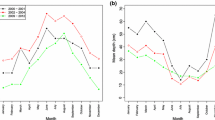
Abstract
There are few examples of host population regulation by macroparasites in stable communities; however, strong impact of parasites on the host individual is obvious in many cases (for example increased mortality, a reduction in fecundity up to the complete castration). Associations between the host populations (periwinkle Littorina obtusata) and prevalence of trematodes was investigated using long-term data (1982–1997) of two L. obtusata populations in the White Sea, northwest Russia. We hypothesized that high prevalence of trematodes will reduce future host population density, and increase mortality. Using a general linear model, we found a significant negative correlation between host population density and the prevalence of the most abundant parasite, Microphallus piriformes in the previous year. We found no correlation between snail reproduction and the prevalence, but observed a significant reduction in middle-aged mollusk abundance which was associated with high prevalence. This indicates the importance of parasite-induced mortality for the dynamics of the host population. There was an association between trematode infection and L. obtusata populations that influence their distribution within littoral zone. The ‘source’ population, located in the lower section of the macrophyte zone, appears to be self-sustaining, controlling the whole population recruitment and dynamics.


Similar content being viewed by others
References
Albon, S. D., A. Stein, R. J. Irvine, R. Langvatn, E. Ropstad & O. Halvorsen, 2002. The role of parasites in the dynamics of a reindeer population. Proceedings of the Royal Society B 269: 1625–1632.
Berger, V. Ya., 1986. Adaptations of the Marine Mollusks to the Salinity of the Environment. Nauka, Leningrad. (in Russian).
Berger, V. Ya., 2007. Production Potential of the White Sea. St. Petersburg Zoological Institute RAS, Saint-Petersburg. (in Russian).
Bostanci, A., 2005. Wildlife biology. A devil of a disease. Science 307: 1035.
Brown, K. M., B. K. Leathers & D. J. Minchella, 1988. Trematode prevalence and the population dynamic of freshwater pond snails. American Midland Naturalist 120: 289–300.
Burnham, K. P. & D. R. Anderson, 2002. Model Selection and Multimodel Inference: A Practical Information—Theoretic Approach, 2nd ed. Springer, New York.
Burnham, K. P. & D. R. Anderson, 2004. Multimodel inference understanding AIC and BIC in model selection. Sociological Methods & Research 33: 261–304.
Daszak, P., A. A. Cunningham & A. D. Hyatt, 2003. Infectious disease and amphibian population declines. Diversity and Distribution 9: 141–150.
Duffy, M. A., 2007. Selective predation, parasitism, and trophic cascades in a bluegill–Daphnia–parasite system. Oecologia 153: 453–460.
Ebert, D., M. Lipsitch & K. L. Mangin, 2000. The effect of parasites on host population density and extinction: experimental epidemiology with Daphnia and six microparasites. American Naturalist 156: 459–477.
Finner, H. & K. Strassburger, 2002. The partitioning principle: a powerful tool in multiple decision theory. Annals of Statistics 30: 1194–1213.
Fredensborg, B. L., K. N. Mouritsen & R. Poulin, 2005. Impact of trematodes on host survival and population density in the intertidal gastropod Zeacumantus subcarinatus. Marine Ecology Progress Series 290: 109–117.
Galaktionov, K. V., 1993. The Life Cycles of Trematodes as the Components of Ecosystems. Kola Scientific Centre, Apatity. (in Russian).
Ganzha, E. V. & A. I. Granovitch, 2008. Infection with trematode partenites leads to change in penial gland structure in males of littoral mollusks Littorina saxatilis (Olivi) и L. obtusata (L.). Parazitologiya 42: 13–22. (in Russian).
Granovitch, A. I., N. A. Mikhailova & S. O. Sergievsky, 1987. Age-specific peculiarities of prevalence of Littorina obtusata and L. saxatilis populations by the Trematode parasites. Parazitologiya 21: 721–729. (in Russian).
Granovitch, A. I., S. O. Sergievsky & I. M. Sokolova, 2000. Spatial and temporal variation of trematode infestation in coexisting populations of intertidal gastropods Littorina saxatilis and L.obtusata in the White Sea. Diseases of Aquatic Organisms 41: 53–64.
Granovitch, A. I., E. V. Yagunova, A. N. Maximovich & I. M. Sokolova, 2009. Elevated female fecundity as a possible compensatory mechanism in response to trematode infestation in populations of Littorina saxatilis (Olivi). International Journal for Parasitology 39: 1011–1019.
Grixti, J. C., L. T. Wong & S. A. Cameron, 2009. Decline of bumble bees (Bombus) in the North American Midwest. Biological Conservation 142: 75–84.
