenans: biological properties, chemical modifications and structural analysis—a review. Carbohyd Polym 77:167–180
Chopin T, Kerin BF, Mazerolle R (1999) Phycocolloid chemistry as a taxonomic indicator of phylogeny in the Gigartinales, Rhodophyceae: a review and current developments using Fourier transform infrared diffuse reflectance spectroscopy. Phycol Res 47:167–188
De Ruiter GA, Rudolph B (1997) Carrageenan biotechnology. Trends Food Sci Technol 8:389–395
Freile-Pelegrin Y, Robledo D, Azamar JA (2006) Carrageenan of Eucheuma isiforme (Solieriaceae, Rohodophyta) from Yucatán, Mexico. I. Effect of extraction conditions. Bot Mar 49:65–71
Friedlander M, Zelikovitch (1984) Growth rates, phycocolloid yield and quality of the red seaweeds, Gracilaria sp., Pterocladia capillacea, Hypnea musciformis, and Hypnea cornuta, in field studies in Israel. Aquaculture 40:57–66
Gonzalez ME, Alarcon B, Carrasco L (1987) Polysaccharides as antiviral agents: antiviral activity of carrageenan. Antimicrob Agents Chemother 31:1388–1393
Graham LE, Wilcox LW (2000) Algae. Prentice Hall, Upper Saddle River
Hilliou L, Larotonda F, Abreu P, Ramos A, Sereno A, Goncalves M (2006) Effect of extraction parameters on the chemical structure and gel properties of k/i-hybrid carrageenans obtained from Mastocarpus stellatus. Biomol Eng 23:201–208
Imeson A (2000) Carrageenan. Handbook of Hydrocolloids. P. W. GO Phillips. CRC Press, Boca Raton, pp 87–102
Jackson SG, McCandless EL (1978) Simple, rapid, turbidometric determination of inorganic sulfate and/or protein. Anal Biochem 90:802–808
Kim EY, Kim YR, Nam TJ, Kong IS (2012) Antioxidant and DNA protection activities of a glycoprotein isolated from a seaweed, Saccharina japonica. Intl J Food Sci Technol 47:1020–1027
Knutsen SH, Myslabodski DE, Larsen B, Usov AI (1994) A modified system of nomenclature for red algal galactans. Bot Mar 37:163–169
Knutsen S, Murano E, D’Amato M, Toffanin R, Rizzo R, Paoletti S (1995) Modified procedures for extraction and analysis of carrageenan applied to the red alga Hypnea musciformis. J Appl Phycol 7:565–576
Mabeau S (1989) Lafiliere algue francaise en 1988: atouts et point de blovage. Oceanis 15:673–692
Michel G, Nyval-Collen P, Barbeyron T, Czjzek M, Helbert W (2006) Bioconversion of red seaweed galactans: a focus on bacterial agarases and carrageenase. Appl Microbiol Biotechnol 71:23–33
Mtolera MSP, Buriyo AS (2004) Studies on Tanzanian Hypneaceae: seasonal variation in kappa-carrageenan content and properties in Hypnea musciformis (Gigartinales, Rhodophyta). Western Indian Ocean J Mar Sci 3:43–49
Navarro D, Stortz C (2003) Determination of the configuration of 3,6-anhydrogalactose and cyclizable α-galactose 6-sulfate units in red seaweed galactans. Carbohyd Res 338:2111–2118
Navarro D, Stortz C (2005) Microwave-assisted alkaline modification of red seaweed galactans. Carbohyd Polym 62:187–191
Oyaizu M (1986) Antioxidative activities of products of browning reaction prepared from glucosamine. Jpn J Nutri 44:307–315
Pereira L, Sousa A, Coelho H, Amado A, Ribeiro-Claro P (2003) Use of FTIR, FR-Ramman and 13C-NMR spectroscopy for identification of some seaweed phycocolloids. Biomol Eng 20:223–228
