Abstract
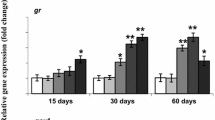
Glutathione S-transferases (GST) are considered among the most controversial biomarkers of water pollutants in fish with little known about factors influencing their activities. The objective of this study was to investigate how gender, dose, ploidy, and sampling time alter hepatic GST activities in African catfish (Clarias gariepinus) following β-naphthoflavone (β-NF) injection. Newly matured male and female diploid and triploid fish were intraperitoneally (i.p.) injected with 0, 15, or 75 mg/kg of β-NF, and livers were excised 24, 48, and 72 h post-injection. Results showed that hepatic GST activities were significantly inhibited by both doses of β-NF. Inhibition was greater in females than males, but no significant differences were observed between diploid and triploid fish. Enzymatic activities differed over time with lowest levels 72 h post-injection. These results extend our understanding of GST activity in fish and highlight the necessity of considering confounding factors when comparing different studies.






Similar content being viewed by others
References
Ali, M., & Jauncey, K. (2005). Approaches to optimizing dietary protein to energy ratio for African catfish Clarias gariepinus (Burchell, 1822). Aquaculture Nutrition, 11(2), 95–101.
Almeida, J. R., Gravato, C., & Guilhermino, L. (2014). Effects of temperature in juvenile seabass (Dicentrarchus labrax L.) biomarker responses and behaviour: implications for environmental monitoring. Estuaries and Coasts, 38, 45–55.
Atkins, M. E., & Benfey, T. J. (2008). Effect of acclimation temperature on routine metabolic rate in triploid salmonids. Comparative Biochemistry and Physiology - Part A: Molecular & Integrative Physiology, 149(2), 157–161.
Banni, M., Bouraoui, Z., Ghedira, J., Clerandeau, C., Guerbej, H., Narbonne, J., & Boussetta, H. (2009). Acute effects of benzo[a]pyrene on liver phase I and II enzymes, and DNA damage on sea bream Sparus aurata. Fish Physiology and Biochemistry, 35(2), 293–299.
Benfey, T. J. (1999). The physiology and behavior of triploid fishes. Reviews in Fisheries Science, 7(1), 39–67.
Benfey, T. J. (2001). Use of sterile triploid Atlantic salmon (Salmo salar L.) for aquaculture in New Brunswick, Canada. ICES Journal of Marine Science: Journal du Conseil, 58(2), 525–529.
Bradford, M. M. (1976). A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Analytical Biochemistry, 72(1-2), 248–254.
Carrera, E. P., García-López, A., Martín del Río, M. P., Martínez-Rodríguez, G., Solé, M., & Mancera, J. M. (2007). Effects of 17β-estradiol and 4-nonylphenol on osmoregulation and hepatic enzymes in gilthead sea bream (Sparus auratus). Comparative Biochemistry and Physiology Part C: Toxicology & Pharmacology, 145(2), 210–217.
Celander, M., Leaver, M. J., George, S. G., & Förlin, L. (1993). Induction of cytochrome P450 1A1 and conjugating enzymes in rainbow trout (Oncorhynchus mykiss) liver: a time course study. Comparative Biochemistry and Physiology Part C: Pharmacology, Toxicology and Endocrinology, 106(2), 343–349.
Celander, M. C. (2011). Cocktail effects on biomarker responses in fish. Aqutic Toxicology, 105(3), 72–77.
Clasen, B., Leitemperger, J., Murussi, C., Pretto, A., Menezes, C., Dalabona, F., Marchezan, E., Adaime, M. B., Zanella, R., & Loro, V. L. (2014). Carbofuran promotes biochemical changes in carp exposed to rice field and laboratory conditions. Ecotoxicology and Environmental Safety, 101, 77–82.
de Bittencourt, P. H., Curi, R., & Williams, J. F. (1998). Glutathione metabolism and glutathione S‐conjugate export ATPase (MRP1/GS‐X pump) activity in cancer. International Union of Biochemistry and Molecular Biology Life, 45(6), 1227–1241.