Hernandez, A. D. & M. V. K. Sukhdeo, 2008. Parasites alter the topology of a stream food web across seasons. Oecologia 156: 613–624.
Hudson, P. J., A. P. Dobson & D. Newborn, 1998. Prevention of population cycles by parasite removal. Science 282: 2256–2258.
Huxham, M., D. Raffaelli & A. Pike, 1993. The influence of Cryptocotyle lingua (Digenea: Platyhelminthes) infections on the survival and fecundity of Littorina littorea (Gastropoda: Prosobranchia); an ecological approach. Journal of Experimental Marine Biology and Ecology 168: 223–238.
Jensen, K. H., M. van de Bildt & H. H. Dietz, 2002. Another phocine distemper outbreak in Europe. Science 297: 209.
Jokela, J. & C. M. Lively, 1995. Parasites, sex, and early reproduction in a mixed population of freshwater snails. Evolution 49: 1268–1271.
Kohler, S. L. & W. K. Hoiland, 2001. Population regulation in an aquatic insect: the role of disease. Ecology 82: 2294–2305.
Kohler, S. L. & M. J. Wiley, 1992. Parasite-induced collapse of populations of a dominant grazer in Michigan streams. Oikos 65: 443–449.
Kuznetsov, V. V., 1960. The White Sea and biological peculiarities of its flora and fauna. Academy of Sciences USSR, Moscow. (in Russian).
Lafferty, K. D., 1993. Effect of parasitic castration on growth, reproduction and population dynamics of the marine snail Cerithidea californica. Marine Ecology Progress Series 96: 229–237.
Lauckner, G., 1980. Diseases of Mollusca: Gastropoda. In Kinne, O. (ed.), Diseases of Marine Animals, Vol. 1. Biologische Anstalt Helgoland, Hamburg: 311–424.
Miller, R. G., 1981. Simultaneous Statistical Inference, 2nd ed. Springer, New York.
Poulin, R. & K. N. Mouritsen, 2006. Climate change, parasitism and the structure of intertidal ecosystems. Journal of Helminthology 80: 183–191.
Reid, D. G., 1996. Systematics and Evolution of Littorina. The Ray Society, London.
Schloegel, L. M., J.-M. Hero, L. Berger, R. Speare, K. McDonald & P. Daszak, 2006. The decline of the sharp-snouted day frog (Taudactylus acutirostris): the first documented case of extinction by infection in a free-ranging wildlife species? EcoHealth 3: 35–40.
Schmid-Hempel, P., 2011. Evolutionary Parasitology. Oxford University Press, Oxford.
Sergievsky, S. O., A. I. Granovitch & N. A. Mikhailova, 1986. Impact of trematode invasion on survival of mollusks Littorina obtusata (L.) и L.saxatilis (Olivi) under conditions of extremely low salinity. Parazitologiya 20: 202–207. (in Russian).
Sokolova, I. M., 1995. Influence of trematodes on the demography of Littorina saxatilis (Gastropoda: Prosobranchia: Littorinidae) in the White Sea. Diseases of Aquatic Organisms 21: 91–101.
Sousa, W. P., 1983. Host life history and the effect of parasitic castration on growth: a field study of Cerithidea californica Haldeman (Gastropoda: Prosobranchia) and its trematode parasites. Journal of Experimental Marine Biology and Ecology 73: 273–296.
Tompkins, D. M., A. M. Dunn, M. J. Smith & S. Telfer, 2010. Wildlife diseases: from individuals to ecosystems. Journal of Animal Ecology 80: 19–38.
Acknowledgments
The authors are grateful to the administration of the Kandalaksha Nature Reserve for their support of our long-term study on Rjazhkov Island. We also wish to thank Prof. V. Berger and Prof. A. Sukhotin (White Sea Biological Station of the Zoological Institute of Russian Academy of Sciences) and M. Levitin (Marine Biological Station of St. Petersburg State University) for providing logistic support, Dr. Sergievsky and Dr. Mikhailova who played a key role in the long-term monitoring of Littorina populations in the White Sea, and all the students who helped us during this study. We also thank unknown referees for providing useful criticism and linguistic corrections of the manuscript. During the work on this manuscript the authors were funded in part by the St. Petersburg University Research Grants No. 1.37.80.2011 and No. 1.0.140.2010 and RFBR Grant No. 12-04-00312a.
Author information
Authors and Affiliations
Corresponding author
Additional information
Guest editor: A. A. Sukhotin / Long-term research on marine ecosystems in the White Sea, Russia
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Granovitch, A.I., Maximovich, A.N. Long-term population dynamics of Littorina obtusata: the spatial structure and impact of trematodes. Hydrobiologia 706, 91–101 (2013). https://doi.org/10.1007/s10750-012-1411-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-012-1411-7