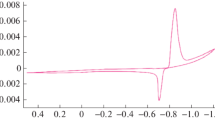
Abstract
The kinetics of zinc dissolution in concentrated potassium hydroxide solution were determined as a function of KOH concentration, amount of added ZnO, and temperature through linear sweep voltammetry (LSV) and electrochemical impedance spectroscopy (EIS). The measurements were evaluated with a simplified reaction scheme in which an intermediate ZnI species is formed on the electrode surface that subsequently reacts to a soluble ZnII species. Analysis of the LSV data with a two-step Butler–Volmer kinetics showed that the transfer coefficients and the surface coverage of the intermediate are approximately constant in the entire range of operation conditions, whereas the exchange current density for the first reaction step is about five times larger than for the second step. Analysis of the dynamic EIS measurements resulted in very similar current densities than obtained from the quasi-stationary LSV method. For the first time, activation energies describing the temperature dependence of the exchange current density were also determined.
Graphic abstract











Similar content being viewed by others
Abbreviations
- A :
-
Kinetic parameter (s Ω−1)
- B :
-
Kinetic parameter (s−1)
- j :
-
Current density (mA cm−2)
- R :
-
Ideal gas constant (J mol−1, K−1)
- R :
-
Resistance (Ω cm2)
- T :
-
Temperature (K (°C))
- F :
-
Faraday constant (C mol−1)
- n :
-
Electron transfer number
- Z :
-
Impedance (Ω cm2)
- η :
-
Overpotential (V)
- γ :
-
Surface coverage
- E a :
-
Activation energy (kJ mol−1)
- j 0 :
-
Exchange current density (mA cm−2)
- b :
-
Tafel slope
- α :
-
Transfer coefficient
- k :
-
Reaction
- overall:
-
Overall reaction
- t :
-
Transient
References
Mainar Aroa R, Boyano Iker, de Meatza Iratxe et al (2016) Alkaline aqueous electrolytes for secondary zinc–air batteries: an overview. Int J Energy Res 40:1032–1049. https://doi.org/10.1002/er.3499
Harting K, Kunz U, Turek T (2012) Zinc-air batteries: prospects and challenges for future improvement. Z Phys Chem 226:151. https://doi.org/10.1524/zpch.2012.0152
Gu P, Zheng M, Zhao Q et al (2017) Rechargeable zinc–air batteries: a promising way to green energy. J Mater Chem A 5(17):7651–7666. https://doi.org/10.1039/C7TA01693J
Bockelmann M, Reining L, Kunz U et al (2017) Electrochemical characterization and mathematical modeling of zinc passivation in alkaline solutions: a review. Electrochim Acta 237:276–298. https://doi.org/10.1016/j.electacta.2017.03.143
Wang K, Pei P, Ma Z et al (2014) Morphology control of zinc regeneration for zinc–air fuel cell and battery. J Power Sour 271:65–75. https://doi.org/10.1016/j.jpowsour.2014.07.182
Riede J-C, Turek T, Kunz U (2018) Critical zinc ion concentration on the electrode surface determines dendritic zinc growth during charging a zinc air battery. Electrochim Acta 269:217–224. https://doi.org/10.1016/j.electacta.2018.02.110
Neburchilov V, Wang H, Martin JJ et al (2010) A review on air cathodes for zinc–air fuel cells. J Power Sour 195(5):1271–1291. https://doi.org/10.1016/j.jpowsour.2009.08.100
Pei P, Wang K, Ma Z (2014) Technologies for extending zinc–air battery’s cyclelife: a review. Appl Energy 128:315–324. https://doi.org/10.1016/j.apenergy.2014.04.095
Han X, Li X, White J et al (2018) Metal-air batteries: from static to flow system. Adv Energy Mater 8(27):1801396. https://doi.org/10.1002/aenm.201801396
Bockelmann M, Kunz U, Turek T (2016) Electrically rechargeable zinc-oxygen flow battery with high power density. Electrochem Commun 69:24–27. https://doi.org/10.1016/j.elecom.2016.05.013
Bockris J, Nagy Z, Damjanovic A (1972) On the deposition and dissolution of zinc in alkaline solutions. J Electrochem Soc 119(3):285–295. https://doi.org/10.1149/1.2404188
Cachet C, Wiart R, Zoppas-Ferreira J (1993) Zinc deposition and dissolution in a flow-through porous electrode. Electrochim Acta 38(2–3):311–318. https://doi.org/10.1016/0013-4686(93)85145-O
Cachet C, Ströder U, Wiart R (1982) The kinetics of zinc electrode in alkaline zincate electrolytes. Electrochim Acta 27(7):903–908. https://doi.org/10.1016/0013-4686(82)80214-4
Dirkse TP (1981) The behavior of the zinc electrodes in alkaline solutions. J Electrochem Soc 128(7):1412–1415. https://doi.org/10.1149/1.2127654
