Abstract
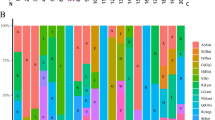
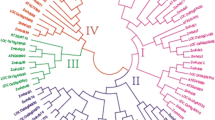
OsGW7 (also known as OsGL7) is homologous to the Arabidopsis thaliana gene that encodes LONGIFOLIA protein, which regulates cell elongation, and is involved in regulating grain length in rice. However, our knowledge on its ortholog in wheat, TaGW7, is limited. In this study, we identified and mapped TaGW7 in wheat, characterized its nucleotide and protein structures, predicted the cis-elements of its promoter, and analysed its expression patterns. The GW7 orthologs in barley (HvGW7), rice (OsGW7), and Brachypodium distachyon (BdGW7) were also identified for comparative analyses. TaGW7 mapped onto the short arms of group 2 chromosomes (2AS, 2BS, and 2DS). Multiple alignments indicated GW7 possesses five exons and four introns in all but two of the species analysed. An exon–intron junction composed of introns 3–4 and exons 4–5 was highly conserved. GW7 has a conserved domain (DUF 4378) and two neighbouring low complexity regions. GW7 was mainly expressed in wheat spikes and stems, in barley seedling crowns, and in rice anthers and embryo-sacs during early development. Drought and heat significantly increased and decreased GW7 expression in wheat, respectively. In barley, GW7 was significantly down-regulated in paleae and awns but up-regulated in seeds under drought treatment and down-regulated under Fusarium and stem rust inoculation. In rice, OsGW7 expression differed significantly under drought treatments. Collectively, these results provide insights into GW7 structure and expression in wheat, barley and rice. The GW7 sequence structure and expression data are the foundation for manipulating GW7 and uncovering its roles in plants.





Similar content being viewed by others
References
Azimzadeh J, Nacry P, Christodoulidou A, Drevensek S, Camilleri C, Amiour N, Parcy F, Pastuglia M, Bouchez D (2008) Arabidopsis TONNEAU1 proteins are essential for preprophase band formation and interact with centrin. Plant Cell 20:2146–2159
Borrill P, Ramirez-Gonzalez R, Uauy C (2016) expVIP: a customisable RNA-seq data analysis and visualisation platform opens up gene expression analysis. Plant Physiol. doi:10.1104/pp.15.01667
Choi HK, Mun JH, Kim DJ, Zhu H, Baek JM, Mudge J, Roe B, Ellis N, Doyle J, Kiss GB (2004) Estimating genome conservation between crop and model legume species. Proc Natl Acad Sci USA 101:15289–15294
Cui F, Zhao C, Ding A, Li J, Wang L, Li X, Bao Y, Li J, Wang H (2014) Construction of an integrative linkage map and QTL mapping of grain yield-related traits using three related wheat RIL populations. Theor Appl Genet 127:659–675
Hu B, Jin J, Guo A-Y, Zhang H, Luo J, Gao G (2014) GSDS 2.0: an upgraded gene feature visualization server. Bioinformatics 31:1296
International Barley Genome Sequencing Consortium (2012) A physical, genetic and functional sequence assembly of the barley genome. Nature 491:711–716
International Wheat Genome Sequencing Consortium (2014) A chromosome-based draft sequence of the hexaploid bread wheat (Triticum aestivum) genome. Science 345:1251788
Jaiswal V, Gahlaut V, Mathur S, Agarwal P, Khandelwal MK, Khurana JP, Tyagi AK, Balyan HS, Gupta PK (2015) Identification of novel SNP in promoter sequence of TaGW2-6A associated with grain weight and other agronomic traits in wheat (Triticum aestivum L.). PLoS ONE 10(6):e0129400. doi:10.1371/journal.pone.0129400
Jiang QT, Liu T, Ma J, Wei YM, Lu ZX, Lan XJ, Dai SF, Zheng YL (2011) Characterization of barley Prp1 gene and its expression during seed development and under abiotic stress. Genetica 139:1283–1292
Keren H, Lev-Maor G, Ast G (2010) Alternative splicing and evolution: diversification, exon definition and function. Nat Rev Genet 11:345–355
Kumar S, Stecher G, Tamura K (2016) MEGA7: molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol Biol Evol. doi:10.1093/molbev/msw1054