Di Bello, D., Vaccaro, E., Longo, V., Regoli, F., Nigro, M., Benedetti, M., Gervasi, P. G., & Pretti, C. (2007). Presence and inducibility by β-naphthoflavone of CYP1A1, CYP1B1 and phase II enzymes in Trematomus bernacchii, an Antarctic fish. Aqutic Toxicology, 84(1), 19–26.
Dixit, R., Riviere, J., Krishnan, K., & Andersen, M. (2003). Toxicokinetics and physiologically based toxicokinetics in toxicology and risk assessment. Journal of Toxicology and Environmental Health, Part B, 6(1), 1–40.
Ferreira, R. S., Monserrat, J. M., Ferreira, J. L. R., Kalb, A. C., Stegeman, J., Bainy, A. C. D., & Zanette, J. (2012). Biomarkers of organic contamination in the South American fish Poecilia vivipara and Jenynsia multidentata. Journal of Toxicology and Environmental Health, Part A, 75(16-17), 1023–1034.
Frasco, M. F., & Guilhermino, L. (2002). Effects of dimethoate and beta-naphthoflavone on selected biomarkers of Poecilia reticulata. Fish Physiology and Biochemistry, 26(2), 149–156.
Garcia, C., Moreira-Filho, O., Bertollo, L. A. C., & Centofante, L. (2003). B chromosomes and natural triploidy in Rhamdia sp. (Pisces, Siluriformes, Heptapteridae). Cytologia, 68(4), 403–411.
Genovese, G., Regueira, M., Piazza, Y., Towle, D. W., Maggese, M. C., & Lo Nostro, F. (2012). Time-course recovery of estrogen-responsive genes of a cichlid fish exposed to waterborne octylphenol. Aqutic Toxicology, 114, 1–13.
Greco, L., Serrano, R., Blanes, M. A., Serrano, E., & Capri, E. (2010). Bioaccumulation markers and biochemical responses in European sea bass (Dicentrarchus labrax) raised under different environmental conditions. Ecotoxicology and Environmental Safety, 73(1), 38–45.
Habig, W. H., Pabst, M. J., & Jacoby, W. B. (1974). Glutathione S-transferase. The first enzymatic step in mercapturic acid formation. Journal of biological chemistry, 249, 7130–7139.
Harayashiki, C. A. Y., Junior, A. S. V., de Souza Machado, A. A., da Costa Cabrera, L., Primel, E. G., Bianchini, A., & Corcini, C. D. (2013). Toxic effects of the herbicide Roundup in the guppy Poecilia vivipara acclimated to fresh water. Aquatic Toxicology, 142, 176--184.
Hodson, P. V., Klopper-Sams, P. J., Munkittrick, K. R., Lockhart, W. L., Metner, D. A., Luxon, P. L., Smith, I. R., Gangon, M. M., Servos, M., & Payne, J. F. (1991). Protocols for measuring mixed function oxygenases of fish liver. Canadian Technical Reports of Fisheries and Aquatic Sciences.
Hughes, E. M., & Gallagher, E. P. (2004). Effects of β-naphthoflavone on hepatic biotransformation and glutathione biosynthesis in largemouth bass (Micropterus salmoides). Marine Environmental Research, 58(2), 675–679.
Hyndman, C. A., Kieffer, J. D., & Benfey, T. J. (2003a). The physiological response of diploid and triploid brook trout to exhaustive exercise. Comparative Biochemistry and Physiology Part A: Molecular & Integrative Physiology, 134(1), 167–179.
Hyndman, C. A., Kieffer, J. D., & Benfey, T. J. (2003b). Physiology and survival of triploid brook trout following exhaustive exercise in warm water. Aquaculture, 221(1-4), 629–643.
Javed, M., Usmani, N., Ahmad I., Ahmad, M. (2015). Studies on the oxidative stress and gill histopathology in Channa punctatus of the canal receiving heavy metal-loaded effluent of Kasimpur Thermal Power Plant. Environmental Monitoring and Assessment, 187(1), 1--11.