Prieto P, Pineda M, Aguilar M (1999) Spectrophotometric quantitation of antioxidant capacity through the formation of a phosphomolybdenum complex: specific application to the determination of vitamin E. Anal Biochem 269:337–341
Rafiquzzaman SM, Kim EY, Kim YR, Nam TJ, Kong IS (2013) Antioxidant activity of glycoprotein purified from Undaria pinnatifida measured by an in vitro digestion model. Int J Biol Macromol 62:265–272
Reis R, Caldeira A, Paula A, Barros-Barreto M (2006) Potential para maricultura da carragenófita Hypnea musciformis (Wulfen) J. V. Lamour. (Gigartinales, Rhodophyta) na Ilha da Marambaia, Baía de Sepetiba, RJ, Brasil. Acta Bot Bras 4:763–769
Reis R. Yoneshigue-Valentin Y, Dos Santos C (2008) Spatial and temporal variation of Hypnea musciformis carrageenan (Rhodophyta-Gigartinales) from natural beds in Rio de Janeiro state. Brazil J Appl Phycol 20:1–8
Rinaudo M (2007) Seaweed polysaccharides. In: Kamerling JP (ed) Comprehensive Glycoscience Vol 2. Elsevier, Amsterdam, pp 691–735
Samaram S, Mirhosseini H, Tan CP, Ghazali HM, Bordbar S, Serjouie A (2015) Optimisation of ultrasound-assisted extraction of oil from papaya seed by response surface methodology: oil recovery, radical scavenging antioxidant activity, and oxidation stability. Food Chem 172:7–17
Sokolova EV, Barabanova AO, Bogdanovich RN, Khomenko VA, Solov’eva TF, Yermak IM (2011) In vitro antioxidant properties of red algal polysaccharides. Biomed Prevent Nutri 1:161–167
Stortz CA, Cerezo AS (2000) Novel findings in carragenans, agaroids and “hybrid” red seaweed galactans. Curr Topics Phytochem 4:121–134
Van de Velde F, De Ruiter GA (2002) Carrageenan. In: Steinbuchel A, DeBaets S, VanDamme EJ (eds) Polysaccharides II: Polysaccharides from eukaryotes Vol. 6. Wiley-VCH, Weinheim, pp 245–274
Vázquez-Delfin E, Robledo D, Freile-Pelegrin Y (2014) Microwave–assisted extraction of the carrageenan from Hypnea musciformis (Cystocloniaceae, Rohodophyta). J Appl Phycol 26:901–907
Wang Y, Cheng Z, Mao J, Fan M, Wu X (2009) Optimization of ultrasonic assisted extraction process of Poria cocos polysaccharides by response surface methodology. Carbohyd Polym 77:713–717
Wong K, Cheung P (2000) Nutritional evaluation of some subtropical red and green seaweeds: part I—proximate composition, amino acid profiles and some physico-chemical properties. Food Chem 71:475–482
Yang W, Fang Y, Liang J, Hu Q (2011) Optimization of ultrasonic extraction of Flammulina velutipes polysaccharides and evaluation of its acetylcholine esterase inhibitory activity. Food Res Intl 44:1269–1275
Yaphe W (1960) Colorimetric determination of 3,6-anhydrogalactose and galactose in marine algal polysaccharides. Anal Chem 10:1327–1329
Yuan H, Song J, Zhang W, Li X, Lia N, Gao X (2006) Antioxidant activity and cytoprotective effect of κ-carrageenan oligosaccharaides and their derivatives. Bioorg Med Chem Lett 16:1329–34
Acknowledgments
This work was supported by a Research Grant of Pukyong National University (2015 year).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rafiquzzaman, S.M., Ahmed, R., Lee, J.M. et al. Improved methods for isolation of carrageenan from Hypnea musciformis and its antioxidant activity. J Appl Phycol 28, 1265–1274 (2016). https://doi.org/10.1007/s10811-015-0605-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10811-015-0605-6