Abstract
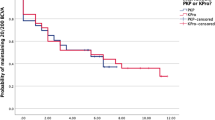
The purpose of this study was to evaluate the outcomes of the Boston type 1 keratoprosthesis (Kpro-1) in eyes with failed keratoplasty. A retrospective review was performed of every patient treated with a Kpro-1 at a tertiary eye care center between January 1, 2008 and July 1, 2013. Eyes with a failed keratoplasty originally performed for corneal edema, trauma, or keratoconus were included in the statistical analysis. The main outcome measures were visual outcome, prosthesis retention, and postoperative complications. Twenty-four eyes met the inclusion criteria, including 13 eyes with corneal edema, 8 eyes with trauma, and 3 eyes with keratoconus. After a mean follow-up period of 28.9 months (range 7–63 months), the median best corrected visual acuity (BCVA) was 20/125. The BCVA was ≥20/40 in 4 (16.7 %) eyes, ≥20/70 in 9 (37.5 %) eyes, and ≥20/200 in 14 (58.3 %) eyes. Overall, the postoperative BCVA improved in 17 (70.9 %) eyes, was unchanged in 3 (12.5 %) eyes, and was worse in 4 (16.7 %) eyes. The initial Kpro-1 was retained in 22 (91.7 %) eyes, and was successfully repeated in the other 2 eyes. One or more serious prosthesis- or sight-threatening complications occurred in 8 (33.3 %) eyes. These included 1 case of wound dehiscence leading to prosthesis extrusion, 1 case of fungal keratitis leading to prosthesis extrusion, 4 cases of endophthalmitis, and 5 retinal detachments. The Boston Kpro-1 is associated with an excellent prognosis for prosthesis retention and satisfactory visual improvement in eyes with previous failed keratoplasty.
Similar content being viewed by others
References
Thompson RW Jr, Price MO, Bowers PJ, Price FW Jr (2003) Long-term survival probability after penetrating keratoplasty. Ophthalmology 110:1396–1402
Sugar A, Tanner JP, Dontchey M, Tennant B, Schultze RL, Dunn SP, Lindquist TD, Gal RL, Beck RW, Kollman C, Mannis MJ, Holland EJ, Cornea Donor Study Investigator Group (2009) Recipient risk factors for graft failure in the cornea donor study. Ophthalmology 116:1023–1028
Arentsen JJ, Donoso R, Laibson PR, Cohen EJ (1987) Penetrating keratoplasty for the treatment of pseudophakic corneal edema associated with posterior chamber lens implantation. Trans Am Ophthalmol Soc 85:393–404
Pineros O, Cohen EJ, Rapuano CJ, Laibson PR (1996) Long-term results after penetrating keratoplasty for Fuchs’ endothelial dystrophy. Arch Ophthalmol 114:15–18
Doren GS, Cohen EJ, Brady SE, Arentsen JJ, Laibson PR (1990) Penetrating keratoplasty after ocular trauma. Am J Ophthalmol 110:408–411
Kirkness CM, Ficker LA, Steele AD, Rice NS (1990) The success of penetrating keratoplasty for keratoconus. Eye 4:673–688
Pramanik S, Musch DC, Sutphin JE, Farjo AA (2006) Extended long-term outcomes of penetrating keratoplasty for keratoconus. Ophthalmology 113:1633–1638
Yaghouti F, Nouri M, Abad JC, Power WJ, Doane MC, Dohlman CH (2001) Keratoprosthesis: preoperative prognostic categories. Cornea 20:19–23
Zerbe BL, Belin MW, Ciolino JB, Boston Type 1 Keratoprosthesis Study Group (2006) Results from the multicenter Boston type 1 keratoprosthesis study. Ophthalmology 113:1779
Aldave AJ, Kamal KM, Vo RC, Yu F (2009) The Boston type I keratoprosthesis: improving outcomes and expanding indications. Ophthalmology 116:640–651
Chew HF, Ayres BD, Hammersmith KM, Rapuano CJ, Laibson PR, Myers JS, Jin YP, Cohen EH (2009) Boston keratoprosthesis outcomes and complications. Cornea 28:989–996
Colby KA, Koo EB (2011) Expanding indications for the Boston keratoprosthesis. Curr Opin Ophthalmol 22:267–273
Robert MC, Harissi-Dagher M (2011) Boston type 1 keratoprosthesis: the CHUM experience. Can J Ophthalmol 46:164–168
Greiner MA, Li JY, Mannis MJ (2011) Longer-term vision outcomes and complications with the Boston type 1 keratoprosthesis at the University of California, Davis. Ophthalmology 118:1543–1550
Patel AP, Wu EI, Ritterband DC, Seedor JA (2012) Boston type 1 keratoprosthesis: the New York eye and ear experience. Eye (Lond) 26:418–425
Aldave AJ, Sangwan V, Basu S, Basak SK, Hovakimyan A, Gevorgyan O, Kharashi SA, Jindan MA, Tandon R, Mascarenhas J, Malyugin B, Padilla MD, Maskati Q, Agarwala N, Hatauruk J, Shamra M, Yu F (2012) International results with the Boston type 1 keratoprosthesis. Ophthalmology 119:1530–1538
Aquavella JV, Qian Y, McCormick GJ, Palakuru JR (2005) Keratoprosthesis: the Dohlman-Doane device. Am J Ophthalmol 140:1032–1038
Birkholz ES, Goins KM (2012) Boston keratoprosthesis: an option for patients with multiple failed corneal grafts. http://webeye.ophth.uiowa.edu/eyeforum/cases/94-Boston-Keratoprosthesis-Failed-Corneal-Grafts.htm. Accessed 26 Mar 2014
Wandling GR, Rauen MP, Goins KM, Kitzmann AS, Sutphin JE, Kwon YH, Alward WL, Wagoner MD (2012) Glaucoma therapy escalation in eyes with pseudophakic corneal edema after penetrating keratoplasty and Descemet’s stripping automated endothelial keratoplasty. Int Ophthalmol 32:9–14
Dohlman CH, Dudenhoefer EJ, Khan BF, Morneault S (2002) Protection of the ocular surface after keratoprosthesis surgery: the role of soft contact lenses. CLAO J 28:72–74
Khan BF, Harissa-Dagher M, Khan DM, Dohlman CH (2007) Advances in Boston keratoprosthesis: enhancing retention and prevention of infection and inflammation. Int Ophthalmol Clin 113(2):61–71
Ward MS, Goins KM, Greiner MA, Kitzmann SA, Sutphin JE, Alward WLM, Greenlee EC, Kwon YH, Zimmerman MD, Wagoner MD (2014) Graft survival versus glaucoma treatment after penetrating or Descemet’s stripping automated endothelial keratoplasty. Cornea 33(8):785–789
Acknowledgments
All authors participated in the experimental design, collections and analysis of the data, preparation of the manuscript, and review and approval of the final document. None of the authors have any financial interests to disclose. There was no external funding or other sources of financial support for this project.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hager, J.L., Phillips, D.L., Goins, K.M. et al. Boston type 1 keratoprosthesis for failed keratoplasty. Int Ophthalmol 36, 73–78 (2016). https://doi.org/10.1007/s10792-015-0078-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10792-015-0078-2