Jebali, J., Sabbagh, M., Banni, M., Kamel, N., Ben-Khedher, S., M’hamdi, N., & Boussetta, H. (2013). Multiple biomarkers of pollution effects in Solea solea fish on the Tunisia coastline. Environmental Science and Pollution Research, 20(6), 3812–3821.
Jerez, S., Rodriguez, C., Cejas, J., Bolanos, A., & Lorenzo, A. (2006). Lipid dynamics and plasma level changes of 17β-estradiol and testosterone during the spawning season of gilthead seabream (Sparus aurata) females of different ages. Comparative Biochemistry and Physiology Part B: Biochemistry and Molecular Biology, 143(2), 180–189.
Karami, A., Christianus, A., Ishak, Z., Courtenay, S., Syed, M. A., Noor, A., & Noorshinah, H. (2010). Effect of triploidization on juvenile African catfish (Clarias gariepinus). Aquaculture International, 18(5), 851–858.
Karami, A., Christianus, A., Ishak, Z., Shamsuddin, Z. H., Masoumian, M., & Courtenay, S. C. (2012). Use of intestinal Pseudomonas aeruginosa in fish to detect the environmental pollutant benzo[a]pyrene. Journal of Hazardous Materials, 215–216, 108–114.
Karami, A., Christianus, A., Ishak, Z., Syed, M. A., & Courtenay, S. (2011a). The effects of intramuscular and intraperitoneal injections of benzo[a]pyrene on selected biomarkers in Clarias gariepinus. Ecotoxicology and Environmental Safety, 74(6), 1558–1566.
Karami, A., Christianus, A., Zokaeifar, H., Saad, K., Imraan, F., Shakibazadeh, S., Negarestan, H., & Courtenay, S. (2011b). Ovaprim treatment promotes oocyte development and milt fertilization rate in diploid and triploid African catfish (Clarias gariepinus). Aquaculture International, 19(6), 1025–1034.
Karami, A., Eghtesadi Araghi, P., Syed, M., & Wilson, S. (2015a). Chromosome preparation in fish: effects of fish species and larval age. International Aquatic Research, 1-10.
Karami, A., Teh, S. J., Zakaria, M. P., & Courtenay, S. C. (2015b). Ploidy-, gender-, and dose-dependent alteration of selected biomarkers in Clarias gariepinus treated with benzo[a]pyrene. Journal of Environmental Sciences. doi:10.1016/j.jes.2015.05.009.
Karlsen, O. A., Puntervoll, P., & Goksøyr, A. (2012). Mass spectrometric analyses of microsomal cytochrome P450 isozymes isolated from β-naphthoflavone-treated Atlantic cod (Gadus morhua) liver reveal insights into the cod CYPome. Aquatic Toxicology, 108, 2–10.
Kim, W.-K., Park, J.-W., Lim, E.-S., Lee, S.-K., Kim, J., Kim, S., Lee, S.-W., Choi, K., & Jung, J. (2014). Tissue-specific antioxidant responses in pale chub (Zacco platypus) exposed to copper and benzo[a]pyrene. Bulletin of Environmental Contamination and Toxicology, 92(5), 540–545.
Koenig, S., & Solé, M. (2012). Natural variability of hepatic biomarkers in Mediterranean deep-sea organisms. Marine Environmental Research, 79, 122–131.
Konishi, T., Kato, K., Araki, T., Shiraki, K., Takagi, M., & Tamaru, Y. (2005). A new class of glutathione S-transferase from the hepatopancreas of the red sea bream Pagrus major. Biochemical Journal, 388, 299–307.
Leggatt, R. A., & Iwama, G. K. (2003). Occurrence of polyploidy in the fishes. Reviews in Fish Biology and Fisheries, 13(3), 237–246.
Leggatt, R. A., Scheer, K. W., Afonso, L. O., & Iwama, G. K. (2006). Triploid and diploid rainbow trout do not differ in their stress response to transportation. North American Journal of Aquaculture, 68(1), 1–8.
