Abstract
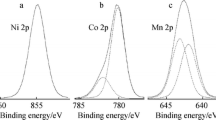
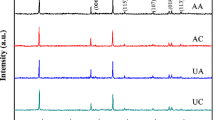
The LiNi1/3Co1/3Mn1/3O2 powders required for the present study, obtained by coprecipitation method has been surface coated with boron and aluminum. The morphology and crystal structure of powders have been characterized using scanning electron microscopy, X-ray diffraction and X-ray photoelectron spectroscopy techniques. The elemental distribution of the coated samples analyzed by transmission electron microscopy images and nano secondary ion mass spectrometry indicates a thin uniform layer of [B, Al]2O3 on the surface of spherical LiNi1/3Co1/3Mn1/3O2. The surface-modified LiNi1/3Co1/3Mn1/3O2 has been explored as a cathode material for lithium secondary ion battery applications. The electrochemical charge–discharge results reveal that the capacity retention rate of coated LiNi1/3Co1/3Mn1/3O2 after 40 cycles at 1 C rate maintains 93% of the initial discharge capacity while the rate of bare LiNi1/3Co1/3Mn1/3O2 maintains only 88%. It is noticed that the small amounts of boron and aluminum coatings on the surface of LiNi1/3Co1/3Mn1/3O2 can significantly improve the electrochemical properties of electrode materials because of the suppression of reaction between the cathode and the electrolytes.







Similar content being viewed by others
References
Scrosati B (1995) Nature 373:557
Tarascon JM, Armand M (2001) Nature 414:359
Guyomard D (2000) New trends in electrochemical technology: energy storage systems for electronics. Gordon & Breach Science Publishers, New York
Wakihara W, Yamamoto O (1998) Lithium ion batteries—fundamentals and performance. Kodansha-Wiley-VCH, Weinheim
Bruce PG, Scrosti B, Tarascon JM (2008) Angew Chem Int Ed 47:2930
Antolini E (2004) Solid State Ionics 170:159
Jeong ED, Kim HJ, Ahn CW, Ha MG, Hong TE, Kim HG, Jin JS, Bae JS, Hong KS, Kim YS, Kim HJ, Doh CH, Yang HS (2009) J Nanosic Nanotechnol 9:4467
Ahn CW, Ha MG, Hong KS, Lee DJ, Doh CH, Doh KY, Na JM, Song BH, Jeon HM, Cho YG, Yang HS, Jeong ED (2010) Defect Diffus Forum 297:906
Hong KS, Yu SM, Ha MG, Ahn CW, Hong TE, Jin JS, Kim HG, Jeong ED, Kim YS, Kim HJ, Doh CH, Yang HS, Jung H (2009) Bull Korean Chem Soc 30:1719
Cho PJ, Jeong ED, Shim YB (1998) Bull Korean Chem Soc 19:39
Jeong ED, Won MS, Shim YB (1998) J Power Sources 70:70
Stewart SG, Srinivasan V, Newman J (2008) J Electrochem Soc 155:A664
He SS, Ma ZF, Liao XZ, Jiang Y (2007) J Power Sources 163:1053
Martha SK, Markevich E, Burgel V, Salitra G, Zingigrad E, Markovsky B, Sclar H, Pramovich Z, Heik O, Aurbach D, Exnar I, Buqa H, Drezen T, Semrau G, Schmidt M, Kovacheva D, Saliyski N (2009) J Power Sources 189:288
Martha SK, Sclar H, Framowitz ZS, Kovacheva D, Saliyski N, Gofer Y, Sharon P, Golik E, Markovsky B, Aurbach D (2009) J Power Sources 189:248
Ohzuku T, Makimura Y (2001) Chem Lett 7:642
Yabuuchi N, Ohzuku T (2003) J Power Sources 119:171
Kim J, Park C, Sun Y (2003) Solid State Ionics 164:43
Huang SH, Wen ZY, Zhang JC, Gu ZH, Xu XH (2006) Solid State Ionics 177:851
Dominko R, Gaberscek M, Bele U, Mihailovic D, Jamnik J (2007) J Eur Ceram Soc 27:909
Liu H, Feng Y, Wang K, Xie JY (2008) J Phys Chem Solids 69:2037
Huang JJ, Jiang ZY (2008) Electrochim Acta 53:7756
Yang LZ, Gao LJ (2009) J Alloys Comp 485:93
Wolfenstine J, Lee U, Allen JL (2006) J. Power Sources 154:287
Liu DT, Quyang CY, Shu J, Jiang J, Wang ZX, Chen LQ (2006) Phys Stat Sol (b) 243:1935
Hao YJ, Lai QY, Lu JZ, Ji XY (2007) Ionics 13:369
Kubiak P, Garcia A, Womes M, Aldon L, Olivier-Fourcade J, Lippens PE, Jumas JC (2003) J Power Sources 119:626
Huang SH, Wen ZY, Gu ZH, Zhu XJ (2005) Electrochim Acta 50:4057
Huang SH, Wen ZY, Zhu XJ, Lin ZX (2007) J Power Sources 165:408
Yu HY, Zhang XF, Jalbout AF, Yan XD, Pan XM, Xie HM, Wang RS (2008) Electrochim Acta 53:4200
Tabuchi T, Yasuda H, Yamachi M (2006) J Power Sources 162:813
Wolfenstine J, Allen JL (2008) J Power Sources 180:582
Wu Y, Manthiram A (2009) Solid State Ionics 180:50
Liu J, Manthiram A (2009) Chem Mater 21:1695
Lee MH, Kang YJ, Myung ST, Sun YK (2004) Electrochem Acta 50:939
Cho TH, Park SM, Yoshio M (2004) Chem Lett 700
Huang ZD, Liu XM, Zhang B, Oh S, Ma PC, Kim JK (2011) Scripta Mater 64:122
Kim GH, Myung ST, Kim HS, Sun YK (2006) Electrochim Acta 51:2447
Chang Z, Chen Z, Wu F, Tang H, Zhu Z, Yuan ZZ, Wang H (2008) Electochim Acta 53:5927
Zhang S, Deng C, Fu BL, Yang SY, Ma L (2010) Powder Technol 198:373
Acknowledgment
This study was supported by KBSI Grant (T31601) to T. E. Hong.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hong, T.E., Jeong, E.D., Baek, S.R. et al. Nano SIMS characterization of boron- and aluminum-coated LiNi1/3Co1/3Mn1/3O2 cathode materials for lithium secondary ion batteries. J Appl Electrochem 42, 41–46 (2012). https://doi.org/10.1007/s10800-011-0369-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10800-011-0369-x