Abstract
Background and aims
Pseudocirrhosis is a poorly understood acquired morphologic change of the liver that occurs in the setting of metastatic malignancy and radiographically resembles cirrhosis. Pseudocirrhosis has been primarily described in metastatic breast carcinoma, with few case reports arising from other primary malignancies. We present 29 cases of pseudocirrhosis, including several cases from primary malignancies not previously described.
Methods
Radiologic, clinical, demographic, and biomedical data were collected retrospectively and analyzed. We compared clinical and radiologic characteristics and outcomes between patients with pseudocirrhosis arising in metastatic breast cancer and non-breast primary malignancies.
Results
Among the 29 patients, 14 had breast cancer and 15 had non-breast primaries including previously never reported primaries associated with pseudocirrhosis, melanoma, renal cell carcinoma, appendiceal carcinoid, and cholangiocarcinoma. Median time from cancer diagnosis to development of pseudocirrhosis was 80.8 months for patients with primary breast cancer and 29.8 months for non-breast primary (p = 0.02). Among all patients, 15 (52%) had radiographic features of portal hypertension. Radiographic evidence of portal hypertension was identified in 28.6% of breast cancer patients, compared to 73.3% of those with non-breast malignancies (p = 0.03).
Conclusion
Pseudocirrhosis has most commonly been described in the setting of metastatic breast cancer but occurs in any metastatic disease to the liver. Our study suggests that portal hypertensive complications are more common in the setting of non-breast primary cancers than in metastatic breast cancer. Prior exposure to multiple chemotherapeutic agents, and agents known to cause sinusoidal injury, is a common feature but not essential for the development of pseudocirrhosis.
Similar content being viewed by others
Introduction
Pseudocirrhosis describes morphologic changes of the liver seen radiographically that mimic cirrhosis but arise in the setting of metastatic malignancy. The imaging features of pseudocirrhosis include nodular liver surface contour, capsular retraction, parenchymal atrophy, and caudate lobe hypertrophy [1,2,3,4]. Despite similar radiographic findings and clinical manifestations, pseudocirrhosis is a distinct clinical entity from cirrhosis that correlates with several different histopathological findings [1].
In 1924, the earliest report of irregular, lobulated hepatic pathologic changes in a case of metastatic breast cancer described the findings as “hepar lobatum carcinomatosum” [5, 6]. This term was borrowed from the similar gross pathologic appearance with “hepar lobatum,” a complication of gummatous tertiary syphilis resulting in areas of hepatic fibrosis and hyperplasia [7,8,9]. The same phenomenon of cirrhosis-like morphologic changes in metastatic liver disease has been described using different terminologies in subsequent decades. “Metastatic carcinomatous liver cirrhosis” was first used in the 1950s [10,11,12,13]. The term “pseudocirrhosis” was used in the 1920s–1950s in reference to cirrhotic-like changes arising secondary to pericarditis or tuberculous disease [14, 15]. In 1994, Young et al. first applied the term “pseudocirrhosis” to describe cirrhosis-like hepatic changes in metastatic disease [16]. In recent history, “pseudocirrhosis” has been the dominant term in the literature used to describe this clinical and radiographic finding [6, 17].
Imaging features of cirrhosis have been described since the early days of cross-sectional imaging [18, 19]. While cross-sectional imaging signs using computed tomography (CT) and magnetic resonance imaging (MRI) were more commonly described, the technological advances and higher resolution of ultrasound in the USA led to a significant increase in its clinical use [20]. The combination of its relative low cost, safety, and the evolving quantitative metrics that led to increased specificity has led to the increased use of ultrasound as a screening tool [21].
Hepatic complications of chemotherapy, including nodular regenerative hyperplasia (NRH) and sinusoidal obstruction syndrome (SOS)/veno-occlusive disease (VOD) had been well described when the earliest applications of the term “pseudocirrhosis” to refer to cirrhosis-like morphologic changes arising in metastatic malignancy appeared in the 1990s [16, 22, 23]. Radiologic findings of NRH may range from normal liver to multiple nodules or large masses that are often hypodense on CT without significant enhancement [24].
