Abstract
Background
The aim of the meta-analysis was to derive a range of mean normal clinical electrooculogram (EOG) values from a systematic review of published EOG studies that followed the guidelines of the ISCEV standard for clinical electro-oculography.
Methods
A systematic literature review was performed using four relevant databases limited to peer-reviewed articles in English between 1967 and February 2017. Studies reporting clinical EOG or FO normal values were included when the report used a standard 30° horizontal saccade, a retinal luminance of between 100 and 250 cd m−2, and had > 10 subjects in their normative values. The search identified 1145 articles after duplicates were removed with subsequent screening of the abstracts excluding a further 1098, resulting in 47 full-text articles that were then assessed by the author (PC) with a final nine articles meeting the inclusion criteria. An overall effect estimate using inverse variance-weighted meta-analysis was performed to estimate the mean values for the light peak/dark trough ratio (LP:DT ratio) (dilated and undilated), the time to the LP, the amplitude of the LP, dark trough (DT) and the fast oscillation (FO) peak-to-trough ratio from the included studies.
Results
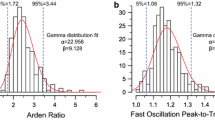
The mean dilated LP:DT ratio was 2.35 (95% CI 2.28–2.42); undilated LP:DT ratio was 2.37 (95% CI 2.28–2.45); LP amplitude was 835 (95% CI 631–1039) µV and the mean time to the LP being 8.2 (95% CI 7.7–8.7) min. The mean DT amplitude was 358 (95% CI 292–424) µV, and the mean FO peak-to-trough ratio was 1.13 (95% CI 1.11–1.16). The results of the LP/DT ratio are drawn from studies with a mean ± standard deviation (SD) age of 34.08 ± 12.93 years for dilated and 33.65 ± 12.28 years for undilated LP/DT ratios.
Conclusions
The meta-analysis of EOG studies has generated a reference range of normal mean values for clinicians to refer to when using the ISCEV clinical EOG. It provides a potential method to generate similar data sets from published normal values in related visual electrophysiology tests.







Similar content being viewed by others
References
Constable PA, Bach M, Frishman LJ, Jeffrey BG, Robson AG (2017) ISCEV Standard for clinical electro-oculography (2017 update). Doc Ophthalmol 134(1):1–9
McCulloch DL, Marmor MF, Brigell MG, Hamilton R, Holder GE, Tzekov R et al (2015) 2015) ISCEV Standard for full-field clinical electroretinography (2015 update. Doc Ophthal 130(1):1–12
Odom JV, Bach M, Brigell M, Holder GE, McCulloch DL, Mizota A et al (2016) ISCEV standard for clinical visual evoked potentials: (2016 update). Doc Ophthalmol 133(1):1–9
Hood DC, Bach M, Brigell M, Keating D, Kondo M, Lyons JS, et al (2012) ISCEV standard for clinical multifocal electroretinography (mfERG) (2011 edition). Doc Ophthalmol 124(1):1-13
Brigell M, Bach M, Barber C, Moskowitz A, Robson J (2003) Guidelines for calibration of stimulus and recording parameters used in clinical electrophysiology of vision. Doc Ophthal 107(2):185–193
Marmor MF, Zrenner E (1993) Standard for clinical electro-oculography. International Society for Clinical Electrophysiology of Vision. Doc Ophthalmol 85(2):115–124
Marmor MF (1998) Standardization notice: EOG standard reapproved. Electro-oculogram. Doc Ophthalmol. 95(1):91–92
Brown M, Marmor M, Vaegan Zrenner E, Brigell M, Bach M (2006) ISCEV Standard for Clinical Electro-oculography (EOG). Doc Ophthalmol 113(3):205–212
Marmor MF, Brigell MG, McCulloch DL, Westall CA, Bach M (2011) ISCEV standard for clinical electro-oculography (2010 update). Doc Ophthalmol 122(1):1–7
Arden GB, Barrada A, Kelsey JH (1962) A new clinical test of retinal function based upon the standing potential of the eye. Brit J Ophthalmol 46:449–467
Arden GB, Constable PA (2006) The electro-oculogram. Progress in Retinal & Eye Research 25(2):207–248
Constable PA (2014) A perspective on the mechanism of the light-rise of the electrooculogram. IOVS 55(4):2669–2673