Lemaire, P., Fӧrlin, L., & Livingstone, D. R. (1996). Responses of hepatic biotransformation and antioxidant enzymes to CYP1A-inducers (3-methylcholanthrene,[beta]-naphthoflavone) in sea bass (Dicentrarchus labrax), dab (Limanda limanda) and rainbow trout (Oncorhynchus mykiss). Aquatic Toxicology, 36(3-4), 141–160.
Lincoln, R. F., Scott, A. P. (1984). Sexual maturation in triploid rainbow trout, Salmo gairdneri Richardson. Journal of Fish Biology, 25, 385--392.
Lindström-Seppä, P. (1985). Seasonal variation of the xenobiotic metabolizing enzyme activities in the liver of male and female vendace (Coregonus albula L.). Aquatic Toxicology, 6(4), 323–331.
Martínez-Gómez, C., Campillo, J., Benedicto, J., Fernández, B., Valdés, J., García, I., & Sánchez, F. (2006). Monitoring biomarkers in fish (Lepidorhombus boscii and Callionymus lyra) from the northern Iberian shelf after the Prestige oil spill. Marine Pollution Bulletin, 53(5), 305–314.
Mathew, N., Kalyanasundaram, M., & Balaraman, K. (2006). Glutathione S-transferase (GST) inhibitors. Expert Opinion on Therapeutic Patents, 16(4), 431–444.
Olsvik, P. A., Lie, K. K., Sturve, J., Hasselberg, L., & Andersen, O. K. (2009). Transcriptional effects of nonylphenol, bisphenol A and PBDE-47 in liver of juvenile Atlantic cod (Gadus morhua). Chemosphere, 75(3), 360–367.
Otitoloju, A., Olagoke, O. (2011). Lipid peroxidation and antioxidant defense enzymes in Clarias gariepinus as useful biomarkers for monitoring exposure to polycyclic aromatic hydrocarbons. Environmental Monitoring and Assessment, 182(1), 205--213.
Otto, D. M., & Moon, T. W. (1996). Endogenous antioxidant systems of two teleost fish, the rainbow trout and the black bullhead, and the effect of age. Fish Physiology and Biochemistry, 15(4), 349–358.
Pereira, P., de Pablo, H., Dulce Subida, M., Vale, C., & Pacheco, M. (2009). Biochemical responses of the shore crab (Carcinus maenas) in a eutrophic and metal-contaminated coastal system (Óbidos lagoon, Portugal). Ecotoxicology and Environmental Safety, 72(5), 1471–1480.
Pfeifer, S., Schiedek, D., & Dippner, J. W. (2005). Effect of temperature and salinity on acetylcholinesterase activity, a common pollution biomarker, in Mytilus sp. from the south-western Baltic Sea. Journal of Experimental Marine Biology and Ecology, 320(1), 93–103.
Pham, R. T., Barber, D. S., & Gallagher, E. P. (2004). GSTA is a major glutathione S-transferase gene responsible for 4-hydroxynonenal conjugation in largemouth bass liver. Marine Environmental Research, 58(2), 485–488.
Ploemen, J., Van Ommen, B., van Iersel, M., Rompelberg, C., Verhagen, H., van Bladeren, P. (1996). Irreversible inhibition of cytosolic glutathione S-transferases. In Glutathione S-transferases: structure, functions and clinical applications (pp. 143–153). London: Taylor and Francis.
Richter, C. J. J., Henken, A. M., Eding, E. H., Van Doesum, J. H., De Boer, P. (1986). Induction of triploidy by cold-shocking eggs and performance of triploids in the African catfish, Clarias gariepinus (Burchell 1822). EIFAC/FAO Symposium on Selection, Hybridization and Genetic Engineering in Aquaculture of Fish and Shellfish for Consumption and Stocking (pp. 24). Bordeaux.
