Summary
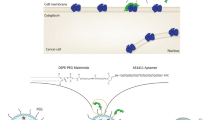
This study aimed to prepare SLC7A5 transporters targeted liposomes of Ribociclib (RB) by stear(o)yl conjugation of Phe, Asp, Glu amino acids to liposomes as targeting moieties. The liposomes were optimized for their formulations. Cell analysis on two cell lines of MCF-7 and NIH-3T3 were done including; cell viability test by MTT assay, cellular uptake, and cell cycle arrest by flow cytometry. The optimal liposomes showed the particle size of 123.6 ± 1.3 nm, drug loading efficiency and release efficiency of 83.87% ± 1.33% and 60.55% ± 0.46%, respectively. The RB loaded liposomes showed no hemolysis activity. Targeted liposomes increased cytotoxicity on MCF-7 cells more significantly than NIH-3T3 cells. Cell flow cytometry indicated that targeted liposomes uptake was superior to plain (non-targted) liposomes and free drug. Free drug and RB-loaded liposomes interrupted cell cycle in G1. However, amino acid-targeted liposomes arrested cells more than the free drug at this stage. Targeted liposomes reduced cell cycle with more interruption in the G2/M phase compared to the negative control.
Graphical abstract










Similar content being viewed by others
Data availability
All data are available on request.
References
Lima SM et al (2021) Global breast cancer incidence and mortality trends by region, age-groups, and fertility patterns. EClinMedicine 38:100985
Al-Mahmood S et al (2018) Metastatic and triple-negative breast cancer: challenges and treatment options. Drug Deliv Transl Res 8(5):1483–1507
Finn RS et al (2016) Targeting the cyclin-dependent kinases (CDK) 4/6 in estrogen receptor-positive breast cancers. Breast Cancer Res 18(1):17
Tosello G et al (2018) Breast surgery for metastatic breast cancer. Cochrane Database System Rev 3:CD011276
Slamon DJ et al (2020) Overall survival with Ribociclib plus fulvestrant in advanced breast cancer. New Eng J Med 382(6):514–524
Shah A et al (2018) FDA approval: Ribociclib for the treatment of postmenopausal women with hormone receptor-positive, HER2-negative advanced or metastatic breast cancer. Clin Cancer Res 24(13):2999–3004
Arora S et al (2017) Project ECHO: an effective means of increasing palliative care capacity. Am J Manag Care 23(7):SP267–SP271
Liu D et al (2016) The smart drug delivery system and its clinical potential. Theranostics 6(9):1306–1323
Hurler J et al (2013) The effect of lipid composition and liposome size on the release properties of liposomes-in-hydrogel. Int J Pharm 456(1):49–57
Guimarães D et al (2021) Design of liposomes as drug delivery system for therapeutic applications. Int J Pharm 601:120571
Yin Y et al (2022) Choline-induced SLC5A7 impairs colorectal cancer growth by stabilizing p53 protein. Cancer Let 525:55–66
Gynther M et al (2010) Brain uptake of ketoprofen-lysine prodrug in rats. Int J Pharm 399(1–2):121–128
Wu W et al (2015) Aspartate-modified doxorubicin on its N-terminal increases drug accumulation in LAT1-overexpressing tumors. Cancer Sci 106(6):747–756
Shennan DB et al (2004) L-Leucine transport in human breast cancer cells (MCF-7 and MDA-MB-231): Kinetics, regulation by estrogen and molecular identity of the transporter. Biochim et Biophy Acta - Biomembranes 1664(2):206–216
Metselaar JM et al (2003) A novel family of l-amino acid-based biodegradable polymer-lipid conjugates for the development of long-circulating liposomes with effective drug-targeting capacity. Bioconjug Chem 14(6):1156–1164
Li L et al (2016) Large amino acid transporter 1 mediated glutamate modified docetaxel-loaded liposomes for glioma targeting. Colloid Surf B: Biointerf 141:260–267
Li L et al (2017) Targeting tumor highly-expressed LAT1 transporter with amino acid-modified nanoparticles: Toward a novel active targeting strategy in breast cancer therapy. Nanomed Nanotech Biol Med 13(3):987–998
