Abstract
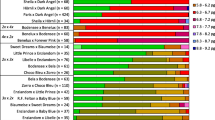
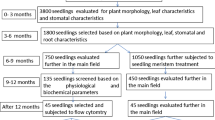
The relationship between the ploidy level and phenotype in haskap (Lonicera caerulea L. subsp. edulis (Turcz. ex Herder) Hultén) is mostly unclear. Therefore, this study investigated the fruit and leaf phenotypes of diploid, tetraploid, pentaploid, hexaploid, and aneuploid (2n = 6x − 3) haskap plants. A concomitant positive increase in the ploidy level was observed between diploids and tetraploids in fresh fruit weight and size, but this trend was not observed in higher-order ploidy levels above pentaploids. The same tendency was observed in the soluble solids content of the fruit. No relationship was found among the other traits with ploidy levels. Therefore, in the phenotype of haskap plants, the trend accompanying the change in ploidy level may differ between individuals below tetraploid and above pentaploid, and the influence of genotype is probably greater than that of ploidy levels. Generally, higher-order polyploid plants have low fertility, especially plants with odd ploidy, and the production of progeny is difficult. In this study, seeds of higher-order ploidy haskap plants demonstrated their ability to germinate and grow. Furthermore, the progeny of these hyperploid lineages showed polyploidy or aneuploidy between tetraploidy and hexaploidy. This study focuses on the higher-order polyploid haskap plants and discusses the genetic findings, such as the phenotype, fertility, and ploidy of the progeny, obtained from these plants. This study will provide basic information on breeding by using polyploid plants.





Similar content being viewed by others
Abbreviations
- LMA:
-
Leaf mass per area
- QTL:
-
Quantitative trait loci
- SPAD:
-
Soil plant analysis development
- SSC:
-
Soluble solids content
References
Anikina EV, Syrchina AI, Vereshchagin AL, Larin MF, Semenov AA (1988) Bitter iridoid glucoside from the fruit of Lonicera caerulea L. Chem Nat Compd 24:512–513. https://doi.org/10.1007/BF00598552
Bors B, Thomson J, Sawchuk E, Reimer P, Sawatzky R, Sander T (2012) Haskap breeding and production–final report. Saskatchewan Agric Regina 1–142
Chen ZJ (2007) Genetic and epigenetic mechanisms for gene expression and phenotypic variation in plant polyploids. Annu Rev Plant Biol 58:377–406. https://doi.org/10.1146/annurev.arplant.58.032806.103835
Cheng CH, Seal AG, Boldingh HL, Marsh KB, MacRae EA, Murphy SJ, Ferguson AR (2004) Inheritance of taste characters and fruit size and number in a diploid Actinidia chinensis (kiwifruit) population. Euphytica 138:185–195. https://doi.org/10.1023/B:EUPH.0000046802.28347.41
Comai L (2005) The advantages and disadvantages of being polyploid. Nat Rev Genet 6:836–846. https://doi.org/10.1038/nrg1711
De Storme N, Mason A (2014) Plant speciation through chromosome instability and ploidy change: cellular mechanisms, molecular factors and evolutionary relevance. Curr Plant Biol 1:10–33. https://doi.org/10.1016/j.cpb.2014.09.002
Fox DT, Soltis DE, Soltis PS, Ashman TL, Van de Peer Y (2020) Polyploidy: a biological force from cells to ecosystems. Trends Cell Biol 30:688–694. https://doi.org/10.1016/j.tcb.2020.06.006
Griffin DK (1996) The incidence, origin, and etiology of aneuploidy. Int Rev Cytol 167:263–296. https://doi.org/10.1016/S0074-7696(08)61349-2
Hirano T, Hoshino Y (2010) Sperm dimorphism in terms of nuclear shape and microtubule accumulation in Cyrtanthus mackenii. Sex Plant Reprod 23:153–162. https://doi.org/10.1007/s00497-009-0123-2
Ji X, Lelivelt C, Wijnker E, de Jong H (2021) Meiotic Aberrations leading to aneuploidy in cauliflower (Brassica oleracea L. var. botrytis). In: Ji X, Numerical and structural chromosome aberrations in cauliflower (Brassica oleracea var. botrytis) and Arabidopsis thaliana. PhD Thesis, Wageningen University, Wageningen. ISBN 978-94-6257-160-0. https://doi.org/10.21203/rs.3.rs-1067592/v1
Lattier JD, Contreras RN (2022) Flower forms and ploidy levels impact fertility in althea. HortScience 57:558–570. https://doi.org/10.21273/HORTSCI16478-21
Li D, Zhong C, Liu Y, Huang H (2010) Correlation between ploidy level and fruit characters of the main kiwifruit cultivars in China: implication for selection and improvement. N Z J Crop Hortic Sci 38:137–145. https://doi.org/10.1080/01140671.2010.482966
Miyashita T, Hoshino Y (2010) Interspecific hybridization in Lonicera caerulea and Lonicera gracilipes: the occurrence of green/albino plants by reciprocal crossing. Sci Hortic 125:692–699. https://doi.org/10.1016/j.scienta.2010.05.032
