Abstract
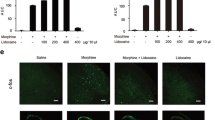
Artemisinin (ART) was initially described for the control of inflammation and pain. However, the mechanisms involved with its antinociceptive effect are still poorly understood. Thus, this present study aimed to investigate the effect of ART in both free and nanocapsulated form on postoperative pain, as well as the participation of the spinal Toll-like receptor 4 (TLR4) in this process. Postoperative pain was induced using the skin/muscle incision retraction (SMIR) model in male Swiss mice. After 3 and 28 days of SMIR, the animals received an intrathecal injection of free or nanocapsulated ART, and the nociceptive threshold was evaluated by von Frey filament test. To evaluate the involvement of the microglia, astrocytes, and TLR4, minocycline (a microglia inhibitor), fluorocitrate (an astrocyte inhibitor), and Lipopolysaccharide Rhodobacter sphaeroides (LPS-RS), a TLR4 antagonist, were intrathecally injected on the third day of SMIR. The levels of spinal TLR4 protein and proinflammatory cytokines tumor necrosis factor-alpha (TNF-α), and interleukin-1-beta (IL-1β) were quantified by western blot and enzyme-linked immunosorbent assay, respectively. The results showed that free ART reduced postoperative pain (P < 0.001, F5,30 = 7.49, 16.66% for 1000 ng dose; and P < 0.01, F5,30 = 7.49, 14.58% for 500 ng dose) on the 3rd day of SMIR; while the ART nanocapsule had this effect on both the third (P < 0.001; F5,30 = 4.94; 43.75, 39.58 and 72.91% for the 250, 500 and 1000 ng doses, respectively) and 28th (P < 0.05; F5,30 = 7.71; 29.16 and 33.33% for the 500 and 1000 ng doses, respectively) day. The ART nanocapsule had a more potent and longer antinociceptive effect than free ART or morphine. Postoperative pain was also reduced by minocycline and LPS-RS. The ART nanocapsule also reduced the increased levels of TLR4, TNF-α, and IL-1β induced by SMIR. These data suggest that the ART nanocapsule has a potent analgesic effect on postoperative pain at the spinal level, and this response involves the inhibition of TLR4 and the proinflammatory cytokines TNF-α and IL-1β.









Similar content being viewed by others
Availability of data and material
The data and material that support the findings of this study are available from the corresponding author, upon reasonable request.
Code availability
Not applicable.
References
Apfelbaum JL, Chen C, Mehta SS, Gan TJ (2003) Postoperative pain experience: results from a national survey suggest postoperative pain continues to be undermanaged. Anesth Analg 97:534–540. https://doi.org/10.1213/01.ANE.0000068822.10113.9E
Charlie-Silva I, Fraceto LF, de Melo NFS (2018) Progress in nano-drug delivery of artemisinin and its derivatives: towards to use in immunomodulatory approaches. Artif Cells Nanomed Biotechnol 46:S611–S620. https://doi.org/10.1080/21691401.2018.1505739
Chen G, Luo X, Qadri MY, Berta T, Ji RR (2018) Sex-Dependent glial signaling in pathological pain: distinct roles of spinal microglia and astrocytes. Neurosci Bull 34:98–108. https://doi.org/10.1007/s12264-017-0145-y
Correll D (2017) Chronic postoperative pain: recent findings in understanding and management. F1000Research 6:1054. https://doi.org/10.12688/f1000research.11101.1
Dai YF, Zhou W-W, Meng J et al (2017) The pharmacological activities and mechanisms of artemisinin and its derivatives: a systematic review. Med Chem Res 26:867–880. https://doi.org/10.1007/s00044-016-1778-5