Demet EM, Albers L, Sokolski KN, Chicz-Demet A, Chen CC, Reist C (1997) Apomorphine induced alteration in corneofundal potentials in depression. Prog Neuro-Psychopharmacology & Biological Psychiatry 21(5):775–788
Inoue M, Tamai A, Hatta S, Sasaki Y (2003) Slow and Fast Oscillation Electrooculography in Central Retinal Vein Occlusion: a Comparison between Affected Eyes and Fellow Intact Eyes. Yonago Acta Med 46(3):65–75
Nakao H, Miki N, Nagata M, Sasaki Y, Yamasaki A, Endo M et al (1995) Fast oscillations of the electro-oculogram in a series of normal subjects. Jap J Ophthalmol 39(2):113–119
Ozaki N, Rosenthal NE, Moul DE, Schwartz PJ, Oren DA (1993) Effects of phototherapy Winter seasonal affective-disorder on electrooculographic ratio in Winter seasonal affective-disorder. Psychiat Res 49(2):99–107
Türksever C, Orgül S, Todorova MG (2015) Comparing short-duration electro-oculograms with and without mydriasis in healthy subjects. Klin Monatsbl Augenheilkd 232(4):471–476
Schneck ME, Fortune B, Adams AJ (2000) The fast oscillation of the electrooculogram reveals sensitivity of the human outer retina/retinal pigment epithelium to glucose level. Vis Res 40(24):3447–3453
Zobor D, Strasser T, Zobor G, Schober F, Messias A, Strauss O et al (2015) Ophthalmological assessment of cannabis-induced persisting perception disorder: is there a direct retinal effect? Doc Ophthalmol 130(2):121–130
Moher D, Shamseer L, Clarke M, Ghersi D, Liberati A, Petticrew M et al (2015) Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Systematic Reviews 4(1):1
Krogh E (1975) Normal values in clinical electrooculography: i. Material, method, methodological investigations and distribution of the potential and time parameters. Acta Ophthalmol 53(4):563–575
Krogh E (1976) Normal values in clinical electrooculography: iI. Analysis of Potential and Time Parameters and Their Relation to Other Variables. Acta Ophthalmol 54(4):389–400
Momirov D, van Lilh GHM, van der Torren K, Vijfvinkel-Bruinenga S (1982) Normal values of the basic level of the standing potential and its light rise. Ophthalmologica 184(4):225–231
Thaler A, Heilig P, Gordesch J (1976) Light peak to dark trough ratio in clinical electro-oculography: influence of dark oscillations on the following light peak. Bibliotheca ophthalmologica: supplementa ad ophthalmologica. 85:110–114
Ohn YH, Katsumi O, Kruger-Leite E, Larson EW, Hirose T (1991) Electrooculogram in central retinal vein obstruction. Ophthalmologica 203(4):189–195
Papakostopoulos D, Bloom PA, Grey RH, Dean Hart JC (1992) The electro-oculogram in central retinal vein occlusion. Brit J Ophthalmol 76(9):515–519
Deng Y, Zhou H, Duan J (1994) Electro-oculogram of retinal vein occlusion. Yen Ko Hsueh Pao 10(1):60–64
Hara A, Miura M (1994) Decreased inner retinal activity in branch retinal vein occlusion. Doc Ophthalmol 88(1):39–47
Hozo SP, Djulbegovic B, Hozo I (2005) Estimating the mean and variance from the median, range, and the size of a sample. BMC Med Res Methodol 5(1):13
Jackson SA (1979) The optimum illuminance level for clinical electro-oculography. Acta Ophthalmol 57(4):665–668
Timmins N, Marmor MF (1992) Studies on the stability of the clinical electro-oculogram. Doc Ophthalmol 81(2):163–171
Lam RW, Beattie CW, Buchanan A, Remick RA, Zis AP (1991) Low electrooculographic ratios in patients with seasonal affective disorder. Amer J Psychiat 148(11):1526–1529
Seggie J, MacMillan H, Griffith L, Shannon HS, Martin J, Simpson J et al (1991) Retinal pigment epithelium response and the use of the EOG and arden ratio in depression. Psychiat Res 36(2):175–185
Lubinski W, Zajaczek S, Sych Z, Penkala K, Palacz O, Lubinski J (2004) Supernormal electro-oculograms in patients with neurofibromatosis type 1. Hered 2(4):193–196
Marmor MF, Wu KH (2005) Alcohol- and light-induced electro-oculographic responses: variability and clinical utility. Doc Ophthalmol 110(2–3):227–236
Arden GB, Wolf JE (2000) The human electro-oculogram: interaction of light and alcohol. IOVS 41(9):2722–2729
