Abstract
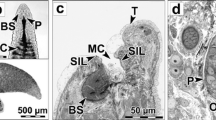
The tub gurnard Chelidonichthys lucerna (Linnaeus, 1758), Triglidae, is an opportunistic, demersal carnivorous fish. Data on the digestive enzymes of tub gurnard have not been reported in the literature. Therefore, the aim of this research was to investigate the distribution and intensity of alkaline phosphatase, acid phosphatase, non-specific esterase, and aminopeptidase in the digestive tract of tub gurnard. To investigate data about those enzymes tissue samples of the esophagus, anterior and posterior part of the stomach, pyloric caeca, anterior, middle and posterior part of the intestine proper, and rectum were taken. Azo-coupling methods were used to detect the enzymatic reactions. The intensities of the reactions were measured using ImageJ software. Alkaline phosphatase, acid phosphatase, and non-specific esterase activities were found in all parts of the digestive tract. The brush border of the pyloric caeca and intestine proper were the main sites of alkaline phosphatase reaction, with intensity decreasing toward the posterior parts of the digestive tract. The high intensities of acid phosphatase were found in the epithelium of the anterior part of the stomach, pyloric caeca, anterior part of the intestine proper, and in the rectum. The intensity of non-specific esterase was mainly increased from the anterior to the posterior parts of the digestive tract. Aminopeptidase activity was found in the esophagus, pyloric caeca, and intestine proper. Our results suggest that the entire digestive tract of the tub gurnard is involved in the digestion and absorption of dietary components.




Similar content being viewed by others
Data availability
The data supporting the results of this study are available from the corresponding author upon request.
References
Bakke AM, Glavar C, Krogdahl Å (2011) Feeding, digestion and absorption of nutrients. In: Grosell M, Farrell AF, Brauner CJ (eds) The multifunctional gut of fish, 1st edn. Elsevier, Amsterdam, Boston, Heidelberg, London, Oxford, New York, Paris, San Diego, San Francisco, Singapore, Sydney, Tokyo, pp 57–110
Boudaya L, Neifar L, Rizzo P, Badalucco C, Bouain A, Fiorentino F (2008) Growth and reproduction of Chelidonichthys lucerna (Linnaeus) (Pisces: Triglidae) in the Gulf of Gabès, Tunisia. J Appl Ichthyol 24(5):581–588. https://doi.org/10.1111/j.1439-0426.2008.01095.x
Chakrabarti I, Gani M, Chaki KK, Sur R, Misra KK (1995) Digestive enzymes in 11 freshwater teleost fish species in relation to food habit and niche segregation. Comp Biochem Physiol 112A:167–177. https://doi.org/10.1016/0300-9629(95)00072-F
Deimling OV, Böcking A (1976) Esterases in histochemistry and ultrahistochemistry. Histochem J 8:215–252. https://doi.org/10.1007/BF01003814
Dulčić J, Grubišić L, Katavić I, Skakelja N (2001) Embryonic and larval development of the tub gurnard Trigla lucerna (Pisces : Triglidae). J Mar Biol Assoc UK 81:313–316. https://doi.org/10.1017/S0025315401003794
Faccioli CK, Chedid RA, Mori RH, Amaral AC, Franceschini-Vicentini IB, Vicentini CA (2016) Acid and alkaline phosphatase localization in the digestive tract mucosa of the Hemisorubim platyrhynchos. Acta Histochem 118:722–728. https://doi.org/10.1016/j.acthis.2016.08.001
Ferreira I, Santos D, Moreira C, Feijó D, Rocha A, Correia AT (2019) Population structure of Chelidonichthys lucerna in Portugal mainland using otolith shape and elemental signatures. Mar Biol Res 15:500–512. https://doi.org/10.1080/17451000.2019.1673897
German DP, Gawlicka AK, Horn MH (2014) Evolution of ontogenetic dietary shifts and associated gut features in prickleback fishes (Teleostei: Stichaeidae). Comp Biochem Phys B 168:12–18. https://doi.org/10.1016/j.cbpb.2013.11.006
Gisbert E, Morais S, Moyano FJ (2013) Feeding and digestion. In: Qin JG (ed) Larval fish aquaculture. Nova Publishers, New York, pp 73–124
Hani YMI, Marchand A, Turies C, Kerambrun E, Palluel O, Bado-Nilles A, Beaudouin R, Porcher J, Geffard A, Dedourge-Geffard O (2018) Digestive enzymes and gut morphometric parameters of threespine stickleback (Gasterosteus aculeatus): influence of body size and temperature. Plos One 13(4):e0194932. https://doi.org/10.1371/journal.pone.0194932