de Magalhães PM (1996) Seleção, melhoramento e nutrição da Artemisia annua L., para cultivo em região intertropical. Universidade Estadual de Campinas, Campinas
dos Celeghini RMS, de Sousa IMO, da Silva AP, Rodrigues RAF, Foglio MA (2009) Desenvolvimento e validação de metodologia analítica por CLAE-IR para determinação de artemisinina em Artemisia annua L. Quim Nova 32:875–878. https://doi.org/10.1590/S0100-40422009000400009
Dos Santos RS, Veras FP, Ferreira DW et al (2020) Involvement of the Hsp70/TLR4/IL-6 and TNF-α pathways in delayed-onset muscle soreness. J Neurochem. https://doi.org/10.1111/jnc.15006.10.1111/jnc.15006
Eidson LN, Murphy AZ (2013) Blockade of toll-like receptor 4 attenuates morphine tolerance and facilitates the pain relieving properties of morphine. J Neurosci 33:15952–15963. https://doi.org/10.1523/JNEUROSCI.1609-13.2013
Fang RH, Kroll AV, Gao W, Zhang L (2018) Cell membrane coating nanotechnology. Adv Mater 30:1706759. https://doi.org/10.1002/adma.201706759
Favero FF, Grando R, Nonato FR et al (2014) Artemisia annua L.: evidence of sesquiterpene lactones’ fraction antinociceptive activity. BMC Complement Altern Med 14:266. https://doi.org/10.1186/1472-6882-14-266
Flatters SJL (2008) Characterization of a model of persistent postoperative pain evoked by skin/muscle incision and retraction (SMIR). Pain 135:119–130. https://doi.org/10.1016/j.pain.2007.05.013
Fu JC (2017) Artemisinin and Chinese medicine as Tu science. Endeavour 41:127–135. https://doi.org/10.1016/j.endeavour.2017.06.005
Gerbershagen HJ, Pogatzki-Zahn E, Aduckathil S et al (2014) Procedure-specific risk factor analysis for the development of severe postoperative pain. Surv Anesthesiol 58:315–316. https://doi.org/10.1097/01.SA.0000455108.38947.b8
Gosselin RD, Suter MR, Ji RR, Decosterd I (2010) Glial cells and chronic pain. Neuroscience 16:519–531. https://doi.org/10.1177/1073858409360822
Grillo R, Rosa AH, Fraceto LF (2014) Poly(ε-caprolactone) nanocapsules carrying the herbicide atrazine: effect of chitosan-coating agent on physico-chemical stability and herbicide release profile. Int J Environ Sci Technol 11:1691–1700. https://doi.org/10.1007/s13762-013-0358-1
Haight ES, Forman TE, Cordonnier SA, James ML, Tawfik VL (2019) Microglial modulation as a target for chronic pain: from the bench to the bedside and back. Anesth Analg 128:737–746. https://doi.org/10.1016/j.neuron.2018.11.009
Hylden JLK, Wilcox GL (1980) Intrathecal morphine in mice: a new technique. Eur J Pharmacol 67:313–316. https://doi.org/10.1016/0014-2999(80)90515-4
Ji RR, Chamessian A, Zhang YQ (2016) Pain regulation by non-neuronal cells and inflammation. Science 354:572–577. https://doi.org/10.1126/science.aaf8924
Jin SX, Zhuang ZY, Woolf CJ, Ji RR (2003) p38 mitogen-activated protein kinase is activated after a spinal nerve ligation in spinal cord microglia and dorsal root ganglion neurons and contributes to the generation of neuropathic pain. J Neurosci 23:4017–4022. https://doi.org/10.1523/jneurosci.23-10-04017.2003
Kawai T, Akira S (2007) TLR signaling. Semin Immunol 19:24–32. https://doi.org/10.1016/j.smim.2006.12.004
Kothamasu P, Kanumur H, Ravur N, Maddu C, Parasuramrajam R, Thangavel S (2012) Nanocapsules: the weapons for novel drug delivery systems. Bioimpacts 2:71–81
Kuzmich NN, Sivak KV, Chubarev VN, Porozov YB, Savateeva-Lyubimova TN, Peri F (2017) TLR4 signaling pathway modulators as potential therapeutics in inflammation and sepsis. Vaccines 5:34
