Abstract
Background
Studies have uncovered LCN2 as a marker of inflammation strongly related to obesity, insulin resistance, and abnormal glucose metabolism in humans, and is involved in vascular diseases, inflammatory diseases, and neurological diseases. In recent years, studies have shown that elevated levels of LCN2 have a strong association with diabetic retinopathy (DR), but the pathogenesis is unknown. Here, we reviewed the relevant literature and compiled the pathogenesis associated with LCN2-induced DR.
Methods
We searched PubMed and Web of Science electronic databases using “lipocalin-2, diabetic retinopathy, retinal degeneration, diabetic microangiopathies, diabetic neuropathy and inflammation” as subject terms.
Results
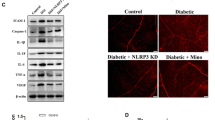
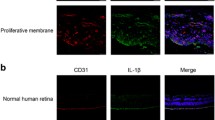
In diabetic retinal neuropathy, LCN2 causes impaired retinal photoreceptor function and retinal neurons; in retinal microangiopathy, LCN2 induces apoptosis of retinal vascular endothelial cells and promotes angiogenesis; in retinal inflammation, increased secretion of LCN2 recruits inflammatory cells and induces pro-inflammatory cytokines. Moreover, LCN2 has the potential as a biomarker for DR. Recent studies have shown that retinal damage can be attenuated by silencing LCN2, which may be associated with the inhibition of caspase-1-mediated pyroptosis, and LCN2 may be a new target for the treatment of DR.
Conclusions
In conclusion, LCN2, involved in the development of diabetic retinopathy, is a key factor in diabetic retinal microangiopathy, neurodegeneration, and retinal inflammation. LCN2 is likely to be a novel molecular target leading to DR, and a more in-depth study of the pathogenesis of DR caused by LCN2 may provide considerable benefits for clinical research and potential drug development.

Similar content being viewed by others
References
Olsen TW, Al-Rajhi A, Ambrus A et al (2020) Diabetic retinopathy preferred practice pattern (R). Ophthalmology 127:P66–P145. https://doi.org/10.1016/j.ophtha.2019.09.025
Cheung N, Mitchell P, Wong TY (2010) Diabetic retinopathy. Lancet 376:124–136. https://doi.org/10.1016/S0140-6736(09)62124-3
Heng LZ, Comyn O, Peto T et al (2013) Diabetic retinopathy: pathogenesis, clinical grading, management and future developments. Diabet Med 30:640–650. https://doi.org/10.1111/dme.12089
Kollias AN, Ulbig MW (2010) Diabetic retinopathy: early diagnosis and effective treatment. Dtsch Arztebl Int. https://doi.org/10.3238/arztebl.2010.0075
Wong TY, Cheung CMG, Larsen M et al (2016) Diabetic retinopathy. Nat Rev Dis Primers 2:16012. https://doi.org/10.1038/nrdp.2016.12
Hammes H-P (2018) Diabetic retinopathy: hyperglycaemia, oxidative stress and beyond. Diabetologia 61:29–38. https://doi.org/10.1007/s00125-017-4435-8
Simó R, Stitt AW, Gardner TW (2018) Neurodegeneration in diabetic retinopathy: does it really matter? Diabetologia 61:1902–1912. https://doi.org/10.1007/s00125-018-4692-1
Lu F, Inoue K, Kato J et al (2019) Functions and regulation of lipocalin-2 in gut-origin sepsis: a narrative review. Crit Care 23:269. https://doi.org/10.1186/s13054-019-2550-2
Guardado S, Ojeda-Juárez D, Kaul M et al (2021) Comprehensive review of lipocalin 2-mediated effects in lung inflammation. Am J Physiol Lung Cell Mol Physiol 321:L726–L733. https://doi.org/10.1152/ajplung.00080.2021
Wang Y, Lam KSL, Kraegen EW et al (2007) Lipocalin-2 is an inflammatory marker closely associated with obesity, insulin resistance, and hyperglycemia in humans. Clin Chem 53:34–41
Jenkins AJ, Joglekar MV, Hardikar AA et al (2015) Biomarkers in diabetic retinopathy. Rev Diabet Stud 12:159–195. https://doi.org/10.1900/RDS.2015.12.159
Chou JC, Rollins SD, Ye M et al (2014) Endothelin receptor-A antagonist attenuates retinal vascular and neuroretinal pathology in diabetic mice. Invest Ophthalmol Vis Sci 55:2516–2525. https://doi.org/10.1167/iovs.13-13676
Chung T-W, Choi H-J, Kim C-H et al (2013) Lipocalin-2 elicited by advanced glycation end-products promotes the migration of vascular smooth muscle cells. Biochim Biophys Acta 1833:3386–3395. https://doi.org/10.1016/j.bbamcr.2013.10.011
Bhusal A, Lee W-H, Suk K (2021) Lipocalin-2 in diabetic complications of the nervous system: physiology, pathology, and beyond. Front Physiol 12:638112. https://doi.org/10.3389/fphys.2021.638112