Hidalgo MC, Urea E, Sanz A (1999) Comparative study of digestive enzymes in fish with different nutritional habits. Proteolytic and amylase activities. Aquaculture 170:267–283. https://doi.org/10.1016/S0044-8486(98)00413-X
Hopwood D, Logan KR, Milne G (1978) The light and electron microscopic distribution of acid phosphatase activity in human normal oesophageal epithelium. Histochem J 10:159–170. https://doi.org/10.1007/BF01003301
Ismen A, Ismen P, Basusta N (2004) Age, growth and reproduction of tub gurnard (Chelidonichthys lucerna L. 1758) in the Bay of Iskenderun in the eastern Mediterranean. Turk J Vet Anim Sci 28:289–295
Kiernan JA (2015) Histological and Histochemical Methods, 5th edn. Scion Publishing Limited, United Kingdom, pp 342–406
Kozarić Z, Kužir S, Nejedli S, Petrinec Z, Srebočan E (2004) Histochemical distribution of digestive enzymes hake, Merluccius merluccius L. 1758. Vet Arhiv 74:299–308
Kozarić Z, Petrinec Z, Kužir S, Gjurčević E, Baždarić B (2011) Histochemical analyses of digestive enzymes in the intestine of adult large-scaled gurnard (Lepidotrigla cavillone, Lacepède, 1801). Anat Histo Embryol 40:314–320. https://doi.org/10.1111/j.1439-0264.2011.01074.x
Kužir S, Gjurčević E, Nejedli S, Baždarić B, Kozarić Z (2012) Morphological and histochemical study of intestine in wild and reared European eel (Anguilla anguilla L.). Fish Physiol Biochem 38:625–633. https://doi.org/10.1007/s10695-011-9543-7
Kuz’mina VV (1996) Influence of age on digestive enzyme activity in some freshwater teleosts. Aquaculture 148:25–37. https://doi.org/10.1016/S0044-8486(96)01370-1
Lallès JP (2019) Intestinal alkaline phosphatase in the gastrointestinal tract of fish: biology, ontogeny, and environmental and nutritional modulation. Rev Aquacult 12:555–581. https://doi.org/10.1111/raq.12340
Loyda Z, Gossrau R, Schiebler TH (1979) Enzyme histochemistry. In: A laboratory manual. Springer-Verlag, Berlin, Heidelberg, New York. https://doi.org/10.1007/978-3-642-67234-7
Lucas A (2002) Cellular Bioenergetics. In: Priede IG (ed) Bioenergetics of Aquatic Animals. Taylor & Francis, London, Bristol, pp 21–40. https://doi.org/10.1201/9781482295313
McCarthy ID, Marriott AL (2018) Age, growth and maturity of tub gurnard (Chelidonichthys lucerna Linnaeus 1758; Triglidae) in the inshore coastal waters of Northwest Wales, UK. J Appl Ichthyol 34:581–589. https://doi.org/10.1111/jai.13614
Montanini S, Stagioni M, Benni E, Vallisneri M (2017a) Feeding strategy and ontogenetic changes in diet of gurnards (Teleostea: Scorpaeniformes: Triglidae) from the Adriatic Sea. The European Zoological J 84:356–367. https://doi.org/10.1080/24750263.2017.13353
Montanini S, Stagioni M, Benni E, Vallisneri M (2017b) Ontogenetic changes in otolith morphology and shape analyses in Chelidonichthys cuculus (Linnaeus, 1758) and Chelidonichthys lucerna (L., 1758). J Appl Ichthyol 33:217–220. https://doi.org/10.1111/jai.13299
Ostaszewska T, Kamaszewski M (2019) Digestive system. In: Kirschbaum F, Formicki K (eds) The Histology of Fishes. CRC Press, Boca Raton, London, New York, pp 88–100
Pearse AGE (1968) Histochemistry. Theoretical and Applied. J. & A. Churchill Ltd., London
Roncarati A, D'Andrea M, Pilla F, Felici A, Melotti P (2013) Tub gurnard Chelidonichthys lucerna L.: a new fish species suitable for farming? First answers evaluating the growth of juveniles reared at different stocking densities, welfare and fillet quality. Aquac Res 44:1140–1151. https://doi.org/10.1111/j.1365-2109.2012.03238.x
Roncarati A, Felici A, Mariotti F, Melotti P (2014) Flesh qualitative traits of tub gurnard (Chelidonichthys lucerna L.), a promising species candidate for aquaculture, captured in the middle Adriatic Sea in different seasons. Ital J Anim Sci 13:352–356. https://doi.org/10.4081/ijas.2014.3159
Roncarati A, Mariotti F, Felici A, Meligrana M, Melotti P (2016) Suitability of artisanal fishery discards as feed for juvenile tub gurnard (Chelidonichthys lucerna L.) reared in sea bottom cages in the mid Adriatic Sea. Mediterr Mar Sci 17:644–650. https://doi.org/10.12681/mms.1750