Laflamme N, Rivest S (2001) Toll-like receptor 4: the missing link of the cerebral innate immune response triggered by circulating gram-negative bacterial cell wall components. FASEB J 15:155–163. https://doi.org/10.1096/fj.00-0339com
Lee JY, Ye J, Gao Z et al (2003) Reciprocal modulation of toll-like receptor-4 signaling pathways involving myd88 and phosphatidylinositol 3-Kinase/AKT by saturated and polyunsaturated fatty acids. J Biol Chem 278:37041–37051. https://doi.org/10.1074/jbc.M305213200
Lehnardt S, Massillon L, Follett P et al (2003) Activation of innate immunity in the CNS triggers neurodegeneration through a Toll-like receptor 4-dependent pathway. Proc Natl Acad Sci 100:8514–8519. https://doi.org/10.1073/pnas.1432609100
Lewis SS, Hutchinson MR, Rezvani N et al (2010) Evidence that intrathecal morphine-3-glucuronide may cause pain enhancement via toll-like receptor 4/MD-2 and interleukin-1β. Neuroscience 165:569–583. https://doi.org/10.1016/j.neuroscience.2009.10.011
Mahdian Dehkordi F, Kaboutari J, Zendehdel M, Javdani M (2019) The antinociceptive effect of artemisinin on the inflammatory pain and role of GABAergic and opioidergic systems. Korean J Pain 32:160–167. https://doi.org/10.3344/kjp.2019.32.3.160
Masaki E, Mizuta K, Ohtani N, Kido K (2016) Early postoperative nociceptive threshold and production of brain-derived neurotrophic factor induced by plantar incision are not influenced with minocycline in a rat: role of spinal microglia. Neurosignals 24:15–24. https://doi.org/10.1159/000442608
Meyer RA, Ringkamp M, Campbell JN, Raja SN (2005) Neural mechanisms of hyperalgesia after tissue injury. Johns Hopkins APL Tech Dig Appl Phys Lab 26:56–66
Moraes CM, de Paula E, Rosa AH, Fraceto LF (2010) Physicochemical stability of poly(lactide-co-glycolide) nanocapsules containing the local anesthetic Bupivacaine. J Braz Chem Soc 21:995–1000. https://doi.org/10.1590/S0103-50532010000600008
Moraes TR, Elisei LS, Malta IH, Galdino G (2020) Participation of CXCL1 in the glial cells during neuropathic pain. Eur J Pharmacol 875:173039. https://doi.org/10.1016/j.ejphar.2020.173039
Nicholas M, Vlaeyen JWS, Rief W et al (2019) The IASP classification of chronic pain for ICD-11. Pain 160:28–37. https://doi.org/10.1097/j.pain.0000000000001390
Nicotra L, Loram LC, Watkins LR, Hutchinson MR (2012) Toll-like receptors in chronic pain. Exp Neurol 234:316–329. https://doi.org/10.1016/j.expneurol.2011.09.038
Piccinini AM, Midwood KS (2010) DAMPening inflammation by modulating TLR signalling. Mediat Inflamm 2010:672395
Qiang W, Cai W, Yang Q et al (2018) Artemisinin B improves learning and memory impairment in AD dementia mice by suppressing neuroinflammation. Neuroscience 395:1–12. https://doi.org/10.1016/j.neuroscience.2018.10.041
Reddi D (2016) Preventing chronic postoperative pain. Anaesthesia 71:64–71. https://doi.org/10.1111/anae.13306
Rodrigues RAF, Foglio MA, Boaventura Júnior S, Santos AS, Rehder VLG (2006) Otimização do processo de extração e isolamento do antimalárico artemisinina a partir de Artemisia annua L. Quim Nova 29:368–372. https://doi.org/10.1590/S0100-40422006000200030
Sakakihara M, Imamachi N, Saito Y (2016) Effects of intrathecal κ-opioid receptor agonist on morphine-induced itch and antinociception in mice. Reg Anesth Pain Med 41:69–74. https://doi.org/10.1097/AAP.0000000000000326
Staud R, Robinson ME, Price DD (2007) Temporal summation of second pain and its maintenance are useful for characterizing widespread central sensitization of fibromyalgia patients. J Pain 8:893–901
