Abstract
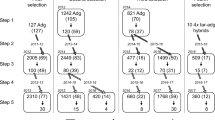
Most wild and cultivated diploid potato species are self-incompatible, preventing inbreeding and promoting outcrossing. To generate diploid inbred lines, the tuber bearing self-compatible species Solanum chacoense (M6 line) has been used to overcome self-incompatibility. In this study, self-compatibility was introgressed from a set of four diploid self-compatible (SC) donor lines with S. chacoense background into eight introgression species lines to develop a multi-species potato germplasm pool using a recurrent selection (RS) breeding strategy. Concurrently, tuber and adaptation-related traits were selected under Michigan field conditions prior to self-compatibility assessment. After six cycles of RS, we observed a significant increase in self-compatibility (16% in cycle 0–86% in cycle 5) inheritance and improvement of tuber-related traits. Reproductive fitness was increased, reducing male sterility from 32% in cycle 0 to 0% in cycle 5. Progeny with no-flowers decreased from 19 to 3% from RS cycles 0 to cycle 5. Additionally, 4,885 single-nucleotide polymorphism (SNPs) markers were used to access the linkage disequilibrium (LD), heterozygosity, and population structure over the RS population. SNP heterozygosity moderately decreased through the six cycles of RS cycles. Hierarchical clustering and Principal Components Analysis showed that selections become more uniform as the cycles advanced and that LD had been reduced across each potato chromosome. We observed significant increase in earliness for vine maturity in cycle 4. The SC germplasm developed in this study can be used to introgress self-compatibility, select tuber-related traits, and develop inbred lines in F1 hybrid programs using a genetically diverse diploid germplasm.






Similar content being viewed by others
References
Alsahlany M, Zarka D, Coombs J, Douches DS (2019) Comparison of methods to distinguish diploid and tetraploid potato in applied diploid breeding. Am J Potato Res 96:244–254. https://doi.org/10.1007/s12230-018-09710-7
Benites FRG, Pinto CABP (2011) Genetic gains for heat tolerance in potato in three cycles of recurrent selection. Crop Breed Appl Biotechnol 11:133–140. https://doi.org/10.1590/S1984-70332011000200005
Berdugo-Cely J, Valbuena RI, Sánchez-Betancourt E et al (2017) Genetic diversity and association mapping in the colombian central collection of Solanum tuberosum L. PLoS One, Andigenum group using SNPs markers. https://doi.org/10.1371/journal.pone.0173039
Bonierbale MW, Plaisted RL, Tanksley SD (1993) A test of the maximum heterozygosity hypothesis using molecular markers in tetraploid potatoes. Theor Appl Genet 86:481–491
Bradshaw JE, Bryan GJ, Ramsay G (2006) Genetic resources (including wild and cultivated Solanum species) and progress in their utilisation in potato breeding. Potato Res 49:49–65. https://doi.org/10.1007/s11540-006-9002-5
Bradshaw JE, Finlay M, Dale B, Mackay GR (2009) Improving the yield, processing quality and disease and pest resistance of potatoes by genotypic recurrent selection. Euphytica 170:215–227. https://doi.org/10.1007/s10681-009-9925-4
Braun SR, Endelman JB, Haynes KG, Jansky SH (2017) Quantitative trait loci for resistance to common scab and cold-induced sweetening in diploid potato. Plant Genome 10:1–9. https://doi.org/10.3835/plantgenome2016.10.0110
Byers DL, Meagher TR (1992) Mate availability in small populations of plant species with homomorphic sporophytic self-incompatibility. Heredity (Edinb) 68:353–359. https://doi.org/10.1038/hdy.1992.50
Cubillos AG, Plaisted RL (1976) Heterosis for yield in hybrids betweenS. Tuberosum ssp.Tuberosum andtuberosum ssp. Andigena Am J Potato Res 53:143–150
De Haan S, Forbes A, Amoros W, et al (2014) Procedures for Standard Evaluation and Data Management of Advanced Potato Clones. Module 2: Healthy Tuber Yield Trials.: International Cooperators Guide. International Potato Center.
