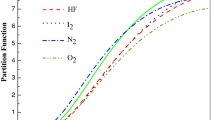
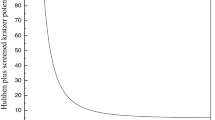
Abstract
In this paper, analytical equation for predicting molar entropy of gaseous substances is presented. The average absolute deviation from experimental data (AAD) is employed as accuracy indicator. The model is used to fit experimental data of four diatomic molecules: ICl (X 1Σg+), BBr (X 1Σ+), NaH (X 1Σ+), and LiH (X 1Σ+). The AADs obtained for the diatomic molecules are 0.0901, 0.2010, 0.5261 and 0.6560%, respectively. The results show that the proposed equation is a near perfect model in predicting experimental data of the diatomic molecules investigated and is approximately equivalent to the improved q-deformed Scarf II potential in modeling molar entropy of the selected diatomic molecules.




Similar content being viewed by others
Availability of Data and Material
Not applicable.
Code Availability
Not applicable.
References
P. Li, Biomed. Eng. Online 18, 30 (2019). https://doi.org/10.1186/s12938-019-0650-5
R.L. Hadimani, in Magnetic Nanostructured Materials, ed. By A.A. El-Gendy, J.M. Barandiaràn, R.L. Hadimani (Elsevier, Amsterdam, 2018), p. 269 https://doi.org/10.1016/B978-0-12-813904-2.00009-7
M. Hasanpour, M. Hatami, Adv. Colloid Interface Sci. 284, 102247 (2020). https://doi.org/10.1016/j.cis.2020.102247
C.S. Jia, L.H. Zhang, X.L. Peng, J.X. Luo, Y.L. Zhao, J.Y. Liu, J.J. Guo, L.D. Tang, Chem. Eng. Sci. 202, 70 (2019). https://doi.org/10.1016/j.ces.2019.03.033
X.L. Peng, R. Jiang, C.S. Jia, L.H. Zhang, Y.L. Zhao, Chem. Eng. Sci. 190, 122 (2018). https://doi.org/10.1016/j.ces.2018.06.027
C.S. Jia, C.W. Wang, L.H. Zhang, X.L. Peng, H.M. Tang, R. Zeng, Chem. Eng. Sci. 183, 26 (2018). https://doi.org/10.1016/j.ces.2018.03.009
R. Jiang, C.S. Jia, Y.Q. Wang, X.L. Peng, L.H. Zhang, Chem. Phys. Lett. 715, 186 (2019). https://doi.org/10.1016/j.cplett.2018.11.044
J. Wang, C.S. Jia, C.J. Li, X.L. Peng, L.H. Zhang, J.Y. Liu, ACS Omega 4, 19193 (2019). https://doi.org/10.1021/acsomega.9b02488
C.S. Jia, J. Li, Y.S. Liu, X.L. Peng, X. Jia, L.H. Zhang, R. Jiang, X.P. Li, J.Y. Liu, Y.L. Zhao, J. Mol. Liq. 315, 113751 (2020). https://doi.org/10.1016/j.molliq.2020.113751
C.W. Wang, J. Wang, Y.S. Liu, X.L. Peng, C.S. Jia, L.H. Zhang, L.Z. Yi, J.Y. Liu, C.S. Jia, X. Jia, J. Mol. Liq. 321, 114912 (2021). https://doi.org/10.1016/j.molliq.2020.114912
C.S. Jia, Y.T. Wang, L.S. Wei, C.W. Wang, X.L. Peng, L.H. Zhang, ACS Omega 4, 20000 (2019). https://doi.org/10.1021/acsomega.9b02950
J.F. Wang, X.L. Peng, L.H. Zhang, C.W. Wang, C.S. Jia, Chem. Phys. Lett. 686, 131 (2017). https://doi.org/10.1016/j.cplett.2017.08.047
C.S. Jia, L.H. Zhang, X.L. Peng, Int. J. Quantum Chem. 117, e25383 (2017). https://doi.org/10.1002/qua.25383
H. Yanar, A. Taş, M. Salti, O. Aydogdu, Eur. Phys. J. Plus 135, 292 (2020). https://doi.org/10.1140/epjp/s13360-020-00297-9
E.S. Eyube, P.P. Notani, M.M. Izam, Mol. Phys. (2021). https://doi.org/10.1080/00268976.2021.1979265
E.S. Eyube, G.G. Nyam, P.P. Notani, Phys. Scr. 96, 125017 (2021). https://doi.org/10.1088/1402-4896/ac2eff
National Institute of Standards and Technology (NIST), NIST Chemistry WebBook, NIST Standard Reference Database Number 69 (2017) https://doi.org/10.18434/T42S31
F.J. Gordillo-Vázquez, J.A. Kunc, J. Appl. Phys. 84, 4693 (1998). https://doi.org/10.1063/1.368712
R. Khordad, A. Ghanbari, Int. J. Thermophys. 42, 115 (2021). https://doi.org/10.1007/s10765-021-02865-2
M.L. Strekalov, Chem. Phys. Lett. 439, 209 (2007). https://doi.org/10.1016/j.cplett.2007.03.052
J.A. Coxon, M.A. Wicramaaratchi, J. Mol. Spectrosc. 79, 380 (1980). https://doi.org/10.1016/0022-2852(80)90220-9
N.T. Hunt, W.Y. Fan, Z. Liu, P.B. Davies, J. Mol. Spectrosc. 191, 326 (1998). https://doi.org/10.1006/jmsp.1998.7632
P.G. Hajigeorgiou, J. Mol. Spectrosc. 263, 101 (2010). https://doi.org/10.1016/j.jms.2010.07.003
R. Horchani, S.A. Shafil, H. Friha, H. Jelassi, Int. J. Thermophys. 42, 84 (2021). https://doi.org/10.1007/s10765-021-02839-4
M. Habibinejad, R. Khordad, A. Ghanbari, Physica B 613, 412940 (2021). https://doi.org/10.1016/j.physb.2021.412940
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
Eyube E.S.: Conceptualization, methodology, validation, data curation, formal analysis, project administration, supervision, writing – original draft, writing – review and editing. B.M. Bitrus: formal analysis, validation, data curation, writing – review and editing. H. Samaila: methodology, validation, data curation, visualization, supervision, writing – review and editing. P.P. Notani: Methodology, formal analysis, validation, data curation, writing – review and editing.
Corresponding author
Ethics declarations
Conflict of interest
We have no known competing financial interest or personal relationships that could have appeared to influence the work reported in this paper.
Ethical Approval
Not applicable.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Eyube, E.S., Bitrus, B.M., Samaila, H. et al. Model Entropy Equation for Gaseous Substances. Int J Thermophys 43, 55 (2022). https://doi.org/10.1007/s10765-022-02980-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10765-022-02980-8