Letunic I, Doerks T, Bork P (2015) SMART: recent updates, new developments and status in 2015. Nucleic Acids Res 43:D257–D260
Li Y, Fan C, Xing Y, Jiang Y, Luo L, Sun L, Shao D, Xu C, Li X, Xiao J (2011) Natural variation in GS5 plays an important role in regulating grain size and yield in rice. Nat Genet 43:1266–1269
Li M, Wang Z, Liang Z, Shen W, Sun F, Xi Y, Liu S (2015) Quantitative trait loci analysis for kernel-related characteristics in common wheat (L.). Crop Sci 55:1485–1493
Lin Q, Hao C, Jian H, Wang Y, Tian L, Wang L, Ma Z, Zhang X (2014) Homologous haplotypes, expression, genetic effects and geographic distribution of the wheat yield gene TaGW2. BMC Plant Biol 14:1–19
Long XY, Wang JR, Ouellet T, Rocheleau H, Wei YM, Pu Z, Jiang QT, Lan XJ, Zheng YL (2010) Genome-wide identification and evaluation of novel internal control genes for Q-PCR based transcript normalization in wheat. Plant Mol Biol 74:307–311
Ma J, Jiang Q-T, Zhang X-W, Lan X-J, Pu Z-E, Wei Y-M, Liu C, Lu Z-X, Zheng Y-L (2013a) Structure and expression of barley starch phosphorylase genes. Planta 238:1081–1093
Ma J, Stiller J, Berkman P.J, Wei Y, Rogers J, Feuillet C, Dolezel J, MayerK, Eversole K, Zheng, Y. (2013b). Sequence-based analysis of translocations and inversions in bread wheat (Triticum aestivum L.). PloS ONE 8(11): e79329. doi:10.1371/journal.pone.0079329.
Ma J, Gao S, Jiang QT, Yang Q, Sun M, Wang JR, Qi PF, Liu YX, Li W, Pu ZE (2016) Structure and expression of phosphoglucan phosphatase genes of Like Sex Four1 and Like Sex Four2 in barley. Genetica 144:1–11
Mao H, Sun S, Yao J, Wang C, Yu S, Xu C, Li X, Zhang Q (2010) Linking differential domain functions of the GS3 protein to natural variation of grain size in rice. Proc Natl Acad Sci USA 107:19579–19584
Murray M, Thompson WF (1980) Rapid isolation of high molecular weight plant DNA. Nucleic Acids Res 8:4321–4326
Okamoto Y, Nguyen AT, Yoshioka M, Iehisa JC, Takumi S (2013) Identification of quantitative trait loci controlling grain size and shape in the D genome of synthetic hexaploid wheat lines. Breed Sci 63: 423–429
Pearce S, Vazquez-Gross H, Herin SY, Hane D, Wang Y, Gu YQ, Dubcovsky J (2015) WheatExp: an RNA-seq expression database for polyploid wheat. BMC Plant Biol 15:1
Postel D, Vanlemmens P, Gode P, Ronco G, Villa P (2002) PlantCARE, a database of plant cis-acting regulatory elements and a portal to tools for in silico analysis of promoter sequences. Nucleic Acids Res 30:325–327
Qi P, Lin YS, Song XJ, Shen JB, Huang W, Shan JX, Zhu MZ, Jiang L, Gao JP, Lin HX (2012) The novel quantitative trait locus GL3.1 controls rice grain size and yield by regulating Cyclin-T1;3. Cell Res 22:1666–1680
Reddy AS (2007) Alternative splicing of pre-messenger RNAs in plants in the genomic era. Annu Rev Plant Biol 58:267–294
Reynolds M, Foulkes J, Furbank R, Griffiths S, King J, Murchie E, Parry M, Slafer G (2012) Achieving yield gains in wheat. Plant Cell Environ 35:1799–1823
Rushton PJ, Torres JT, Parniske M, Wernert P, Hahlbrock K, Somssich I (1996) Interaction of elicitor-induced DNA-binding proteins with elicitor response elements in the promoters of parsley PR1 genes. EMBO J 15:5690–5700
Simmonds J, Scott P, Brinton J, Mestre TC, Bush M, Blanco AD, Dubcovsky J, Uauy C (2016) A splice acceptor site mutation in TaGW2-A1 increases thousand grain weight in tetraploid and hexaploid wheat through wider and longer grains. Theor Appl Genet 129:1–14
Song XJ, Huang W, Shi M, Zhu MZ, Lin HX (2007) A QTL for rice grain width and weight encodes a previously unknown RING-type E3 ubiquitin ligase. Nat Genet 39:623–630
Su Z, Hao C, Wang L, Dong Y, Zhang X (2011) Identification and development of a functional marker of TaGW2 associated with grain weight in bread wheat (Triticum aestivum L.). Theor Appl Genet 122:211–223