De Koeyer DL, Phillips RL, Stuthman DD (1999) Changes in genetic diversity during seven cycles of recurrent selection for grain yield in oat, Avena sativa L. Plant Breed 118:37–43
Domański L (2001) Assessment of morphological characters of potato tubers. Monografie i Rozprawy Naukowe IHAR, Radzików. Poland 10a, 92–95. 92–95.
Driscoll J, Coombs J, Hammerschmidt R et al (2009) Greenhouse and field nursery evaluation for potato common scab tolerance in a tetraploid population. Am J Potato Res 86:96–101. https://doi.org/10.1007/s12230-008-9065-8
EncisoRodriguez F, Douches D, LopezCruz M et al (2018) Genomic selection for late blight and common scab resistance in Tetraploid Potato (Solanum tuberosum). G3 Genes|Genomes|Genetics 8:2471–2481. https://doi.org/10.1534/g3.118.200273
Entani T, Iwano M, Shiba H et al (2003) Comparative analysis of the self-incompatibility (S-) locus region of Prunus mume: Identification of a pollen-expressed F-box gene with allelic diversity. Genes Cells 8:203–213. https://doi.org/10.1046/j.1365-2443.2003.00626.x
Fujii S, Kubo KI, Takayama S (2016) Non-self- and self-recognition models in plant self-incompatibility. Nat Plants 2(9):16130. https://doi.org/10.1038/nplants.2016.130
Gebhardt C, Ballvora A, Walkemeier B et al (2004) Assessing genetic potential in germplasm collections of crop plants by marker-trait association: a case study for potatoes with quantitative variation of resistance to late blight and maturity type. Mol Breed 13:93–102
Goldraij A, Kondo K, Lee CB, Hancock CN, Sivaguru M, Vazquez-Santana S, McClure B (2006) Compartmentalization of S-RNase and HT-B degradation in self-incompatible Nicotiana. Nature 439:805
Hawkes JG (1958) Significance of wild species and primitive forms for potato breeding. Euphytica 7:257–270. https://doi.org/10.1007/BF00025267
Haynes KG (2001) Variance components for yield and specific gravity in a diploid potato population after two cycles of recurrent selection. Am J Potato Res 78:69–75. https://doi.org/10.1007/BF02874827
Haynes KG, Qu X (2018) Three Cycles of Recurrent Maternal Half-Sib Selection Continue to Reduce Foliar Late Blight in a Diploid Hybrid Solanum phureja-S. stenotomum Population. Am J Potato Res. doi: https://doi.org/10.1007/s12230-013-9345-9
Hermsen JGT (1989) Current use of potato collections. In: Proc. IBPGR Workshop The use of plant genetic resources, A.H.D. Brown, O.H. Frankel, D.R. Marshall, J.T. Williams (eds.). Montpellier, France, 1986. Cambridge University Press
Hirsch CN, Hirsch CD, Felcher K et al (2013) Retrospective view of north american potato Solanum tuberosum L breeding in the 20th and 21st Centuries. G3 Genes|Genomes|Genetics 3:1003–1013. https://doi.org/10.1534/g3.113.005595
Hosaka K, Hanneman RE Jr (1998) Genetics of self-compatibility in a self-incompatible wild diploid potato species Solanum chacoense. 1. Detection of an S locus inhibitor (Sli) gene. Euphytica 99:191–197. https://doi.org/10.1023/a:1018353613431
Hull FH (1945) Recurrent Selection and Specific Combining Ability in Corn 1. Am Soc Agron 37:134–145
Jackson MT, Hawkes JG, Rowe PR (1980) An ethnobotanical field study of primitive potato varieties in Peru. Euphytica 29:107–113
Jansky S, Charkowski AO, Douches DS et al (2016) Reinventing potato as a diploid inbred line-based crop. Crop Sci 56:1412–1422. https://doi.org/10.2135/cropsci2015.12.0740
Jansky S, Chung YS, Kittipadukal P (2014) M6: A diploid potato inbred line for use in breeding and genetics research. J Plant Regist 8:195–199. https://doi.org/10.3198/jpr2013.05.0024crg
Jenkins MT (1940) The Segregation of Genes Affecting Yield of Grain in Maize. J Am Soc Agron 32:55–63
Jombart T, Kamvar ZN, Collins C, et al (2020) Package ‘ adegenet .’