Ruus, A., Sandvik, M., Ugland, K. I., & Skaare, J. U. (2002). Factors influencing activities of biotransformation enzymes, concentrations and compositional patterns of organochlorine contaminants in members of a marine food web. Aqutic Toxicology, 61(1), 73–87.
Stegeman, J. J., Brouwer, M., Richard, T. D. G., Forlin, L., Fowler, B. A., Sanders, B. M., & van Veld, P. A. (1992). Molecular responses to environmental contamination: enzyme and protein systems as indicators of chemical exposure and effect. In R. J. Huggett, R. A. Kimerly, P. M. J. Mehrle, & H. L. Bergman (Eds.), Biomarkers: biochemical, physiological and histological markers of anthropogenic stress (pp. 235–335). Chelsea: Lewis Publishers.
Teles, M., Gravato, C., Pacheco, M., & Santos, M. (2004). Juvenile sea bass biotransformation, genotoxic and endocrine responses to β-naphthoflavone, 4-nonylphenol and 17β-estradiol individual and combined exposures. Chemosphere, 57(2), 147–158.
Tian, X., Song, E., Pi, R., Zhu, X., Liu, L., Ma, X., Dong, H., Liu, J., & Song, Y. (2012). Polychlorinated biphenyls and their different level metabolites as inhibitors of glutathione S-transferase isoenzymes. Chemico-biological interactions, 198(1), 1–8.
Vaccaro, E., Meucci, V., Intorre, L., Soldani, G., Di Bello, D., Longo, V., Gervasi, P. G., & Pretti, C. (2005). Effects of 17β-estradiol, 4-nonylphenol and PCB 126 on the estrogenic activity and phase I and II biotransformation enzymes in male sea bass (Dicentrarchus labrax). Aquatic Toxicology, 75(4), 293–305.
Van der Oost, R., Beyer, J., & Vermeulen, N. P. E. (2003). Fish bioaccumulation and biomarkers in environmental risk assessment: a review. Environmental Toxicology and Pharmacology, 13(2), 57–149.
Van Veld, P. A., Ko, U., Vogelbein, W. K., & Westbrook, D. J. (1991). Glutathione S-transferase in intestine, liver and hepatic lesions of mummichog (Fundulus heteroclitus) from a creosote-contaminated environment. Fish Physiology and Biochemistry, 9(4), 369–376.
Vuorinen, P. J., Keinänen, M., Vuontisjärvi, H., Barsiene, J., Broeg, K., Förlin, L., Gercken, J., Kopecka, J., Köhler, A., & Parkkonen, J. (2006). Use of biliary PAH metabolites as a biomarker of pollution in fish from the Baltic Sea. Marine Pollution Bulletin, 53(8-9), 479–487.
Washburn, B. S., Vines, C. A., Baden, D. G., Hinton, D. E., & Walsh, P. J. (1996). Differential effects of brevetoxin and β-naphthoflavone on xenobiotic metabolizing enzymes in striped bass (Morone saxatilis). Aquatic Toxicology, 35(1), 1–10.
Zhang, Y., Andersson, T., & Förlin, L. (1990). Induction of hepatic xenobiotic biotransformation enzymes in rainbow trout by β-naphthoflavone. Time-course studies. Comparative Biochemistry and Physiology Part B: Comparative Biochemistry, 95(2), 247–253.
Zhu, L., Dong, X., Xie, H., Wang, J., Wang, J., Su, J., & Yu, C. (2011). DNA damage and effects on glutathione‐S‐transferase activity induced by atrazine exposure in zebrafish (Danio rerio). Environmental Toxicology, 26(5), 480–488.
Acknowledgments
This paper was supported by E-Science grant (No. 01-02-05-SF0017), Ministry of Science and Technology and Innovation (MOSTI), Government of Malaysia.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Karami, A., Courtenay, S.C. Glutathione S-transferase activities in African catfish injected with β-naphthoflavone: effects of ploidy, gender, dose, and sampling time. Environ Monit Assess 187, 681 (2015). https://doi.org/10.1007/s10661-015-4906-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10661-015-4906-7