Thavikulwat AT, Lopez P, Caruso RC, Jeffrey BG (2015) The effects of gender and age on the range of the normal human electro-oculogram. Doc Ophthalmol 131(3):177–188
Caruso RC, Lopez P, Ayres LM, Kaiser-Kupfer MI (2003) Fast and slow oscillations of the electro-oculogram in Stargardt’s disease. IOVS 44:U712-U
Thompson DA, Constable PA, Liasis A, Walters B, Esteban MT (2016) The physiology of the retinal pigment epithelium in Danon disease. Retina 36(3):629–638
Low S, Davidson AE, Holder GE, Hogg CR, Bhattacharya SS, Black GC et al (2011) Autosomal dominant Best disease with an unusual electrooculographic light rise and risk of angle-closure glaucoma: a clinical and molecular genetic study. Mol Vis 17:2272–2282
Arden GB, Kelsey JH (1962) Some observations on the relationship between the standing potential of the human eye and bleaching and regeneration of visual purple. J Physiol (Lond) 161:205–226
Ozaki N, Rosenthal NE, Myers F, Schwartz PJ, Oren DA (1995) Effects of season on electro-oculographic ratio in winter seasonal affective disorder. Psychiat Res 59(1–2):151–155
Higgins JP, Thompson SG, Deeks JJ, Altman DG (2003) Measuring inconsistency in meta-analyses. BMJ 327(7414):557–560
Mergaerts F, Daems E, van Malderen L, Spileers W (2001) Recording of the fast oscillations in the human electro-oculogram. Doc Ophthalmol 103(1):63–72
Constable PA, Lawrenson JG, Arden GB (2006) Light and alcohol evoked electro-oculograms in cystic fibrosis. Doc Ophthalmol 113(3):133–143
De Rouck A, Kayembe D (1981) A clinical procedure for the simultaneous recording of fast and slow EOG oscillations. International Ophthalmol 3(3):179–189
Thorburn W, Nordstrom S (1978) EOG in a large family with hereditary macular degeneration. (Best’s vitelliform macular dystrophy) identification of gene carriers. Acta Ophthalmol 56(3):455–464
Schatz P, Bitner H, Sander B, Holfort S, Andreasson S, Larsen M et al (2010) Evaluation of macular structure and function by OCT and electrophysiology in patients with vitelliform macular dystrophy due to mutations in BEST1. IOVS 51(9):4754–4765
Weleber RG (1989) Fast and Slow oscillations of the electro-oculogram in Best’s Macular Dystrophy and Retinitis Pigmentosa. Arch Ophthalmol 107(4):530–537
Burgess R, Millar ID, Leroy BP, Urquhart JE, Fearon IM, De Baere E et al (2008) Biallelic mutation of BEST1 causes a distinct retinopathy in humans. Amer J Hum Genet 82(1):19–31
Xiao QH, Hartzell HC, Yu KA (2010) Bestrophins and retinopathies. Pflűgers Archiv-Eur J Physiol 460(2):559–569
Boon CJF, van den Born LI, Visser L, Keunen JEE, Bergen AAB, Booij JC et al (2013) Autosomal Recessive Bestrophinopathy: differential diagnosis and treatment options. Ophthalmology 120(4):809–820
Crowley C, Paterson R, Lamey T, McLaren T, De Roach J, Chelva E et al (2014) Autosomal recessive bestrophinopathy associated with angle-closure glaucoma. Doc Ophthalmol 129(1):57–63
Toto L, Boon CJ, Di Antonio L, Battaglia Parodi M, Mastropasqua R, Antonucci I et al (2016) Bestrophinopathy: a spectrum of ocular abnormalities caused by the c.614T > C mutation in the BEST1 Gene. Retina 36(8):1586–1595
Carter DA, Smart MJ, Letton WV, Ramsden CM, Nommiste B, Chen LL et al (2016) Mislocalisation of BEST1 in iPSC-derived retinal pigment epithelial cells from a family with autosomal dominant vitreoretinochoroidopathy (ADVIRC). Sci Rep 6:33792
Goldberg MF, Lee FL, Tso MO, Fishman GA (1989) Histopathologic study of autosomal dominant vitreoretinochoroidopathy. Peripheral annular pigmentary dystrophy of the retina. Ophthalmology 96(12):1736–1746
Yardley J, Leroy BP, Hart-Holden N, Lafaut BA, Loeys B, Messiaen LM et al (2004) Mutations of VMD2 splicing regulators cause nanophthalmos and autosomal dominant vitreoretinochoroidopathy (ADVIRC). IOVS 45(10):3683–3689
Derekoy S, Kenar F, Yavas GF, Kusbeci T, Ozturk F, Aycicek A (2016) The influence of iris color and retina pigment epithelium melanin on allergic rhinitis. Ent Updates. 6(1):23–28
Lubinski W, Zajaczek S, Sych Z, Penkala K, Palacz O, Lubinski J (2001) Electro-oculogram in patients with neurofibromatosis type 1. Doc Ophthalmol 103(2):91–103