Labate JA, Lamkey KR, Lee M, Woodman WL (1997) Molecular genetic diversity after reciprocal recurrent selection in BSSS and BSCB1 maize populations. 37:416–423.
Leisner CP, Hamilton JP, Crisovan E et al (2018) Genome sequence of M6, a diploid inbred clone of the high-glycoalkaloid-producing tuber-bearing potato species Solanum chacoense, reveals residual heterozygosity. Plant J 94:562–570. https://doi.org/10.1111/tpj.13857
Lindhout P, Meijer D, Schotte T et al (2011) Towards F 1 hybrid seed potato breeding. Potato Res 54:301–312. https://doi.org/10.1007/s11540-011-9196-z
Manrique-Carpintero NC, Coombs JJ, Cui Y et al (2015) Genetic map and QTL analysis of agronomic traits in a diploid potato population using single nucleotide polymorphism markers. Crop Sci 55:2566–2579. https://doi.org/10.2135/cropsci2014.10.0745
Marroni F, Pinosio S, Zaina G et al (2011) Nucleotide diversity and linkage disequilibrium in Populus nigra cinnamyl alcohol dehydrogenase (CAD4) gene. Tree Genet Genomes 7:1011–1023. https://doi.org/10.1007/s11295-011-0391-5
McClure B, Cruz-García F, Romero C (2011) Compatibility and incompatibility in S-RNase-based systems. Ann Bot 108:647–658. https://doi.org/10.1093/aob/mcr179
O’Brien M, Kapfer C, Major G, Laurin M, Bertrand C, Kondo K, Matton DP (2002) Molecular analysis of the stylar-expressed Solanum chacoense small asparagine-rich protein family related to the HT modifier of gametophytic self-incompatibility in Nicotiana. Plant J 32(6):985–996
Paradis E, Schliep K (2019) Ape 5.0: An environment for modern phylogenetics and evolutionary analyses in R. Bioinformatics 35:526–528. https://doi.org/10.1093/bioinformatics/bty633
Phumichai C, Mori M, Kobayashi A et al (2005) Toward the development of highly homozygous diploid potato lines using the self-compatibility controlling Sli gene. Genome 48:977–984. https://doi.org/10.1139/g05-066
Plaisted RL, Peterson LC (1963) Two cycles of phenotypic recurrent selection for high specific gravity. Am Potato J 40:396–402
Plaisted RL, Thurston HD, Sieczka JB et al (1981) Rosa: a new golden nematode resistant variety for chipping and tablestock. Am Potato J 58:451–455. https://doi.org/10.1007/BF02874542
Ríos RO (2015) Plant Breeding in the Omics Era. Springer
Ruiz de Galarreta JI, López-Pardo R, Tierno R et al (2015) Disease resistance and nutritional properties of tuber-bearing native potato species and old Spanish cultivars. J Agric Sci Technol 17:935–947
Sanetomo R, Akino S, Suzuki N, Hosaka K (2014) Breakdown of a hybridization barrier between Solanum pinnatisectum Dunal and potato using the S locus inhibitor gene (Sli). Euphytica 197:119–132. https://doi.org/10.1007/s10681-013-1057-1
Sanford LL, Ladd TL (1987) Genetic transmission of potato leafhopper resistance from recurrent selection populations in potato Solanum tuberosum L. Gp tuberosum Am Potato J 64:655–662
Sharma SK, MacKenzie K, McLean K et al (2018) Linkage Disequilibrium and Evaluation of Genome-Wide Association Mapping Models in Tetraploid Potato. G3 Genes|Genomes|Genetics 8:3185–3202. https://doi.org/10.1534/g3.118.200377
Shin JH, Blay S, McNeney B, Graham J (2006) LDheatmap: an R function for graphical display of pairwise linkage disequilibria between single nucleotide polymorphisms. J Stat Softw 16:1–10. https://doi.org/10.18637/jss.v000.i00
Spooner DM, Hijmans RJ (2001) Potato systematics and germplasm collecting, 1989–2000. Am J Potato Res 78:237–268. https://doi.org/10.1007/BF02875691
Spooner DM, Salas A (2006) Structure, iosystematics and genetic resources. Handb potato Prod Improv postharvest Manag New York, USA Haworth’s Press Inc Binghampt 1–39. Doi: https://doi.org/10.1300/5776