Ghosh S, Shang P, Yazdankhah M et al (2017) Activating the AKT2-nuclear factor-κB-lipocalin-2 axis elicits an inflammatory response in age-related macular degeneration. J Pathol 241:583–588. https://doi.org/10.1002/path.4870
Koban Y, Sahin S, Boy F et al (2019) Elevated lipocalin-2 level in aqueous humor of patients with central retinal vein occlusion. Int Ophthalmol 39:981–986. https://doi.org/10.1007/s10792-018-0894-2
Wang H, Lou H, Li Y et al (2020) Elevated vitreous Lipocalin-2 levels of patients with proliferative diabetic retinopathy. BMC Ophthalmol 20:260. https://doi.org/10.1186/s12886-020-01462-5
Chung JO, Park SY, Cho DH et al (2016) Plasma neutrophil gelatinase-associated lipocalin levels are positively associated with diabetic retinopathy in patients with Type 2 diabetes. Diabet Med 33:1649–1654. https://doi.org/10.1111/dme.13141
Lee S, Park J-Y, Lee W-H et al (2009) Lipocalin-2 is an autocrine mediator of reactive astrocytosis. J Neurosci 29:234–249. https://doi.org/10.1523/JNEUROSCI.5273-08.2009
Zhao P, Stephens JM (2013) STAT1, NF-κB and ERKs play a role in the induction of lipocalin-2 expression in adipocytes. Mol Metab 2:161–170. https://doi.org/10.1016/j.molmet.2013.04.003
Moscowitz AE, Asif H, Lindenmaier LB et al (2019) The importance of melanocortin receptors and their agonists in pulmonary disease. Front Med (Lausanne) 6:145. https://doi.org/10.3389/fmed.2019.00145
Devireddy LR, Gazin C, Zhu X et al (2005) A cell-surface receptor for lipocalin 24p3 selectively mediates apoptosis and iron uptake. Cell 123:1293–1305
Li D, Yan Sun W, Fu B et al (2020) Lipocalin-2-The myth of its expression and function. Basic Clin Pharmacol Toxicol 127:142–151. https://doi.org/10.1111/bcpt.13332
Frances L, Tavernier G, Viguerie N (2021) Adipose-derived lipid-binding proteins: the good, the bad and the metabolic diseases. Int J Mol Sci. https://doi.org/10.3390/ijms221910460
Skrypnyk NI, Gist KM, Okamura K et al (2020) IL-6-mediated hepatocyte production is the primary source of plasma and urine neutrophil gelatinase-associated lipocalin during acute kidney injury. Kidney Int 97:966–979. https://doi.org/10.1016/j.kint.2019.11.013
Simó R, Hernández C (2014) Neurodegeneration in the diabetic eye: new insights and therapeutic perspectives. Trends Endocrinol Metab 25:23–33. https://doi.org/10.1016/j.tem.2013.09.005
Makris K, Stefani D, Makri E et al (2015) Evaluation of a particle enhanced turbidimetric assay for the measurement of neutrophil gelatinase-associated lipocalin in plasma and urine on Architect-8000: analytical performance and establishment of reference values. Clin Biochem 48:1291–1297. https://doi.org/10.1016/j.clinbiochem.2015.08.003
Jessen BA, Stevens GJ (2002) Expression profiling during adipocyte differentiation of 3T3-L1 fibroblasts. Gene 299(1–2):95–100
Zhao P, Elks CM, Stephens JM (2014) The induction of lipocalin-2 protein expression in vivo and in vitro. J Biol Chem 289:5960–5969. https://doi.org/10.1074/jbc.M113.532234
Mosialou I, Shikhel S, Liu J-M et al (2017) MC4R-dependent suppression of appetite by bone-derived lipocalin 2. Nature 543:385–390. https://doi.org/10.1038/nature21697
Wen Y, Chen X, Feng H et al (2022) Kdm6a deficiency in microglia/macrophages epigenetically silences Lcn2 expression and reduces photoreceptor dysfunction in diabetic retinopathy. Metabolism 136:155293. https://doi.org/10.1016/j.metabol.2022.155293
Opdenakker G, Abu E-A (2019) Metalloproteinases mediate diabetes-induced retinal neuropathy and vasculopathy. Cell Mol Life Sci 76:3157–3166. https://doi.org/10.1007/s00018-019-03177-3
Srdelić Mihalj S, Kuzmić-Prusac I, Zekić-Tomaš S et al (2015) Lipocalin-2 and matrix metalloproteinase-9 expression in high-grade endometrial cancer and their prognostic value. Histopathology 67:206–215. https://doi.org/10.1111/his.12633
Mohammad G, Vandooren J, Siddiquei MM et al (2014) Functional links between gelatinase B/matrix metalloproteinase-9 and prominin-1/CD133 in diabetic retinal vasculopathy and neuropathy. Prog Retin Eye Res 43:76–91. https://doi.org/10.1016/j.preteyeres.2014.07.002
Eidinger O, Leibu R, Newman H et al (2015) An intronic deletion in the PROM1 gene leads to autosomal recessive cone-rod dystrophy. Mol Vis 21:1295–1306
