Abstract
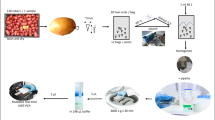
Virus screening is obligatory to avoid the spread of plant viruses regionally and globally. Double-antibody sandwich (DAS)-ELISA is the standard for screening potato viruses owing to its high-throughput potential, robustness, and cost–benefit ratio. However, low virus titers present in dormant potato tubers may not be reliably detected by using DAS-ELISA. Virus enrichment for reliable virus detection by DAS-ELISA assay is time-consuming and can be avoided by switching to more sensitive molecular biological techniques. Therefore, we developed a TaqMan® qPCR-based one-step protocol, termed direct reverse transcription quantitative PCR (DiRT-qPCR) for detection of RNA potato viruses PVY, PLRV and PVS without sophisticated nucleic acid purification and providing a high-throughput potential. Compared with DAS-ELISA, DiRT-qPCR showed up to a 100,000,000-fold higher sensitivity depending on the virus species. We also compared the qualitative results of standard DAS-ELISA used in seed potato certification, performed by sampling leaves of at least 4-weeks-old cultivated tuber eye cuttings, to the 1.5 h long DiRT-qPCR protocol on dormant tubers. The DiRT-qPCR protocol achieved an agreement with the DAS-ELISA procedure of 92.8%, 84.1% and 82.3% for the detection of PLRV, PVY, and PVS, respectively. The investigated different virus species show different multiplication behavior in secondary infected potato tuber eye cuttings, which is assumed to be a reason for the remaining qualitative differences in the outcome of the DiRT-qPCR and DAS-ELISA comparison. In our opinion, DiRT-qPCR protocol can be used as a reliable, work- and resource-saving alternative to DAS-ELISA in qualitative directed virus detection, particularly because no RNA purification is needed and dormant potato tubers can be directly used.

Similar content being viewed by others
References
Agindotan, B., & Perry, K. L. (2007). Macroarray detection of plant RNA viruses using randomly primed and amplified complementary DNAs from infected plants. Phytopathology, 97(1), 119–127. https://doi.org/10.1094/PHYTO-97-0119.
Agindotan, B. O., Shiel, P. J., & Berger, P. H. (2007). Simultaneous detection of potato viruses PLRV, PVA, PVX and PVY from dormant potato tubers by TaqMan real-time RT-PCR. Journal of Virological Methods, 142(1–2), 1–9. https://doi.org/10.1016/j.jviromet.2006.12.012.
Ahouee, K. H., Habibi, M. K., & Mosahebi, G. H. (2010). Detection of potato leafroll virus isolated from potato fields in Tehran province in aphids by immunocapture reverse transcription polymerase chain reaction. African Journal of Biotechnology, 9(16), 2349–2352.
Boonham, N., Laurenson, L., Weekes, R., & Mumford, R. (2009). Direct detection of plant viruses in potato tubers using real-time PCR. Methods in Molecular Biology, 508, 249–258. https://doi.org/10.1007/978-1-59745-062-1_19.
Casper, R. (1988). Luteoviruses. In: The Plant Viruses: Polyhedral virions with monopartite RNA genomes., R. Koenig, ed (Boston, MA: Springer US), pp. 235–258, ISBN:978-1-4612-8246-4.
Chung, B. Y.-W., Miller, W. A., Atkins, J. F., & Firth, A. E. (2008). An overlapping essential gene in the Potyviridae. Proceedings of the National Academy of Sciences of the United States of America, 105(15), 5897–5902. https://doi.org/10.1073/pnas.0800468105.
Cohen, J. (1960). A coefficient of agreement for nominal scales. Educational and Psychological Measurement, 20(1), 37–46. https://doi.org/10.1177/001316446002000104.
Dupuis, B. (2017). The movement of potato virus Y (PVY) in the vascular system of potato plants. European Journal of Plant Pathology, 147(2), 365–373. https://doi.org/10.1007/s10658-016-1008-5.
Faccioli, G., Panopoulos, N. J., & Gold, A. H. (1971). Kinetics of the carbohydrate metabolism in potato leafroll-infected Physalis floridana Rydb. Phytopathologia Mediterranea, 10(1), 1–25.
Foster, G. D., & Mills, P. R. (1992). The 3′-nucleotide sequence of an ordinary strain of potato virus S. Virus Genes, 6(3), 213–220. https://doi.org/10.1007/BF01702560.
Gallup, J. M. (2011). qPCR inhibition and amplification of difficult templates. In S. Kennedy & N. Oswald (Eds.), PCR troubleshooting and optimization: The essential guide. Caister Academic Press 9781904455721.