Villegas CT, Wilsie CP, Frey KJ (1971) Recurrent Selection for High Self-Fertility in Vernal Alfalfa (Medicago Sativa L.). Crop Sci 11:881–883
Vincent Q (2011) A ggplot2 based biplot. R Packag version 055. Available: http://github.com/vqv/ggbiplot
Visser RGF, Bachem CWB, de Boer JM et al (2009) Sequencing the potato genome: Outline and first results to come from the elucidation of the sequence of the world’s third most important food crop. Am J Potato Res 86:417–429. https://doi.org/10.1007/s12230-009-9097-8
Yuan M, Zhou Y, Liu D (2004) Genetic diversity among populations and breeding lines from recurrent selection in Brassica napus as revealed by RAPD markers. Plant Breed 123:9–12
Acknowledgement
The work was funded by USDA NIFA 2014-67013-22434. MA received fellowship from the Higher Committee for Education Development in Iraq (HCED). A special thanks to all Michigan State University Potato Breeding and Genetic Program team members and especially Nick Garrity, Donna Kells, Damen Kurzer, and Azamat Sardarbekov who helped with chloroplast counting and pollination. The authors also would like to thank Greg Steere and Matt Zuehlke for technical assistance. The authors thank Dr. Norma C. Manrique-Carpintero for analysis advice and Dr. Daniel Zarka and Andrea Garcia-Ramirez for SNP genotyping and technical assistance.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Online Resources 1
Comparison of hierarchical clustering between cycle 0 to cycle 5 with parental lines using 4,885 SNPs. (a) cycle 0, (b) cycle 1, (c) cycle 2, (d) cycle 3, (e) cycle 4, and (f) cycle 5. The black color is representing parental lines that used to generate the RS germplasm; blue is representing genotyped progeny from each cycle of RS (TIF 3483 KB)
Online Resources 2
Comparison of principal component analysis between cycle 0 to cycle 5 with parental lines using 4,885 SNPs. (a) cycle 0, (b) cycle 1, (c) cycle 2, (d) cycle 3, (e) cycle 4, and (f) cycle 5. The black color is representing parental lines that used to generate the RS germplasm; Blue is representing genotyped progeny from each cycle of RS (TIF 2042 KB)
Online Resources 3
Linkage disequilibrium (LD) measure r2 in the association panel plotted against the physical map distance (MB) between two pairs of SNPs located on the whole chromosomal region for all 12 chromosomes for cycle 0 of the RS in the upper panels. Nonlinear quantile regression of r2 versus the physical map distance between the SNP markers is showing in the red line. Lower panels: Genome-wide LD scans using 4,885 SNPs across the whole chromosomal for all 12 chromosomes in cycle 0 of the RS. Blue areas show regions with lower LD while the red areas show regions with higher LD (TIF 8028 KB)
Online Resources 4
Linkage disequilibrium (LD) measure r2 in the association panel plotted against the physical map distance (MB) between two pairs of SNPs located on the whole chromosomal region for all 12 chromosomes for cycle 5 of the RS in the upper panels. Nonlinear quantile regression of r2 versus the physical map distance between the SNP markers is showing in the red line. Lower panels: Genome-wide LD scans using 4,885 SNPs across the whole chromosomal for all 12 chromosomes in cycle 5 of the RS. Blue areas show regions with lower LD while the red areas show regions with higher LD. The figure is showing the LD reduction in all 12 chromosomes over six cycles of RS compared with Figure S1 (TIF 7720 KB)
Rights and permissions
About this article
Cite this article
Alsahlany, M., Enciso-Rodriguez, F., Lopez-Cruz, M. et al. Developing self-compatible diploid potato germplasm through recurrent selection. Euphytica 217, 47 (2021). https://doi.org/10.1007/s10681-021-02785-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10681-021-02785-0