Miyashita T, Hoshino Y (2015) Interploid and intraploid hybridizations to produce polyploid Haskap (Lonicera caerulea var. emphyllocalyx) plants. Euphytica 201:15–27. https://doi.org/10.1007/s10681-014-1159-4
Miyashita T, Araki H, Hoshino Y (2011) Ploidy distribution and DNA content variations of Lonicera caerulea (Caplifoliaceae) in Japan. J Plant Res 124:1–9. https://doi.org/10.1007/s10265-010-0341-6
Murashige T, Skoog F (1962) A revised medium for the rapid growth and bioassay with tobacco tissue cultures. Physiol Plant 15:473–497
Nakato N, Masuyama S (2021) Polyploid progeny from triploid hybrids of Phegopteris decursivepinnata (Thelypteridaceae). J Plant Res 134:195–208. https://doi.org/10.1007/s10265-021-01255-x
Orr-Weaver TL (2015) When bigger is better: the role of polyploidy in organogenesis. Trends Genet 31:307–315. https://doi.org/10.1016/j.tig.2015.03.011
Peskoller A, Silbernagl L, Hülber K, Sonnleitner M, Schönswetter P (2021) Do pentaploid hybrids mediate gene flow between tetraploid Senecio disjunctus and hexaploid S. carniolicus s. Str. (S. carniolicus aggregate, Asteraceae)? Alp Botany 131:151–160. https://doi.org/10.1007/s00035-021-00254-x
Plekhanova MN (2000) Blue honeysuckle (Lonicera caerulea L.)-A new commercial berry crop for temperate climate: genetic resources and breeding. Acta Hortic 538:159–164. https://doi.org/10.17660/ActaHortic.2000.538.25
R Core Team (2020) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria
Ramsey J, Schemske DW (1998) Pathways, mechanisms, and rates of polyploid formation in flowering plants. Annu Rev Ecol Syst 29(1):467–501
Rupasinghe HPV, Yu LJ, Bhullar KS, Bors B (2012) Haskap (Lonicera caerulea): a new berry crop with high antioxidant capacity. Can J Plant Sci 92:1311–1317. https://doi.org/10.4141/cjps2012-073
Santos GC, Cardoso FP, Martins AD, Rodrigues FA, Pasqual M, Cossani P, Magno Queiroz Luz JMQ, Rezende RALS, Soares JDR (2020) Polyploidy induction in Physalis alkekengi. Biosci J 36:827–835. https://doi.org/10.14393/BJ-v36n3a2020-47729
Siegel JJ, Amon A (2012) New insights into the troubles of aneuploidy. Annu Rev Cell Dev Biol 28:189–214. https://doi.org/10.1146/annurev-cellbio-101011-155807
Soltis PS, Soltis DE (2000) The role of genetic and genomic attributes in the success of polyploids. Proc Natil Acad Sci USA 97:7051–7057. https://doi.org/10.1073/pnas.97.13.7051
Svarcova I, Heinrich J, Bednar P, Kren V, Cvak L, Ulrichova J, Simanek V, Valentova K (2007) Main components and radical scavenging activity of Lonicera caerulea L. var. kamtschatica Berries. Fifty Years of the Phytochemical Society of Europe, Cambridge, pp 94–95
Thompson MM, Barney DL (2007) Evaluation and breeding of haskap in North America. J Am Pomol Soc 61:25–33
Tsukaya H (2013) Does ploidy level directly control cell size? Counterevidence from arabidopsis genetics. PLoS ONE 8:e83729. https://doi.org/10.1371/journal.pone.0083729
Xiao K, Zhu Z, Zou N, Zhang L, Sun Y, Zhou S (2022) The characteristics of abnormal meiosis and functional aneuploid pollen of odd-allotetraploid lily ‘Honesty’ unveiled using in situ hybridization. Sci Hortic 300:111091. https://doi.org/10.1016/j.scienta.2022.111091
Zhang H, Bian Y, Gou X, Zhu B, Xu C, Qi B, Li N, Rustgi S, Zhou H, Han F, Jiang J, von Wettstein D, Liu B (2013) Persistent whole-chromosome aneuploidy is generally associated with nascent allohexaploid wheat. Proc Natl Acad Sci USA 110:3447–3452. https://doi.org/10.1073/pnas.1300153110
Zhong J, Cai J, Liu S, Wang Z, Yin D, Zhou S (2022) GISH analysis of introgressive hybridization using aneuploids as male parents in Lilium. Euphytica 218:1–9. https://doi.org/10.1007/s10681-022-03116-7
Acknowledgements
The authors thank Emeritus Professor H. Nakashima and Mr. H. Nakano for their support in plant management. This work was partially supported by the Japan Society for the Promotion of Science (JSPS) KAKENHI Grant Number 20K06028 and 16K07584. Jixiao Li received the grant from JST SPRING, Grant Number JPMJSP2119, to proceed with this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, J., Suzuki, Y. & Hoshino, Y. Phenotypic analysis of polyploid and aneuploid haskap (Lonicera caerulea L. subsp. edulis (Turcz. ex Herder) Hultén) plants and their progeny production. Euphytica 219, 52 (2023). https://doi.org/10.1007/s10681-023-03181-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10681-023-03181-6