Gugerli, P., & Gehriger, W. (1980). Enzyme-linked immunosorbent assay (ELISA) for the detection of potato leafroll virus and potato virus Y in potato tubers after artificial break of dormancy. Potato Research, 23(3), 353–359. https://doi.org/10.1007/BF02360674.
Gundry, C.N., & Poulson, M.D. (2011). Obtaining maximum PCR sensitivity and specifity. In: S. Kennedy, N. Oswald (Eds.) PCR troubleshooting and optimization: The essential guide.Caister Academic Press. ISBN:9781904455721.
Hadersdorfer, J., Neumüller, M., Treutter, D., & Fischer, T. C. (2011). Fast and reliable detection of plum pox virus in woody host plants using the blue LAMP protocol. Annals of Applied Biology, 159(3), 456–466. https://doi.org/10.1111/j.1744-7348.2011.00510.x.
Hameed, A., Iqbal, Z., Asad, S., & Mansoor, S. (2014). Detection of multiple potato viruses in the field suggests synergistic interactions among potato viruses in Pakistan. The Plant Pathology Journal, 30(4), 407–415. https://doi.org/10.5423/PPJ.OA.05.2014.0039.
Harrington, R., Katis, N., & Gibson, R. W. (1986). Field assessment of the relative importance of different aphid species in the transmission of potato virus Y. Potato Research, 29(1), 67–76. https://doi.org/10.1007/BF02361982.
Hühnlein, A., Drechsler, N., Steinbach, P., Thieme, T., & Schubert, J. (2013). Comparison of three methods for the detection of potato virus Y in seed potato certification. Journal of Plant Diseases and Protection, 120(2), 57–69. https://doi.org/10.1007/BF03356455.
Hühnlein, A., Schubert, J., Zahn, V., & Thieme, T. (2016). Examination of an isolate of potato leaf roll virus that does not induce visible symptoms in the greenhouse. European Journal of Plant Pathology, 145(4), 829–845. https://doi.org/10.1007/s10658-016-0872-3.
Ju, H.-J. (2011). Simple and rapid detection of potato leafroll virus by reverse transcription loop-mediated isothermal amplification. The Plant Pathology Journal, 27(4), 385–389. https://doi.org/10.5423/PPJ.2011.27.4.385.
Kogovsek, P., Pompe-Novak, M., Petek, M., Fragner, L., Weckwerth, W., & Gruden, K. (2016). Primary metabolism, phenylpropanoids and antioxidant pathways are regulated in potato as a response to potato virus Y infection. PLoS One, 11(1), e0146135. https://doi.org/10.1371/journal.pone.0146135.
Landis, J. R., & Koch, G. G. (1977). The measurement of observer agreement for categorical data. Biometrics, 33(1), 159–174.
Leone, G., van Schijndel, H. B., van Genien, B., & Schoen, C. D. (1997). Direct detection of potato leafroll virus in potato tubers by immunocapture and the isothermal nucleic acid amplification method NASBA. Journal of Virological Methods, 66(1), 19–27. https://doi.org/10.1016/S0166-0934(97)02203-9.
Lin, Y.-H., Johnson, D. A., & Pappu, H. R. (2014). Effect of potato virus S infection on late blight resistance in potato. American Journal of Potato Research, 91(6), 642–648. https://doi.org/10.1007/s12230-014-9394-8.
Lindner, K., Trautwein, F., Kellermann, A., & Bauch, G. (2015). Potato virus Y (PVY) in seed potato certification. Journal of Plant Diseases and Protection, 122(3), 109–119. https://doi.org/10.1007/BF03356539.
Malnoe, P., Farinelli, L., Collet, G. F., & Reust, W. (1994). Small-scale field tests with transgenic potato, cv. Bintje, to test resistance to primary and secondary infections with potato virus Y. Plant Molecular Biology, 25(6), 963–975. https://doi.org/10.1007/BF00014670.
Martelli, G. P., Adams, M. J., Kreuze, J. F., & Dolja, V. V. (2007). Family Flexiviridae: A case study in virion and genome plasticity. Annual Review of Phytopathology, 45, 73–100. https://doi.org/10.1146/annurev.phyto.45.062806.094401.
Matousek, J., Schubert, J., Ptacek, J., Kozlova, P., & Dedic, P. (2005). Complete nucleotide sequence and molecular probing of potato virus S genome. Acta Virologica, 49(3), 195–205.
Mayo, M., Ryabov, E., Fraser, G., & Taliansky, M. (2000). Mechanical transmission of potato leafroll virus. Journal of General Virology, 81(Pt 11), 2791–2795. https://doi.org/10.1099/0022-1317-81-11-2791.
