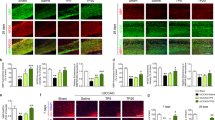
Abstract
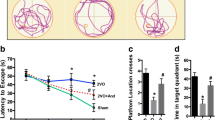
Age-related white matter lesions (WML) frequently present vascular problems by decreasing cerebral blood supply, resulting in the condition known as chronic cerebral hypoperfusion (CCH). This study aimed to investigate the effect of hexahydrocurcumin (HHC) on the processes of demyelination and remyelination induced by the model of the Bilateral Common Carotid Artery Occlusion (BCCAO) for 29 days to mimic the CCH condition. The pathological appearance of myelin integrity was significantly altered by CCH, as evidenced by Transmission Electron Microscopy (TEM) and Luxol Fast Blue (LFB) staining. In addition, CCH activated A1-astrocytes and reactive-microglia by increasing the expression of Glial fibrillary acidic protein (GFAP), complement 3 (C3d) and pro-inflammatory cytokines. However, S100a10 expression, a marker of neuroprotective astrocytes, was suppressed, as were regenerative factors including (IGF-1) and Transglutaminase 2 (TGM2). Therefore, the maturation step was obstructed as shown by decreases in the levels of myelin basic protein (MBP) and the proteins related with lipid synthesis. Cognitive function was therefore impaired in the CCH model, as evidenced by the Morris water maze test. By contrast, HHC treatment significantly improved myelin integrity, and inhibited A1-astrocytes and reactive-microglial activity. Consequently, pro-inflammatory cytokines and A1-astrocytes were attenuated, and regenerative factors increased assisting myelin maturation and hence improving cognitive performance. In conclusion, HHC improves cognitive function and also the integrity of white matter in CCH rats by reducing demyelination, and pro-inflammatory cytokine production and promoting the process of remyelination.
Graphical abstract






Similar content being viewed by others
Availability of data and materials
All data presented in this study are included in the article.
Abbreviations
- 2VO:
-
2 Vessels occlusion
- AD:
-
Alzheimer’s disease
- APOE:
-
Apolipoprotein E
- BBB:
-
Blood–brain barrier
- BCCAO:
-
Bilateral common carotid artery occlusion
- C3d:
-
Complement 3
- CBF:
-
Cerebral blood flow
- CCA:
-
Common carotid artery occlusion
- CCH:
-
Chronic cerebral hypoperfusion
- CNS:
-
Central nervous system
- GFAP:
-
Glial fibrillary acidic protein
- HHC:
-
Hexahydrocurcumin
- Iba-1:
-
Ionized calcium-binding adaptor molecule 1
- IL-1β:
-
Interleukin 1 beta
- IGF-1:
-
Insulin-like growth factor 1
- MBP:
-
Myelin basic protein
- MMP-9:
-
Matrix metalloproteinase 9
- mTOR:
-
Mammalian target of rapamycin
- OPC:
-
Oligodendrocyte precursor cells
- SREBP:
-
Sterol regulatory element-binding protein
- TGM2:
-
Transglutaminase 2
- TNF-α:
-
Tumor necrosis factor alpha
- WML:
-
White matter lesion
- VaD:
-
Vascular dementia
References
Akiguchi I, Tomimoto H, Suenaga T, Wakita H, Budka H (1997) Alterations in glia and axons in the brains of Binswanger’s disease patients. Stroke 28(7):1423–1429. https://doi.org/10.1161/01.STR.28.7.1423
Anand P, Thomas SG, Kunnumakkara AB et al (2008) Biological activities of curcumin and its analogues (Congeners) made by man and mother nature. Biochem Pharmacol 76(11):1590–1611. https://doi.org/10.1016/j.bcp.2008.08.008
Attems J, Jellinger KA (2014) The overlap between vascular disease and Alzheimer’s disease-lessons from pathology. BMC Med 12(1):1–12. https://doi.org/10.1186/s12916-014-0206-2
Baaklini CS, Rawji KS, Duncan GJ, Ho MF, Plemel JR (2019) Central nervous system remyelination: roles of glia and innate immune cells. Front Mol Neurosci 12:225. https://doi.org/10.3389/fnmol.2019.00225