An extensive review of pseudocirrhosis was recently published [6]. Few studies have characterized the natural history of patients with pseudocirrhosis [25, 26]. In contrast to cirrhosis, which is an end-stage manifestation of chronic liver disease, pseudocirrhosis may develop more rapidly, with imaging findings evolving over 1–3 months [1, 16]. In patients with metastatic malignancy, the development of pseudocirrhosis is an ominous finding, which is often associated with portal hypertensive complications, hepatocellular dysfunction, and early discontinuation of chemotherapy [25, 27]. In those who develop features of portal hypertension, the clinical presentation may resemble decompensated liver disease, with ascites, variceal hemorrhage, peripheral edema, and hepatic encephalopathy [1, 28]. Transjugular intrahepatic portosystemic shunt (TIPS) placement has been used for relief of symptoms of portal hypertension, with similar efficacy as in benign hepatic disease [29, 30]. Increased mortality is seen in patients who develop pseudocirrhosis and appears to be related primarily to hepatocellular dysfunction or progression of malignancy [25, 27, 31]. Those patients who develop ascites or esophageal or gastric varices as a manifestation of portal hypertension have also been shown to have poorer overall survival [32, 33].
Among 2.4 million cancer patients, 5% present with synchronous liver metastases [34]. The most common primary site is breast cancers for younger women (ages 20–50) and colorectal cancers for younger men. With older patients, a more heterogeneous population of cancers with liver metastases emerges including esophageal, stomach, small intestine, melanoma, and bladder cancer in addition to the large proportion of lung, pancreatic, and colorectal cancers [34]. The incidence of distant metastasis at initial diagnosis of breast cancer is 5–8%, but in late recurrent breast cancer, 50–80% of patients have metastatic disease involving the liver [35, 36]. The incidence of pseudocirrhosis among breast cancer patients with metastasis to liver has been reported to be as high as 50–55% [25, 37]. Approximately 75% of metastatic breast cancer patients receiving chemotherapy demonstrate some degree of hepatic contour abnormality [4].
Pseudocirrhosis has been most frequently described in the setting of metastatic breast cancer [38,39,40,41,42]. Apart from breast cancer, cases have been reported in primary malignancies, such as pancreatic [43], colorectal [44, 45], gastric [46,47,48], esophageal [49], lung [50, 51], ovarian [52], and medullary thyroid cancers [53].
We report the clinical and radiographic findings of 29 cases of patients with pseudocirrhosis associated with various malignancies, including some previously not reported, from our institution. The goals of this study were to better characterize the radiographic and clinical findings of patients with pseudocirrhosis. In addition, we compare and contrast findings of pseudocirrhosis in breast cancer with non-breast cancer patients. Lastly, we discuss possible pathophysiological causes for its development and identify therapies that may be associated with the development of pseudocirrhosis and its portal hypertensive complications.
Methods
Patients were identified through a search of available radiology examinations of Keck Medical Center of the University of Southern California and Los Angeles County + University of Southern California Medical Center. The keyword “pseudocirrhosis” was identified in radiology reports using the mPower (Nuance, Burlington, MA) search tool for any patient prior to October 2022. Patients were excluded if there was an underlying chronic liver disease (which could independently cause cirrhosis), including chronic viral hepatitis, alcohol-associated hepatitis, or known cirrhosis prior to a radiologic diagnosis of pseudocirrhosis. Patients were also excluded if original imaging studies were not available for review.
Radiologic images were reviewed by board-certified radiologists with additional fellowship training in abdominal radiology for findings of pseudocirrhosis and the presence or absence of features of portal hypertension. For each patient, all radiologic studies available were reviewed to identify the study that initially demonstrated findings of pseudocirrhosis. Each study was assessed for the presence of liver surface nodularity, ascites, splenomegaly, and portosystemic collaterals.
Each patient’s electronic medical record was reviewed and demographic data, including age, gender, and race were collected. Patients’ oncologic history was reviewed, and data collected included primary malignancy, date of diagnosis, histologic findings, sites of metastasis, treatment course, outcomes, and therapeutic agents received. Medical charts were also reviewed for the presence of clinical manifestations of portal hypertension, including ascites, lower extremity edema, hepatic encephalopathy, and varices. Laboratory test results including complete blood count, comprehensive metabolic panel, and coagulation studies were collected from the date of diagnosis of pseudocirrhosis, as well as baseline and final values at the earliest and latest available encounter within each patient’s record.