Su Z, Jin S, Lu Y, Zhang G, Chao S, Bai G (2016) Single nucleotide polymorphism tightly linked to a major QTL on chromosome 7 A for both kernel length and kernel weight in wheat. Mol Breed 36:1–11
Sun X-Y, Wu K, Zhao Y, Kong F-M, Han G-Z, Jiang H-M, Huang X-J, Li R-J, Wang H-G, Li S-S (2009) QTL analysis of kernel shape and weight using recombinant inbred lines in wheat. Euphytica 165:615–624
Thompson JD, Higgins DG, Gibson TJ (1994) CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res 22:4673–4680
Vogel JP, Garvin DF, Mockler TC, Schmutz J, Rokhsar D, Bevan MW, Barry K, Lucas S, Harmon-Smith M, Lail K (2010) Genome sequencing and analysis of the model grass Brachypodium distachyon. Nature 463:763–768
Wang S, Wu K, Yuan Q, Liu X, Liu Z, Lin X, Zeng R, Zhu H, Dong G, Qian Q (2012) Control of grain size, shape and quality by OsSPL16 in rice. Nat Genet 44:950–954
Wang S, Li S, Liu Q, Wu K, Zhang J, Wang S, Wang Y, Chen X, Zhang Y, Gao C (2015a) The OsSPL16-GW7 regulatory module determines grain shape and simultaneously improves rice yield and grain quality. Nat Genet 47:949–954
Wang Y, Xiong G, Hu J, Jiang L, Yu H, Xu J, Fang Y, Zeng L, Xu E, Xu J (2015b) Copy number variation at the GL7 locus contributes to grain size diversity in rice. Nat Genet 47:944–948
Weng JF, Gu SH, Wan XY, Gao H, Guo T, Su N, Lei CL, Zhang X, Cheng ZJ, Guo XP, Wang JL, Jiang L, Zhai HQ, Wan JM (2008) Isolation and initial characterization of GW5, a major QTL associated with rice grain width and weight. Cell Res 18:1199–1209
Wu Q-H, Chen Y-X, Zhou S-H, Fu L, Chen J-J, Xiao Y, Zhang D, Ouyang S-H, Zhao X-J, Cui Y (2015) High-density genetic linkage map construction and QTL mapping of grain shape and size in the wheat population Yanda1817 × Beinong6. PloS ONE 10:e0118144
Yang Z, Bai Z, Li X, Wang P, Wu Q, Yang L, Li L, Li X (2012) SNP identification and allelic-specific PCR markers development for TaGW2, a gene linked to wheat kernel weight. Theor Appl Genet 125:1057–1068
Zhang X, Wang J, Huang J, Lan H, Wang C, Yin C, Wu Y, Tang H, Qian Q, Li J (2012) Rare allele of OsPPKL1 associated with grain length causes extra-large grain and a significant yield increase in rice. Proc Natl Acad Sci USA 109:21534–21539
Zhang X, Chen J, Shi C, Chen J, Zheng F, Tian J (2013) Function of TaGW2-6 A and its effect on grain weight in wheat (Triticum aestivum L.). Euphytica 192:347–357
Acknowledgements
This publication is based upon work supported by the projects from the National Natural Science Foundation of China (31570335 and 31601292) and the National Key Research and Development Program of China (2016YFD0101000, 2016YFD0102000, and 2016YFD0100100). We thank Dr. Cristobal Uauy and Dr. Philippa Borrill for providing the wheat transcriptome data. We appreciate the anonymous referees for critical reading of the manuscript.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
All authors read and agreed with the final manuscript and have no conflicts of interest.
Additional information
Jian Ma, Puyang Ding and Peng Qin have contributed equally to this paper.
Electronic supplementary material
Below is the link to the electronic supplementary material.
10725_2017_258_MOESM1_ESM.tif
Supplementary material 1 Fig. S1: Validation of expression patterns of GW7 in wheat by employment of another processed expression database The y-axis represents the expression value. For (a) Zadoks scale for wheat growth stage was adopted and detailed information for each stage is provided in Table S2. (TIF 2896 KB)
Rights and permissions
About this article
Cite this article
Ma, J., Ding, P., Qin, P. et al. Structure and expression of the TaGW7 in bread wheat (Triticum aestivum L.). Plant Growth Regul 82, 281–291 (2017). https://doi.org/10.1007/s10725-017-0258-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10725-017-0258-3