Behrendt G, Baer K, Buffo A et al (2013) Dynamic changes in myelin aberrations and oligodendrocyte generation in chronic amyloidosis in mice and men. Glia 61(2):273–286. https://doi.org/10.1002/glia.22432
Chida Y, Kokubo Y, Sato S et al (2011) The alterations of oligodendrocyte, myelin in corpus callosum, and cognitive dysfunction following chronic cerebral ischemia in rats. Brain Res 1414:22–31. https://doi.org/10.1016/j.brainres.2011.07.026
Dai J, Bercury KK, Macklin WB (2014) Interaction of mTOR and Erk1/2 signaling to regulate oligodendrocyte differentiation. Glia 62(12):2096–2109. https://doi.org/10.1002/glia.22729
De Jong G, Farkas E, Stienstra C et al (1999) Cerebral hypoperfusion yields capillary damage in the hippocampal CA1 area that correlates with spatial memory impairment. Neuroscience 91(1):203–210. https://doi.org/10.1016/s0306-4522(98)00659-9
Defaux A, Zurich M-G, Honegger P, Monnet-Tschudi F (2011) Minocycline promotes remyelination in aggregating rat brain cell cultures after interferon-γ plus lipopolysaccharide-induced demyelination. Neuroscience 187:84–92. https://doi.org/10.1016/j.neuroscience.2011.04.053
Dempe JS, Scheerle RK, Pfeiffer E, Metzler M (2013) Metabolism and permeability of curcumin in cultured Caco-2 cells. Mol Nutr Food Res 57(9):1543–1549. https://doi.org/10.1002/mnfr.201200113
Di Penta A, Moreno B, Reix S et al (2013) Oxidative stress and proinflammatory cytokines contribute to demyelination and axonal damage in a cerebellar culture model of neuroinflammation. PLoS ONE 8(2):e54722. https://doi.org/10.1371/journal.pone.0054722
Duncombe J, Kitamura A, Hase Y, Ihara M, Kalaria RN, Horsburgh K (2017) Chronic cerebral hypoperfusion: a key mechanism leading to vascular cognitive impairment and dementia. Closing the translational gap between rodent models and human vascular cognitive impairment and dementia. Clin Sci 131(19):2451–2468. https://doi.org/10.1042/CS20160727
Eng LF, Ghirnikar RS (1994) GFAP and astrogliosis. Brain Pathol 4(3):229–237. https://doi.org/10.1111/j.1750-3639.1994.tb00838.x
Fan Y-Y, Huo J (2021) A1/A2 astrocytes in central nervous system injuries and diseases: Angels or devils? Neurochem Int 148:105080. https://doi.org/10.1016/j.neuint.2021.105080
Faulkner JR, Herrmann JE, Woo MJ, Tansey KE, Doan NB, Sofroniew MV (2004) Reactive astrocytes protect tissue and preserve function after spinal cord injury. J Neurosci 24(9):2143–2155. https://doi.org/10.1523/JNEUROSCI.3547-03.2004
Figlia G, Gerber D, Suter U (2018) Myelination and mTOR. Glia 66(4):693–707. https://doi.org/10.1002/glia.23273
Franklin RJ, Ffrench-Constant C (2008) Remyelination in the CNS: from biology to therapy. Nat Rev Neurosci 9(11):839–855. https://doi.org/10.1038/nrn2480
Gaither T, Vargas I, Inada S, Frank M (1987) The complement fragment C3d facilitates phagocytosis by monocytes. Immunology 62(3):405–411
Hu Y, Zhang M, Liu B et al (2023) Honokiol prevents chronic cerebral hypoperfusion induced astrocyte A1 polarization to alleviate neurotoxicity by targeting SIRT3-STAT3 axis. Free Radic Biol Med 202:62–75. https://doi.org/10.1016/j.freeradbiomed.2023.03.018
Hughes AN (2021) Glial cells promote myelin formation and elimination. Front Cell Dev Biol 9:661486. https://doi.org/10.3389/fcell.2021.661486
Imai M, Watanabe M, Suyama K et al (2008) Delayed accumulation of activated macrophages and inhibition of remyelination after spinal cord injury in an adult rodent model. J Neurosurg Spine 8(1):58–66. https://doi.org/10.3171/SPI-08/01/058
Iwama S, Sugimura Y, Suzuki H et al (2011) Time-dependent changes in proinflammatory and neurotrophic responses of microglia and astrocytes in a rat model of osmotic demyelination syndrome. Glia 59(3):452–462. https://doi.org/10.1002/glia.21114
