Abstract
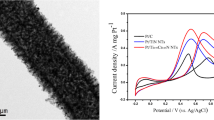
This research aimed to investigate the effect of the calcination temperature and the CeO2 to nanotubes (CNTs) weight ratio on the performance of Pt/CeO2−CNTs hybrid nanotube for methanol oxidation. The catalysts were prepared by microwave-assisted polyol process. Electrochemical results showed that Pt/CeO2−CNTs hybrid nanotube catalyst with the calcination temperature of 280 °C and the weight ratio of 4:4 is more active and stable than others. The properties of catalyst were characterized by X-ray diffraction, high-resolution transmission electron microscopy, and scanning transmission electron microscopy-energy dispersive spectroscopy elemental mapping. The increased catalytic efficiency of Pt in the Pt/CeO2−CNTs hybrid nanotube catalysts resulted from its smaller Pt nanoparticles and good contact with CeO2 and CNTs simultaneously.
Graphical abstract
A novel Pt/CeO2−CNTs hybrid nanotube catalyst with CNTs and CeO2-supported Pt nanoparticles has been successfully prepared by the liquid-phase chemical deposition method. The Pt nanoparticles are homogeneously distributed over CeO2−CNTs-−HN supports through adsorbing adjacent to CeO2, which are homogeneously distributed over CNTs. Further, the Pt nanoparticles are inserted into voids formed by CeO2 other than deposited on CeO2 directly, contacting with CeO2 and carbon simultaneously.










Similar content being viewed by others
References
Heinzel A, Barragan V (1999) A review of the state-of-the-art of the methanol crossover in direct methanol fuel cells. J Power Sources 84:70–74
Qiao Y, Li CM (2011) Nanostructured catalysts in fuel cells. J Mater Chem 21:4027
Shrestha S, Liu Y, Mustain WE (2011) Electrocatalytic activity and stability of Pt clusters on state-of-the-art supports: a review. Catalysis Reviews 53:256–336
Meher SK, Rao GR (2012) Polymer-assisted hydrothermal synthesis of highly reducible shuttle-shaped CeO2: microstructural effect on promoting Pt/C for methanol electrooxidation. ACS Catalysis 2:2795–2809
Yajima T, Uchida H, Watanabe M (2004) In-situ ATR-FTIR spectroscopic study of electro-oxidation of methanol and adsorbed CO at Pt-Ru alloy. J Phys Chem B 108:2654–2659
Carrettin S, Concepción P, Corma A, López Nieto JM, Puntes VF (2004) Nanocrystalline CeO2 increases the activity of Au for CO oxidation by two orders of magnitude. Angew Chem Int Ed 43:2538–2540
Zhang J-J, Wang Z-B, Li C, Zhao L, Liu J, Zhang L-M, Gu D-M (2015) Multiwall-carbon nanotube modified by N-doped carbon quantum dots as Pt catalyst support for methanol electrooxidation. J Power Sources 289:63–70
Wang X, Li X, Liu D, Song S, Zhang H (2012) Green synthesis of Pt/CeO2/graphene hybrid nanomaterials with remarkably enhanced electrocatalytic properties. Chem Commun 48:2885–2887
Lei M, Wang ZB, Li JS, Tang HL, Liu WJ, Wang YG (2014) CeO2 nanocubes-graphene oxide as durable and highly active catalyst support for proton exchange membrane fuel cell. Sci Rep 4:7415
Zhou Y, Gao Y, Liu Y, Liu J (2010) High efficiency Pt-CeO2/carbon nanotubes hybrid composite as an anode electrocatalyst for direct methanol fuel cells. J Power Sources 195:1605–1609
Lou X, Chen J, Wang M, Gu J, Wu P, Sun D, Tang Y (2015) Carbon nanotubes supported cerium dioxide and platinum nanohybrids: layer-by-layer synthesis and enhanced electrocatalytic activity for methanol oxidation. J Power Sources 287:203–210
Sun Z, Wang X, Liu Z, Zhang H, Yu P, Mao L (2010) Pt-Ru/CeO2/carbon nanotube nanocomposites: an efficient electrocatalyst for direct methanol fuel cells. Langmuir 26:12383–12389
Wang J, Yu S, Xi J, Chen L, Zhu W, Qiu X (2007) Preparation of Pt/CeO2−CNTs Through Spontaneous Adsorbing Pt Nanoparticles onto CNTs Aided by CeO2. Electrochem Solid-State Lett 10:B114–B118
Chu Y-Y, Wang Z-B, Gu D-M, Yin G-P (2010) Performance of Pt/C catalysts prepared by microwave-assisted polyol process for methanol electrooxidation. J Power Sources 195:1799–1804
Schmidt T, Gasteiger H, Stäb G, Urban P, Kolb D, Behm R (1998) Characterization of high-surface-area electrocatalysts using a rotating disk electrode configuration. J Electrochem Soc 145:2354–2358
Xing Y (2004) Synthesis and electrochemical characterization of uniformly-dispersed high loading Pt nanoparticles on sonochemically-treated carbon nanotubes. J Phys Chem B 108:19255–19259
Liu Z, Ling XY, Su X, Lee JY (2004) Carbon-supported Pt and PtRu nanoparticles as catalysts for a direct methanol fuel cell. J Phys Chem B 108:8234–8240
Coloma F, Sepulveda-Escribano A, Fierro J, Rodriguez-Reinoso F (1994) Preparation of platinum supported on pregraphitized carbon blacks. Langmuir 10:750–755
Wang J, Yu S, Xi J, Chen L, Zhu W, Qiu X (2007) Preparation of Pt/CeO[sub 2]–CNTs Through Spontaneous Adsorbing Pt Nanoparticles onto CNTs Aided by CeO[sub 2]. Electrochem Solid-State Lett 10:B114
García G, Koper MTM (2011) Carbon monoxide oxidation on Pt single crystal electrodes: understanding the catalysis for low temperature fuel cells. ChemPhysChem 12:2064–2072
Xu C, Zeng R, Shen PK, Wei Z (2005) Synergistic effect of CeO2 modified Pt/C catalysts on the alcohols oxidation. Electrochim Acta 51:1031–1035
Scibioh MA, Kim S-K, Cho EA, Lim T-H, Hong S-A, Ha HY (2008) Pt-CeO2/C anode catalyst for direct methanol fuel cells. Appl Catal B 84:773–782
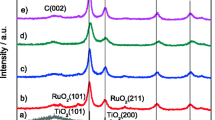
Yu J, Wang B (2010) Effect of calcination temperature on morphology and photoelectrochemical properties of anodized titanium dioxide nanotube arrays. Appl Catal B 94:295–302
Yu J, Yu H, Cheng B, Trapalis C (2006) Effects of calcination temperature on the microstructures and photocatalytic activity of titanate nanotubes. J Mol Catal A 249:135–142
Acknowledgments
This research is financially supported by the National Natural Science Foundation of China (Grant Nos. 21206124 and 21506159) and Natural Science Foundation of Tianjin (No. 15JCYBJC20500).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Yang, J., Chu, Y., Li, L. et al. Effects of calcination temperature and CeO2 contents on the performance of Pt/CeO2−CNTs hybrid nanotube catalysts for methanol oxidation. J Appl Electrochem 46, 369–377 (2016). https://doi.org/10.1007/s10800-016-0931-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10800-016-0931-7