Williams C, Papakostopoulos D (1995) Electro-oculographic abnormalities in amblyopia. BJO 79(3):218–224
Pinckers A, Perret CM, Brink H (1990) The EOG in systemic disease: alopecia areata, lupus erythematodes, rheumatoid arthritis. Neuro-Ophthalmology 10(3):145–151
Leguire LE, Pappa KS, McGregor ML, Rogers GL, Bremer DL (1992) Electro-oculogram in vitamin A deficiency associated with cystic fibrosis. Short communication. Ophthal Paediat Genet 13(3):187–189
Kimizuka Y, Kiyosawa M, Tamai M, Takase S (1993) Retinal changes in myotonic dystrophy: clinical and follow-up evaluation. Retina 13(2):129–135
O’Brien C, Duvall-Young J, Brown M, Short C, Bone M (1993) Electrophysiology of type II mesangiocapillary glomerulonephritis with associated fundus abnormalities. BJO 77(12):778–780
Faktorovich EG, Kim RY (1996) Electrophysiologic and psychophysical abnormalities in a patient with membranoproliferative glomerulonephritis type II (dense-deposit disease) retinopathy. IOVS 37(3):S505
Ikeda H, Head GM, Ellis CJ (1994) Electrophysiological signs of retinal dopamine deficiency in recently diagnosed Parkinson’s disease and a follow up study. Vis Res 34(19):2629–2638
Reist C, Sokolski KN, Chen CC, Coskinas E, Demet EM (1995) The effect of sleep deprivation on motor impairment and retinal adaptation in Parkinson’s disease. Prog Neuro-Psychopharmacol Biol Psychiat 19(3):445–454
Sokolski KN, Reist C, Chen CC, DeMet EM (1995) Antidepressant responses and changes in visual adaptation after sleep deprivation. Psychiat Res 57(3):197–207
Rinaldi M, Libondi T, Cotticelli L, Rinaldi M, D’Onofrio C (1996) Electrofunctional study in Vitiligo patients without ocular changes. IOVS 37(3):S1037
Scuderi GL, Ricci F, Nucci C, Galasso MJ, Cerulli L (1998) Electro-oculography in pigment dispersion syndrome. Ophthalmic Res 30(1):23–29
Greenstein VC, Seiple W, Liebmann J, Ritch R (2001) Retinal pigment epithelial dysfunction in patients with pigment dispersion syndrome: implications for the theory of pathogenesis. Arch Ophthalmol 119(9):1291–1295
Arndt CF, Sari A, Ferre M, Parrat E, Courtas D, De Seze J et al (2001) Electrophysiological effects of corticosteroids on the retinal pigment epithelium. IOVS 42(2):472–475
Arden GB, Barrada A (1962) An analysis of the electrooculograms of a series of normal subjects. BJO 46:468–482
Pinckers A (1979) Clinical electro-oculography. Acta Ophthalmol 57(4):623–632
Adams A (1973) The normal electro-oculogram (E.O.G.). Acta Ophthalmol 51(4):551–561
Krogh E (1977) Normal values in clinical electrooculography: III. Numerical evaluation of two dimensionless EOG Parameters. Acta Ophthalmol 55(2):227–236
Thaler ARG, Lessel MR, Heilig P, Scheiber V (1982) The fast oscillation of the electro-oculogram. Influence of stimulus intensity and adaptation time on amplitude and peak latency. Ophthalmic Res 14(3):210–214
Trimble JL, Ernest JT, Newell FW (1977) Electro-oculography in infants. IOVS 16(7):668–670
Acknowledgements
The authors would like to thank Ms. Nikki May from Flinders University Library services for help in conducting the literature search.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers’ bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licensing arrangements), or non-financial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this manuscript.
Statement on the welfare of animals
This article does not contain any studies with animals performed by any of the authors.
Informed consent
For this type of study, formal consent is not required.
Statement of human rights
As this study does not contain any involvement of human or animal participants, ethical approval is not required or is applicable.
Rights and permissions
About this article
Cite this article
Constable, P.A., Ngo, D., Quinn, S. et al. A meta-analysis of clinical electro-oculography values. Doc Ophthalmol 135, 219–232 (2017). https://doi.org/10.1007/s10633-017-9616-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10633-017-9616-8