Jaworski J, Sheng M (2006) The growing role of mTOR in neuronal development and plasticity. Mol Neurobiol 34:205–219. https://doi.org/10.1385/MN:34:3:205
Jearjaroen P, Pakdeepak K, Tocharus C, Chaichompoo W, Suksamrarn A, Tocharus J (2021) Inhibitory effect of hexahydrocurcumin on memory impairment and amyloidogenesis in dexamethasone-treated mice. Neurotox Res 39(2):266–276. https://doi.org/10.1007/s12640-020-00269-y
Jearjaroen P, Thangwong P, Tocharus C, Lungkaphin A, Chaichompoo W, Srijun J, Suksamrarn A, Tocharus J (2023) Hexahydrocurcumin attenuates neuronal injury and modulates synaptic plasticity in chronic cerebral hypoperfusion in rats. Mol Neurobiol. https://doi.org/10.1007/s12035-023-03821-x
Jiang T, Luo J, Pan X et al (2021) Physical exercise modulates the astrocytes polarization, promotes myelin debris clearance and remyelination in chronic cerebral hypoperfusion rats. Life Sci 278:119526. https://doi.org/10.1016/j.lfs.2021.119526
Kamholz J, Toffenetti J, Lazzarini RA (1988) Organization and expression of the human myelin basic protein gene. J Neurosci Res 21(1):62–70. https://doi.org/10.1002/jnr.490210110
Konat GW, Wiggins RC (1985) Effect of reactive oxygen species on myelin membrane proteins. J Neurochem 45(4):1113–1118. https://doi.org/10.1111/j.1471-4159.1985.tb05530.x
Konno S, Meyer JS, Terayama Y, Margishvili GM, Mortel KF (1997) Classification, diagnosis and treatment of vascular dementia. Drugs Aging 11(5):361–373. https://doi.org/10.2165/00002512-199711050-00004
Lebrun-Julien F, Bachmann L, Norrmén C et al (2014) Balanced mTORC1 activity in oligodendrocytes is required for accurate CNS myelination. J Neurosci 34(25):8432–8448. https://doi.org/10.1523/JNEUROSCI.1105-14.2014
Li F, Nitteranon V, Tang X, Liang J, Zhang G, Parkin KL et al (2012) In vitro antioxidant and anti-inflammatory activities of 1-dehydro-[6]-gingerdione, 6-shogaol, 6-dehydroshogaol and hexahydrocurcumin. Food Chem 135(2):332–337. https://doi.org/10.1016/j.foodchem.2012.04.145
Li M, Meng N, Guo X et al (2020) Dl-3-n-butylphthalide promotes remyelination and suppresses inflammation by regulating AMPK/SIRT1 and STAT3/NF-κB signaling in chronic cerebral hypoperfusion. Front Aging Neurosci 12:137. https://doi.org/10.3389/fnagi.2020.00137
Li R, Liang Q, Yang Q, Dai W, Xiao Y, Pan H et al (2023) Hexahydrocurcumin from Zingiberis rhizoma attenuates lipopolysaccharide-induced acute pneumonia through JAK1/STAT3 signaling pathway. Phytomedicine 7(122):155141. https://doi.org/10.1016/j.phymed.2023.155141
Lian H, Yang L, Cole A et al (2015) NFκB-activated astroglial release of complement C3 compromises neuronal morphology and function associated with Alzheimer’s disease. Neuron 85(1):101–115. https://doi.org/10.1016/j.neuron.2014.11.018
Liang J, Yue Y, Jiang H et al (2018) Genetic variations in the p11/tPA/BDNF pathway are associated with post stroke depression. J Affect Disord 226:313–325. https://doi.org/10.1016/j.jad.2017.09.055
Liddelow SA, Barres BA (2017) Reactive astrocytes: production, function, and therapeutic potential. Immunity 46(6):957–967. https://doi.org/10.1016/j.immuni.2017.06.006
Liddelow SA, Guttenplan KA, Clarke LE et al (2017) Neurotoxic reactive astrocytes are induced by activated microglia. Nature 541(7638):481–487. https://doi.org/10.1038/nature21029
Liddelow SA, Marsh SE, Stevens B (2020) Microglia and astrocytes in disease: dynamic duo or partners in crime? Trends Immunol 41(9):820–835. https://doi.org/10.1016/j.it.2020.07.006
Lloyd AF, Miron VE (2019) The pro-remyelination properties of microglia in the central nervous system. Nat Rev Neurol 15(8):447–458. https://doi.org/10.1038/s41582-019-0184-2