Sun Y, Yang M, Tang H, Ma Z, Liang Y, Li Z (2015) The over-production of TNF-α via Toll-like receptor 4 in spinal dorsal horn contributes to the chronic postsurgical pain in rat. J Anesth 29:734–740. https://doi.org/10.1007/s00540-015-2011-2
Tanga FY, Nutile-McMenemy N, DeLeo JÁ (2005) The CNS role of Toll-like receptor 4 in innate neuroimmunity and painful neuropathy. Proc Natl Acad Sci USA 102:5856–5861. https://doi.org/10.1073/pnas.0501634102
Tavares MR, de Menezes LR, do Nascimento DF, et al (2016) Polymeric nanoparticles assembled with microfluidics for drug delivery across the blood-brain barrier. Eur Phys J Spec Top 225:779–795. https://doi.org/10.1140/epjst/e2015-50266-2
Tsan MF, Gao B (2004) Cytokine function of heat shock proteins. Am J Physiol Cell Physiol 286:C739–C744. https://doi.org/10.1152/ajpcell.00364.2003
Tu Y (2011) The discovery of artemisinin (qinghaosu) and gifts from Chinese medicine. Nat Med 17:1217–1220. https://doi.org/10.1038/nm.2471
Wang J, Zhou H, Zheng J et al (2006) The antimalarial artemisinin synergizes with antibiotics to protect against lethal live Escherichia coli challenge by decreasing proinflammatory cytokine release. Antimicrob Agents Chemother 50:2420–2427. https://doi.org/10.1128/AAC.01066-05
Weiser TG, Regenbogen SE, Thompson KD et al (2008) An estimation of the global volume of surgery: a modelling strategy based on available data. Lancet 372:139–144. https://doi.org/10.1016/S0140-6736(08)60878-8
Wen YR, Suter MR, Ji RR et al (2009) Activation of p38 mitogen-activated protein kinase in spinal microglia contributes to incision-induced mechanical allodynia. Anesthesiology 110:155–165. https://doi.org/10.1097/aln.0b013e318190bc16
Wen YR, Tan PH, Cheng JK, Liu YC, Ji RR (2016) Role of microglia in neuropathic pain, postoperative pain, and morphine tolerance. J Formos Med Assoc 110:487–494. https://doi.org/10.1016/S0929-6646(11)60074-0
Ying Y-L, Wei X-H, Xu X-B et al (2014) Over-expression of P2X7 receptors in spinal glial cells contributes to the development of chronic postsurgical pain induced by skin/muscle incision and retraction (SMIR) in rats. Exp Neurol 261:836–843. https://doi.org/10.1016/j.expneurol.2014.09.007
Ying M, Liu H, Zhang T et al (2017) Effect of artemisinin on neuropathic pain mediated by P2X 4 receptor in dorsal root ganglia. Neurochem Int 108:27–33. https://doi.org/10.1016/j.neuint.2017.02.004
Zimmermann M (1983) Ethical guidelines for investigations of experimental pain in conscious animals. Pain 16:109–110. https://doi.org/10.1016/0304-3959(83)90201-4
Acknowledgements
This work was supported by Fundação de Amparo à Pesquisa do Estado de Minas (FAPEMIG), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) [Grant 001] and by Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP) [Grant: 2014/16008-3].
Funding
This work was supported by Fundação de Amparo à Pesquisa do Estado de Minas (FAPEMIG), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) [Grant 001] and by Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP) [Grant: 2014/16008-3].
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there are no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Elisei, L.M.S., Moraes, T.R., Malta, I.H. et al. Antinociception induced by artemisinin nanocapsule in a model of postoperative pain via spinal TLR4 inhibition. Inflammopharmacol 28, 1537–1551 (2020). https://doi.org/10.1007/s10787-020-00756-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10787-020-00756-w