McDonald, M. S. (1976). Effects of leaf roll virus infection on the development of tubers in the potato plant. Potato Research, 19(4), 349–355. https://doi.org/10.1007/BF02359853.
McDonald, G. J., & Coleman, K. W. (1984). Detection of potato viruses Y and S in tubers by ELISA [enzyme-linked immunosorbent assay] after breaking of dormancy with bromoethane or rindite. American Potato Journal, 61(10), 619–622. https://doi.org/10.1007/BF02855113.
Miller, W. A., & Koev, G. (2000). Synthesis of subgenomic RNAs by positive-strand RNA viruses. Virology, 273(1), 1–8. https://doi.org/10.1006/viro.2000.0421.
Monteiro, L., Bonnemaison, D., Vekris, A., Petry, K. G., Bonnet, J., Vidal, R., Cabrita, J., & Mégraud, F. (1997). Complex polysaccharides as PCR inhibitors in feces: Helicobacter pylori model. Journal of Clinical Microbiology, 35(4), 995–998.
Mortimer-Jones, S. M., Jones, M. G., Jones, R. A., Thomson, G., & Dwyer, G. I. (2009). A single tube, quantitative real-time RT-PCR assay that detects four potato viruses simultaneously. Journal of Virological Methods, 161(2), 289–296. https://doi.org/10.1016/j.jviromet.2009.06.027.
Murashige, T., & Skoog, F. (1962). A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiologia Plantarum, 15(3), 473–497. https://doi.org/10.1111/j.1399-3054.1962.tb08052.x.
Nie, X. (2005). Reverse transcription loop-mediated isothermal amplification of DNA for detection of potato virus Y. Plant Disease, 89(6), 605–610. https://doi.org/10.1094/PD-89-0605.
Nyalugwe, E. P., Wilson, C. R., Coutts, B. A., & Jones, R. A. C. (2012). Biological properties of potato virus X in potato: Effects of mixed infection with potato virus S and resistance phenotypes in cultivars from three continents. Plant Disease, 96(1), 43–54. https://doi.org/10.1094/PDIS-04-11-0305.
Olspert, A., Carr, J. P., & Firth, A. E. (2016). Mutational analysis of the potyviridae transcriptional slippage site utilized for expression of the P3N-PIPO and P1N-PISPO proteins. Nucleic Acids Research, 44(16), 7618–7629. https://doi.org/10.1093/nar/gkw441.
Petrie, A., & Sabin, C. (2009). Medical statistics at a glance. Oxford: Wiley-Blackwell 140518051X.
PflKartV (1986). Pflanzkartoffelverordnung in der Fassung der Bekanntmachung vom 23. November 2004 (BGBl. I S. 2918), die zuletzt durch Artikel 4 der Verordnung vom 9. Juni 2017 (BGBl. I S. 1614) geändert worden ist. Federal Ministry of Justice and Consumer Protection.
Reiter, M., & Pfaffl, M. W. (2011). RT-PCR optimization strategies. In S. Kennedy & N. Oswald (Eds.), PCR troubleshooting and optimization: The essential guide. Caister Academic Press 9781904455721.
Rossen, L., Norskov, P., Holmstrom, K., & Rasmussen, O. F. (1992). Inhibition of PCR by components of food samples, microbial diagnostic assays and DNA-extraction solutions. International Journal of Food Microbiology, 17(1), 37–45.
Schori, M., Appel, M., Kitko, A., & Showalter, A. M. (2013). Engineered DNA polymerase improves PCR results for plastid DNA. Applications in Plant Sciences, 1(2). https://doi.org/10.3732/apps.1200519.
Schrader, C., Schielke, A., Ellerbroek, L., & Johne, R. (2012). PCR inhibitors – Occurrence, properties and removal. Journal of Applied Microbiology, 113(5), 1014–1026. https://doi.org/10.1111/j.1365-2672.2012.05384.x.
Singh, R. P., Nie, X., & Singh, M. (2000). Duplex RT-PCR: Reagent concentrations at reverse transcription stage affect the PCR performance. Journal of Virological Methods, 86(2), 121–129. https://doi.org/10.1016/S0166-0934(00)00138-5.
Singh, R. P., McLaren, D. L., Nie, X., & Singh, M. (2003). Possible escape of a recombinant isolate of potato virus Y by serological indexing and methods of its detection. Plant Disease, 87(6), 679–685. https://doi.org/10.1094/PDIS.2003.87.6.679.