Statistical Analysis
For continuous measurement, histogram and Shapiro–Wilk tests were used to assess data normality. For normally distributed data, the difference between primary breast and nonprimary breast group was tested by independent t test, otherwise Wilcoxon rank sum test was used. For category measurements, chi-square test was used for the group comparison. However, for any cell in the contingency table with sample size < 5, Fisher’s exact test was used. For longitudinal comparisons between the earliest available (baseline) laboratory values, laboratory values at the time of pseudocirrhosis diagnosis, and the last values available in the medical record, mixed-effect model was used for global comparison with post hoc Tukey’s test for correcting multiple comparison errors. Since the data normality was poor, Wilcoxon ranking score transformation was used before mixed model fitting. Model integrity was examined by residual plots. SAS 9.4 was used for all data analysis.
All aspects of this study were approved by the University of Southern California Institutional Review Board.
Results
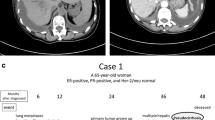
Twenty-nine patients with a radiologic diagnosis of pseudocirrhosis were identified and their demographic and clinical characteristics are shown in Table 1. Fourteen patients (48%) had primary breast cancer, 4 (14%) neuroendocrine (each with pancreas, appendiceal, prostate, and small cell lung, respectively), 4 (14%) colorectal, and 2 had melanoma (7%). There was a single patient with each of the following primary malignancies: prostate, ovary, renal cell carcinoma, and unknown primary. Hormone receptor status was known in 13 of 14 of the patients with breast cancer. Of these, 92% were estrogen receptor positive, 69% progesterone receptor positive, and 31% human epidermal growth factor receptor 2 (HER2) positive. The median age of all patients was 61 (IQR 56–69) years. Twenty patients (69%) were female. Seventeen patients (59%) were white, 4 (14%) Asian, 1 (3%) Black, 2 (7%) Hispanic or Latino. Race was not reported in 5 patients (17%). The overall median time from initial diagnosis of the patient’s malignancy to radiographic diagnosis of pseudocirrhosis was 71.7 (IQR 28–147.1) months. Time to development of pseudocirrhosis for the breast cancer patients was significantly longer than non-breast with a median of 80.8 months (IQR 39.7–211.8) and 29.8 months (IQR 11.9–136.4), respectively (p = 0.02). Figure 1 illustrates the development and progression of pseudocirrhosis and radiographic findings of portal hypertension over the course of 12 months.
Serial changes in the imaging appearances of the liver noted in a 67-year-old female with metastatic breast cancer. All images are axial contrast-enhanced CT images of the upper abdomen. Surgical clips are from an unrelated left nephrectomy. Top left: A few Hypodense lesions consistent with the hepatic metastases. Top right: 3 months later, increasing size and number of lesions are seen with more obvious nodularity of the anterior surface of the liver. Bottom left: 6 months from initial image, increasing nodularity of the hepatic surface is seen. Bottom right: 1 year from the initial image, very prominent nodularity, capsular retraction, heterogeneity of hepatic texture, and presence of perihepatic ascites are seen with increasing splenic size and increased portosystemic collaterals all consistent with the classical appearance of cirrhosis or in this case pseudocirrhosis
Serum liver tests collected at baseline, at the time of initial radiographic diagnosis of pseudocirrhosis, and the last available set of data from each patient’s chart are shown in Table 2. Most patients showed a cholestatic pattern of liver injury at the time of diagnosis, with 25 of 27 patients with R factor < 2 and no patients having R factor > 5 (hepatocellular pattern). All patients had normal bilirubin at diagnosis, but 5 patients (17%) had increased bilirubin level ≥ 3.0 mg/dL at last follow-up.
Initial abdominal imaging studies that diagnosed pseudocirrhosis comprised CT in 25 (86%), ultrasound in 3 (10%), and MRI in one patient (4%). For inclusion into this study, all patients had nodularity of the liver surface. Figure 2 shows axial and coronal CT images of a patient with small cell lung cancer with diffuse hepatic metastases and nodular contour. Similar findings are seen in Fig. 3 that shows a patient with breast cancer and multifocal hypoechoic hepatic metastases with nodular liver surface and perihepatic ascites.