Mangla R, Kolar B, Almast J, Ekholm SE (2011) Border zone infarcts: pathophysiologic and imaging characteristics. Radiographics 31(5):1201–1214. https://doi.org/10.1148/rg.315105014
McDowell AR, Petrova N, Carassiti D et al (2022) High-resolution quantitative MRI of multiple sclerosis spinal cord lesions. Magn Reson Med 87(6):2914–2921. https://doi.org/10.1002/mrm.29152
Miron VE, Boyd A, Zhao J-W et al (2013) M2 microglia and macrophages drive oligodendrocyte differentiation during CNS remyelination. Nat Neurosci 16(9):1211–1218. https://doi.org/10.1038/nn.3469
Misra UK, Singh SK, Kalita J, Kumar A (2020) Astrocyte activation following nitrous oxide exposure is related to oxidative stress and glutamate excitotoxicity. Brain Res 1730:146645. https://doi.org/10.1016/j.brainres.2020.146645
Miyamoto N, Magami S, Inaba T et al (2020) The effects of A1/A2 astrocytes on oligodendrocyte linage cells against white matter injury under prolonged cerebral hypoperfusion. Glia 68(9):1910–1924. https://doi.org/10.1002/glia.23814
Morales NP, Sirijaroonwong S, Yamanont P, Phisalaphong C (2015) Electron paramagnetic resonance study of the free radical scavenging capacity of curcumin and its demethoxy and hydrogenated derivatives. Biol Pharm Bull 38(10):1478–1483. https://doi.org/10.1248/bpb.b15-00209
Nelson KM, Dahlin JL, Bisson J, Graham J, Pauli GF, Walters MA (2017) The essential medicinal chemistry of curcumin: miniperspective. J Med Chem 60(5):1620–1637. https://doi.org/10.1021/acs.jmedchem.6b00975
Norrmen C, Figlia G, Lebrun-Julien F et al (2014) mTORC1 controls PNS myelination along the mTORC1-RXRγ-SREBP-lipid biosynthesis axis in Schwann cells. Cell Rep 9(2):646–660. https://doi.org/10.1016/j.celrep.2014.09.001
O’Brien JT, Thomas A (2015) Vascular dementia. Lancet 386(10004):1698–1706. https://doi.org/10.1016/S0140-6736(15)00463-8
Panthiya L, Tocharus J, Onsa-Ard A, Chaichompoo W, Suksamrarn A, Tocharus C (2022) Hexahydrocurcumin ameliorates hypertensive and vascular remodeling in L-NAME-induced rats. Biochim Biophys Acta (BBA)-Mol Basis Dis 1868(3):166317. https://doi.org/10.1016/j.bbadis.2021.166317
Park J-A, Lee C-H (2017) Temporal changes in mammalian target of rapamycin (mTOR) and phosphorylated-mTOR expressions in the hippocampal CA1 region of rat with vascular dementia. J Vet Sci 18(1):11–16. https://doi.org/10.4142/jvs.2017.18.1.11
Pathak D, Berthet A, Nakamura K (2013) Energy failure: does it contribute to neurodegeneration? Ann Neurol 74(4):506–516. https://doi.org/10.1002/ana.24014
Perez-Alvarez MJ, Villa Gonzalez M, Benito-Cuesta I, Wandosell FG (2018) Role of mTORC1 controlling proteostasis after brain ischemia. Front Neurosci 12:60. https://doi.org/10.3389/fnins.2018.00060
Peterson TR, Sengupta SS, Harris TE et al (2011) mTOR complex 1 regulates lipin 1 localization to control the SREBP pathway. Cell 146(3):408–420. https://doi.org/10.1016/j.cell.2011.06.034
Poh L, Sim WL, Jo D-G et al (2022) The role of inflammasomes in vascular cognitive impairment. Mol Neurodegener 17(1):4. https://doi.org/10.1186/s13024-021-00506-8
Rosenberg GA (2009) Inflammation and white matter damage in vascular cognitive impairment. Stroke 40(3 suppl):S20–S23. https://doi.org/10.1161/STROKEAHA.108.533133
Rosenberg GA, Sullivan N, Esiri MM (2001) White matter damage is associated with matrix metalloproteinases in vascular dementia. Stroke 32(5):1162–1168. https://doi.org/10.1161/01.str.32.5.1162
Shimano H, Sato R (2017) SREBP-regulated lipid metabolism: convergent physiology—divergent pathophysiology. Nat Rev Endocrinol 13(12):710–730. https://doi.org/10.1038/nrendo.2017.91