Luo Y, Wan J, Luo C et al (2021) Higher aqueous levels of resistin and lipocalin-2 indicated worse visual improvement following anti-VEGF therapy in patients with retinal vein occlusion. Curr Eye Res 46:845–854. https://doi.org/10.1080/02713683.2020.1842462
Parmar T, Parmar VM, Arai E et al (2016) Acute stress responses are early molecular events of retinal degeneration in Abca4-/-Rdh8-/-mice after light exposure. Invest Ophthalmol Vis Sci 57:3257–3267. https://doi.org/10.1167/iovs.15-18993
Tonade D, Kern TS (2021) Photoreceptor cells and RPE contribute to the development of diabetic retinopathy. Prog Retin Eye Res 83:100919. https://doi.org/10.1016/j.preteyeres.2020.100919
Strauss O (2005) The retinal pigment epithelium in visual function. Physiol Rev 85:845–881
Costa GN, Vindeirinho J, Cavadas C et al (2012) Contribution of TNF receptor 1 to retinal neural cell death induced by elevated glucose. Mol Cell Neurosci 50:113–123. https://doi.org/10.1016/j.mcn.2012.04.003
Su X, Zhou P, Qi Y (2022) Down-regulation of LCN2 attenuates retinal vascular dysfunction and caspase-1-mediated pyroptosis in diabetes mellitus. Ann Transl Med 10:695. https://doi.org/10.21037/atm-22-2655
Wang X, Li H, Li W et al (2020) The role of Caspase-1/GSDMD-mediated pyroptosis in Taxol-induced cell death and a Taxol-resistant phenotype in nasopharyngeal carcinoma regulated by autophagy. Cell Biol Toxicol 36:437–457. https://doi.org/10.1007/s10565-020-09514-8
Zhao S, Huang Z, Jiang H et al (2022) Sirtuin 1 induces choroidal neovascularization and triggers age-related macular degeneration by promoting LCN2 through SOX9 deacetylation. Oxid Med Cell Longev 2022:1671438. https://doi.org/10.1155/2022/1671438
Wang W, Lo ACY (2018) Diabetic retinopathy: pathophysiology and treatments. Int J Mol Sci. https://doi.org/10.3390/ijms19061816
Moschen AR, Adolph TE, Gerner RR et al (2017) Lipocalin-2: a master mediator of intestinal and metabolic inflammation. Trends Endocrinol Metab 28:388–397. https://doi.org/10.1016/j.tem.2017.01.003
Yang Q, Graham TE, Mody N et al (2005) Serum retinol binding protein 4 contributes to insulin resistance in obesity and type 2 diabetes. Nature 436:356–362
Han W, Wei H, Kong W et al (2020) Association between retinol binding protein 4 and diabetic retinopathy among type 2 diabetic patients: a meta-analysis. Acta Diabetol 57:1203–1218. https://doi.org/10.1007/s00592-020-01535-3
Farjo KM, Farjo RA, Halsey S et al (2012) Retinol-binding protein 4 induces inflammation in human endothelial cells by an NADPH oxidase-and nuclear factor kappa B-dependent and retinol-independent mechanism. Mol Cell Biol 32:5103–5115. https://doi.org/10.1128/MCB.00820-12
Burguillos MA, Deierborg T, Kavanagh E et al (2011) Caspase signalling controls microglia activation and neurotoxicity. Nature 472:319–324. https://doi.org/10.1038/nature09788
Rashad NM, El-Shal AS, Etewa RL et al (2017) Lipocalin-2 expression and serum levels as early predictors of type 2 diabetes mellitus in obese women. IUBMB Life 69:88–97. https://doi.org/10.1002/iub.1594
Parmar T, Parmar VM, Perusek L et al (2018) Lipocalin 2 plays an important role in regulating inflammation in retinal degeneration. J Immunol 200:3128–3141. https://doi.org/10.4049/jimmunol.1701573
Batsos G, Christodoulou E, Christou EE et al (2022) Vitreous inflammatory and angiogenic factors on patients with proliferative diabetic retinopathy or diabetic macular edema: the role of Lipocalin2. BMC Ophthalmol 22:496. https://doi.org/10.1186/s12886-022-02733-z
Zhao M, Gelize E, Levy R et al (2021) Mineralocorticoid receptor pathway and its antagonism in a model of diabetic retinopathy. Diabetes 70:2668–2682. https://doi.org/10.2337/db21-0099
Funding
The authors have not disclosed any funding. A funding declaration is mandatory for publication in this journal.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Literature collection and analysis were performed by YZ, XS and TQ. The first draft of the manuscript was written by YZ, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, Y., Song, X., Qi, T. et al. Review of lipocalin-2-mediated effects in diabetic retinopathy. Int Ophthalmol 44, 78 (2024). https://doi.org/10.1007/s10792-024-03015-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10792-024-03015-x