Sheehan DC, Hrapchak BB (1980) Enzyme histochemistry. In: Theory and practice of Histotechnology, 2nd edn. Battelle Press, Columbus, Ohio, pp 292–310
Solovyev M, Gisbert E (2016) Influence of time, storage temperature and freeze/thaw cycles on the activity of digestive enzymes from gilthead sea bream (Sparus aurata). Fish Physiol Biochem 42:1383–1394. https://doi.org/10.1007/s10695-016-0226-2
Stagioni M, Montanini S, Vallisneri M (2011) Feeding of tub gurnard Chelidonichthys lucerna (Scorpaeniformes: Triglidae) in the north-east Mediterranean. J Mar Biol Assoc UK 92:605–612. https://doi.org/10.1017/S0025315411000671
Suvarna SK, Layton C, Bancroft JD (2019) Bancroft’s theory and practice of histological techniques. Elsevier, Scotland, pp 40–95
Tlak Gajger I, Nejedli S, Kozarić Z (2012) Histochemical distribution of digestive enzymes in the intestine of the common two-banded seabream, Diplodus vulgaris, Geoffroy St-Hilaire 1817. Anat Histol Embryol 42:161–167. https://doi.org/10.1111/j.1439-0264.2012.01179.x
Ugolev AM (1965) Membrane (Contact) Digestion. Physiol Rev 45:555–587. https://doi.org/10.1152/physrev.1965.45.3.555
Uyan A, Turan C (2017) Genetic and morphological analyses of tub gurnard Chelidonichthys lucerna populations in Turkish marine waters. Biochem Syst Ecol 73:35–40. https://doi.org/10.1016/j.bse.2017.06.003
Vallisneri M, Stagioni M, Montanini S, Tommasini S (2011) Body size, sexual maturity and diet in Chelidonichthys lucerna (Osteichthyes: Triglidae) from the Adriatic Sea, north eastern Mediterranean. Acta Adriat 52:141–148. https://doi.org/10.32582/aa.v52i1.297
Wang YZ, Sun JF, Lv AJ, Zhang SL, Sung YY, Shi HY, Hu XC, Chen SJ, Xing KZ (2018) Histochemical distribution of four types of enzymes and mucous cells in the gastrointestinal tract of reared half-smooth tongue sole Cynoglossus semilaevis. J Fish Biol 92:3–16. https://doi.org/10.1111/jfb.13469
Watanabe Y (1984) An ultrastructural study of intracellular digestion of horseradish peroxidase by the rectal epithelium cells in larvae of a freshwater cottid fish Cottus nozawae. B Jpn Soc Sci Fish 50:409–416. https://doi.org/10.1111/j.1095-8649.1991.tb03086.x
Acknowledgements
The authors would like to thank Nada Crnogaj, Department of Anatomy, Histology and Embryology, for her technical assistance.
Funding
This research was carried out as a part of scientific project “Improvement of cooperation between fishermen and scientists for the purpose of introducing advanced technologies of marking of fishing tools, fish health and environmental protection” supported by the Operational Programme for Maritime Affairs and Fisheries of the Republic of Croatia for the programming period 2014-2020. The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
Lucija Bastiančić, Snježana Kužir, and Damir Valić contributed to the conception and design of the study. Marin Lovrić and Damir Valić organized the sampling of the fish. All authors were equally involved in the selection of methodology and supervision. Lucija Bastiančić and Snježana Kužir prepared the material, collected data, and analyzed the samples. Ivan Vlahek performed the statistical analysis of the study data. Lucija Bastiančić and Valerija Benko were responsible for imaging and image analyses. The first draft of the manuscript was written by Lucija Bastiančić and revised by all authors. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
The research is performed in accordance with Animal Protection Act by the Ministry of Agriculture and approved by The Committee for Ethics in Veterinary Medicine, Faculty of Veterinary Medicine (No. 251-61-01/139-20-32).
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Bastiančić, L., Vlahek, I., Benko, V. et al. Histochemical research of enzymes involved in cellular digestion in the digestive tract of tub gurnard, Chelidonichthys lucerna. Fish Physiol Biochem 50, 157–170 (2024). https://doi.org/10.1007/s10695-023-01188-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-023-01188-3