Su S-H, Wu Y-F, Lin Q, Wang D-P, Hai J (2019) URB597 protects against NLRP3 inflammasome activation by inhibiting autophagy dysfunction in a rat model of chronic cerebral hypoperfusion. J Neuroinflammation 16(1):1–15. https://doi.org/10.1186/s12974-019-1668-0
Thangwong P, Jearjaroen P, Govitrapong P, Tocharus C, Tocharus J (2022) Melatonin improves cognitive function by suppressing endoplasmic reticulum stress and promoting synaptic plasticity during chronic cerebral hypoperfusion in rats. Biochem Pharmacol 198:114980. https://doi.org/10.1016/j.bcp.2022.114980
Vorhees CV, Williams MT (2006) Morris water maze: procedures for assessing spatial and related forms of learning and memory. Nat Protoc 1(2):848–858. https://doi.org/10.1038/nprot.2006.116
Wakita H, Tomimoto H, Akiguchi I, Kimura J (1994) Glial activation and white matter changes in the rat brain induced by chronic cerebral hypoperfusion: an immunohistochemical study. Acta Neuropathol 87:484–492. https://doi.org/10.1007/BF00294175
Wicha P, Tocharus J, Janyou A, Jittiwat J, Changtam C, Suksamrarn A et al (2017) Hexahydrocurcumin protects against cerebral ischemia/reperfusion injury, attenuates inflammation, and improves antioxidant defenses in a rat stroke model. PLoS ONE 12(12):e0189211. https://doi.org/10.1371/journal.pone.0189211
Wicha P, Tocharus J, Janyou A, Jittiwat J, Chaichompoo W, Suksamrarn A et al (2020) Hexahydrocurcumin alleviated blood–brain barrier dysfunction in cerebral ischemia/reperfusion rats. Pharmacol Rep 72:659–671. https://doi.org/10.1007/s43440-019-00050-9
Zamanian JL, Xu L, Foo LC et al (2012) Genomic analysis of reactive astrogliosis. J Neurosci 32(18):6391–6410. https://doi.org/10.1523/JNEUROSCI.6221-11.2012
Zhang L-Y, Pan J, Mamtilahun M et al (2020) Microglia exacerbate white matter injury via complement C3/C3aR pathway after hypoperfusion. Theranostics 10(1):74. https://doi.org/10.7150/thno.35841
Zhou D, Meng R, Li SJ et al (2018) Advances in chronic cerebral circulation insufficiency. CNS Neurosci Ther 24(1):5–17. https://doi.org/10.1111/cns.12780
Funding
This work was supported by the Royal Golden Jubilee Ph.D. program (NRCT5-RGJ63004-076) and Thailand Science Research and Innovation (TSRI) (RUG6290031), Thailand. Partial support from Functional Food Research Center for Well-Being, Multidisciplinary Research Institute, Chiang Mai University and the Center of Excellence for Innovation in Chemistry (PERCH-CIC), Ministry of Higher Education, Science, Research and Innovation is gratefully acknowledged.
Author information
Authors and Affiliations
Contributions
PJ: conducted the experiments, performed data analysis, writing—original draft, review & editing. PT: conducted the experiments. CT: performed data analysis, and writing—review & editing. WC: preparation of hexahydrocurcumin. AS: preparation of hexahydrocurcumin, and writing—review & editing. JT: conceived and designed the experiments, performed data analysis, writing—original draft, review & editing, and project administration.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethical approval
All experimental procedures were approved by the Institutional Animal Care and Use Committee at the Faculty of Medicine, Chiang Mai University (Permit number: 31/2563) and performed in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jearjaroen, P., Thangwong, P., Tocharus, C. et al. Hexahydrocurcumin attenuated demyelination and improved cognitive impairment in chronic cerebral hypoperfusion rats. Inflammopharmacol 32, 1531–1544 (2024). https://doi.org/10.1007/s10787-023-01406-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10787-023-01406-7