Hampson NA, Shaw PE, Taylor R (1969) Anodic behaviour of zinc in potassium hydroxide solution: II.*Horizontal anodes in electrolytes containing Zn(II). Br Corros J 4(4):207–211. https://doi.org/10.1179/000705969798325398
Muralidharan VS, Rajagopalan KS (1978) Kinetics and mechanism of corrosion of zinc in sodium hydroxide solutions by steady-state and transient methods. J Electroanal Chem 94(1):21–36. https://doi.org/10.1016/S0022-0728(78)80395-7
Chang Y-C (1985) Anodic dissolution of zinc electrodes in alkaline electrolyte: mass transport effects. J Electrochem Soc 132(2):375. https://doi.org/10.1149/1.2113842
Popova TI, Simonova NA, Kabanov BN (1967) Anodic dissolution of passive zinc in zincate solutions of alkali. Soviet Electrochem 3:1273–1279
Dirkse TP (1955) Electrolytic oxidation of zinc in alkaline solutions. J Electrochem Soc 102(9):497–501. https://doi.org/10.1149/1.2430136
Armstrong RD, Bell MF (1974) The electrochemical behaviour of zinc in alkaline solution. Electrochemistry 4:1–17. https://doi.org/10.1039/9781847557247-00001
Hampson NA, Herdman GA, Taylor R (1970) Some kinetic and thermodynamic studies of the system Zn/Zn(II) ·OH−. J Electroanal Chem 25(1):9–18. https://doi.org/10.1016/S0022-0728(70)80034-1
Cachet C (1992) The behavior of zinc electrode in alkaline electrolytes. J Electrochem Soc 139(3):644. https://doi.org/10.1149/1.2069279
Vetter KJ (1967) Electrochemical kinetics: Theoretical and experimental aspects. Academic Press, New York
Deiss E, Holzer F, Haas O (2002) Modeling of an electrically rechargeable alkaline Zn–air battery. Electrochim Acta 47(25):3995–4010. https://doi.org/10.1016/S0013-4686(02)00316-X
Schröder D, Krewer U (2014) Model based quantification of air-composition impact on secondary zinc air batteries. Electrochim Acta 117:541–553. https://doi.org/10.1016/j.electacta.2013.11.116
Liu L, Zuo R, Sun Q et al (2013) Structure and electrical properties of Mn doped Bi(Mg1/2Ti1/2)O3-PbTiO3 ferroelectric thin films. Appl Surf Sci 268:327–331. https://doi.org/10.1016/j.apsusc.2012.12.087
Farr JPG, Hampson NA (1968) The exchange reaction Zn(II)/Zn(Hg) in alkali. J Electroanal Chem 18(4):407–411. https://doi.org/10.1016/S0022-0728(68)80008-7
Boden DP, Wylie RB, Spera VJ (1971) The electrode potential of zinc amalgam in alkaline zincate solutions. J Electrochem Soc 118(8):1298. https://doi.org/10.1149/1.2408309
Despić AR, Jovanović D, Rakić T (1976) Kinetics and mechanism of deposition of zinc from zincate in concentrated alkali hydroxide solutions. Electrochim Acta 21(1):63–77. https://doi.org/10.1016/0013-4686(76)85111-0
Chang Y-C (1984) A model for the anodic dissolution of zinc in alkaline electrolyte. J Electrochem Soc 131(7):1465. https://doi.org/10.1149/1.2115875
Orazem ME, Tribollet B (2017) Electrochemical impedance spectroscopy: the electrochemical society series. Wiley, Hoboken
Bockelmann M, Becker M, Reining L et al (2019) Passivation of zinc anodes in alkaline electrolyte: part II. Influence of operation parameters. J Electrochem Soc 166(6):A1132–A1139. https://doi.org/10.1149/2.0791906jes
Erdey-Grúz T, Volmer M (1930) Zur Theorie der Wasserstoff Überspannung. Z Phys Chem 150A:203–213. https://doi.org/10.1515/zpch-1930-15020
Danilov FI, Protsenko VS (2010) Actual activation energy of electrochemical reactions at stage charge transfer. Russ J Electrochem 46(2):188–195. https://doi.org/10.1134/S1023193510020102
Protsenko VS, Danilov FI (2011) Activation energy of electrochemical reaction measured at a constant value of electrode potential. J Electroanal Chem 651(2):105–110. https://doi.org/10.1016/j.jelechem.2010.12.014
Acknowledgements
This work was supported by the German Federal Ministry for Economic Affairs and Energy (Bundesministerium für Wirtschaft und Forschung (BMWi), Grant Number 03ESP217 E) during the ZnPLUS project.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Reining, L., Bockelmann, M., Kunz, U. et al. Kinetics of active zinc dissolution in concentrated KOH solutions. J Appl Electrochem 50, 149–158 (2020). https://doi.org/10.1007/s10800-019-01376-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10800-019-01376-1