Singh, M., Singh, R. P., Fageria, M. S., Nie, X., Coffin, R., & Hawkins, G. (2013). Optimization of a real-time RT-PCR assay and its comparison with ELISA, conventional RT-PCR and the grow-out test for large scale diagnosis of potato virus Y in dormant potato tubers. American Journal of Potato Research, 90(1), 43–50. https://doi.org/10.1007/s12230-012-9274-z.
Spiegel, S., & Martin, R. R. (1993). Improved detection of potato leafroll virus in dormant potato tubers and microtubers by the polymerase chain reaction and ELISA. Annals of Applied Biology, 122(3), 493–500.
Stammler, J., Hadersdorfer, J., Neumüller, M., Kellermann, A., & Treutter, D. (2014). Poster: Ansatz zur Optimierung des molekularen Nachweises von Kartoffelviren (approach for optimization of molecular detection of potato viruses). Julius-Kühn-Archiv (447).
Stare, T., Ramšak, Ž., Blejec, A., Stare, K., Turnšek, N., Weckwerth, W., Wienkoop, S., Vodnik, D., & Gruden, K. (2015). Bimodal dynamics of primary metabolism-related responses in tolerant potato-potato virus Y interaction. BMC Genomics, 16, 716. https://doi.org/10.1186/s12864-015-1925-2.
Struik, P. C., & Wiersema, S. G. (1999). Seed potato technology. Wageningen Pers: Wageningen. isbn:9074134653.
Syller, J. (1996). Potato leafroll virus (PLRV): Its transmission and control. Integrated Pest Management Reviews, 1(4), 217–227. https://doi.org/10.1007/BF00139765.
Vallejo, C. D., Gutiérrez, S. P. A., & Marín, M. M. (2016). Genome characterization of a Potato virus S (PVS) variant from tuber sprouts of Solanum phureja Juz. et Buk. Agronomía Colombiana, 34(1), 51–60. https://doi.org/10.15446/agron.colomb.v34n1.53161.
Varga, A., & James, D. (2006). Use of reverse transcription loop-mediated isothermal amplification for the detection of plum pox virus. Journal of Virological Methods, 138(1–2), 184–190. https://doi.org/10.1016/j.jviromet.2006.08.014.
Wardrop, E. A., Gray, A. B., Singh, R. P., & Peterson, J. F. (1989). Aphid transmission of potato virus S. American Potato Journal, 66(8), 449–459. https://doi.org/10.1007/BF02855437.
Wei, T., Lu, G., & Clover, G. (2008). Novel approaches to mitigate primer interaction and eliminate inhibitors in multiplex PCR, demonstrated using an assay for detection of three strawberry viruses. Journal of Virological Methods, 151(1), 132–139. https://doi.org/10.1016/j.jviromet.2008.03.003.
Weller, S. A., Elphinstone, J. G., Smith, N. C., Boonham, N., & Stead, D. E. (2000). Detection of Ralstonia solanacearum strains with a quantitative, multiplex, real-time, fluorogenic PCR (TaqMan) assay. Applied and Environmental Microbiology, 66(7), 2853–2858.
Acknowledgements
We thank Dr. Petr Dedic, who worked at the Potato Research Institute Havlíčkův Brod (Czech Republic) for providing virus infected in vitro potato plant material for assay validation and assay sensitivity comparison, Johannes Hertrich for technical assistance and the technical assistance team of the Bavarian State Research Center for Agriculture (Freising, Germany) for conducting the DAS-ELISA assay. In addition, we want to thank Thomas Eckl (Bavarian State Research Center for Agriculture, Freising, Germany) for support in statistical calculation and Dr. Miriam Prinz (Bavarian State Research Center, Freising, Germany) for article proof reading.
We also thank the former head of the Associate Professorship of Fruit Science Prof. Dr. Dieter Treutter who passed away and the current temporary head Prof. Dr. Wilfried Schwab for their support.
Funding
This work was funded by the Bavarian State Ministry for Food, Agriculture and Forestry (Stmelf). Furthermore, there was a financial contribution by the National Association of seed potato growers Bayern e. V. (Landesverband der Saatkartoffel-Erzeuger-Vereinigungen in Bayern e.V.) and by the potato growers of the German Society for Plant Breeding e.V. (Gesellschaft für Pflanzenzüchtung e.V., GPZ).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Stammler, J., Oberneder, A., Kellermann, A. et al. Detecting potato viruses using direct reverse transcription quantitative PCR (DiRT-qPCR) without RNA purification: an alternative to DAS-ELISA. Eur J Plant Pathol 152, 237–248 (2018). https://doi.org/10.1007/s10658-018-1468-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10658-018-1468-x