Wang Z et al (2019) Tyrosine modified irinotecan-loaded liposomes capable of simultaneously targeting LAT1 and ATB0,+ for efficient tumor therapy. J Control Rel 316:22–33
Brandl M (2001) Liposomes as drug carriers: A technological approach. Biotechnol Annu Rev 7:59–85
Pinto F et al (2018) Design of multifunctional nanostructured lipid carriers enriched with α-tocopherol using vegetable oils. Ind Crops Product 118:149–159
Kępczyński M et al (2008) Which physical and structural factors of liposome carriers control their drug-entrapment efficiency. Chem Phys Lipid 155(1):7–15
Zhao L et al (2015) Preparation of liposomes using supercritical carbon dioxide technology: Effects of phospholipids and sterols. Food Res Int 77:63–72
Hua S, Wu SY (2013) The use of lipid-based nanocarriers for targeted pain therapies. Front Pharmacol 4:143
Accardo A, Morelli G (2015) Peptide-targeted liposomes for selective drug delivery: advantages and problematic issues. Biopolymers 104(5):462–479
Hong SS et al (2015) Effects of triglycerides on the hydrophobic drug loading capacity of saturated phosphatidylcholine-based liposomes. Int J Pharm 483(1–2):142–150
Kaddah S et al (2018) Cholesterol modulates the liposome membrane fluidity and permeability for a hydrophilic molecule. Food Chem Toxicol 113:40–48
Luo Q et al (2016) Transporter occluded-state conformation-induced endocytosis: Amino acid transporter ATB°+-mediated tumor targeting of liposomes for docetaxel delivery for hepatocarcinoma therapy. J Control Rel 243:370–380
Kim CK, Park DK (1987) Stability and drug release properties of liposomes containing cytarabine as a drug carrier. Arch Pharm Res 10(2):75–79
Papageorgiou F et al (2019) Physicochemical study of the protein–liposome interactions: influence of liposome composition and concentration on protein binding. J Liposome Res 29(4):313–321
Arnida et al (2011) Geometry and surface characteristics of gold nanoparticles influence their biodistribution and uptake by macrophages. Eur J Pharm Biopharm 77(3):417–423
Sartaj A et al (2022) Ribociclib nanostructured lipid carrier aimed for breast cancer: formulation optimization, attenuating in vitro specification, and in vivo scrutinization. BioMed Res Int 2022:6009309
Nakanishi T et al (2011) Solute carrier transporters as targets for drug delivery and pharmacological intervention for chemotherapy. J Pharm Sci 100(9):3731–3750
Salatin S et al (2015) Effect of the surface modification, size, and shape on cellular uptake of nanoparticles. Cell Biol Int 39(8):881–890
Dickson MA (2014) Molecular pathways: CDK4 inhibitors for cancer therapy. Clin Cancer Res 20(13):3379–3383
Kommineni N et al (2022) Stealth liposomal chemotherapeutic agent for triple negative breast cancer with improved pharmacokinetics. Nanotheranostics 6(4):424–435
Al-Shdefat R et al (2023) Ribociclib hybrid lipid–polymer nanoparticle preparation and characterization for cancer treatment. Polymers 15(13):2844
Acknowledgements
The authors appreciate financial support of the project by Vice Chancellery of Research of Isfahan University of Medical Sciences.
Funding
This work has been supported by Isfahan University of Medical Sciences (project No: 399182).
Author information
Authors and Affiliations
Contributions
Mahtab Afsharzadeh has done the entire project, Jaleh Varshosaz is the supervisor of the work and corresponding author of the manuscript, Mina Mirian has helped in cellular studies, and Farshid Hassanzadeh has helped in chemical synthesis of amino acid conjugates and interpretation of spectra.
Corresponding author
Ethics declarations
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Afsharzadeh, M., Varshosaz, J., Mirian, M. et al. Targeted delivery of liposomal Ribociclib to SLC7A5 transporters in breast cancer cells. Invest New Drugs 42, 89–105 (2024). https://doi.org/10.1007/s10637-023-01409-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10637-023-01409-9