a, b Axial gray-scale image of the liver in a 32-year-old female with metastatic breast cancer. Color Doppler abdominal ultrasound images show reversal of flow in the main portal vein. Note the multifocal hypoechoic hepatic metastases with nodular liver surface and perihepatic ascites seen on gray-scale ultrasound
Overall, 15 patients (52%) were found to have features of portal hypertension, which was defined by the presence of splenomegaly, abdominopelvic collaterals, or ascites. Patients with ascites alone were only considered to have portal hypertension if there was documentation of serum ascites albumin gradient (SAAG) ≥ 1.1 g/dL. Twenty-one patients had ascites of any volume (72%), 8 had splenomegaly (28%), and 8 had portosystemic collaterals (28%). Among patients with ascites, the largest volume noted on any radiologic study was as follows: 5 patients (24%) had large, 2 (10%) moderate, 10 (48%) small, and 5 (24%) had trace volume ascites. Six patients (29%) underwent therapeutic paracentesis with more than 1 L of ascitic fluid removed. SAAG was available on 4 patients, all of whom had a level > 1.1 g/dL (mean SAAG 1.6 g/dL). Four patients were refractory to diuretic management and two of these had tunneled peritoneal drains placed for continued ascitic fluid removal. One patient had ascitic fluid pathology with atypical cells but did not have SAAG determination. Among the patients with venous collaterals, 4 had peri-esophageal/peri-gastric alone, 1 had peri-splenic alone, 1 had omental alone, and 1 had both peri-splenic and peri-gastric varices. Two patients had documented episodes of variceal hemorrhage and both patients survived the hospitalization.
Radiographic portal hypertension was seen in 29% of breast cancer patients compared to 73% of non-breast cancer patients (p = 0.03). Of 14 breast cancer patients, 10 developed ascites (71%), 2 developed collateralization (14%), and 1 patient developed splenomegaly (7%). 6 of the 10 patients who developed ascites had no other manifestations of portal hypertension and had no SAAG data available, so were not included as having portal hypertension. Among 15 non-breast cancer patients, 11 developed ascites (73%), 7 splenomegaly (47%), and 6 collaterals (40%). Figures 4 and 5 show examples of pseudocirrhosis and splenomegaly in patients with prostate cancer and colorectal cancer, respectively. The presence of portosystemic collaterals is illustrated in Figs. 5 and 6.
Altogether, 15 patients (52%) had radiographic features of portal hypertension. Of these, 10 patients had 2 or more signs of portal hypertension on imaging. Three patients were determined to have portal hypertension by the presence of high-SAAG ascites alone. One patient had ascites without splenomegaly or portosystemic collaterals but was determined to have portal hypertension by the presence of reversal of portal venous flow on Doppler ultrasound. Peripheral edema was documented in 16 patients (55%) and hepatic encephalopathy in one patient (3%). Figure 7 shows a patient with ovarian cancer and pseudocirrhosis who has ascites and portosystemic collaterals.
Twenty-seven patients (93%) received some form of chemotherapy; hormone therapy, and/or immunotherapy prior to the radiographic diagnosis of pseudocirrhosis. Twenty-three patients (79%) received a chemotherapeutic agent known to cause hepatic sinusoidal injury, including sinusoidal obstructive syndrome (reference Livertox.nih.gov). These chemotherapeutic agents included carboplatin (n = 10), paclitaxel (n = 10), palbociclib (n = 8), cyclophosphamide (n = 6), oxaliplatin (n = 7), doxorubicin (n = 5), gemcitabine (n = 5), leucovorin (n = 6), docetaxel (n = 3), methotrexate (n = 2), etoposide (n = 2), cisplatin (n = 1), and vinblastine (n = 1). Among the 23 patients who received one or more agents known to cause sinusoidal injury, 12 (52%) had radiographic findings consistent with portal hypertension in contrast to 3 of 6 (50%) patients that did not receive one of the above agents. Thirteen of 14 patients (93%) with breast cancer and 10 of 15 patients (67%) with non-breast primary received at least one of these agents.
The clinical and radiographic presentation of pseudocirrhosis among patients with breast cancer and other malignancies were compared (Table 3). Splenomegaly and radiographic portal hypertension were statistically significantly higher in non-breast primary patients (46.7%, p = 0.04 and 73.3%, p = 0.03) compared to breast cancer patients (7.1% and 28.6%, respectively).
At the time of data collection, 6 patients (21%) were deceased and 6 patients (21%) had been placed on hospice. For these patients, the average time to death or hospice was 256 days from the initial diagnosis of pseudocirrhosis. 5 patients (17%) were known to be living. Outcomes of the other 12 patients (41%) were unknown.
Discussion
Pseudocirrhosis is a radiologic diagnosis that describes morphologic changes of the liver mimicking cirrhosis that arise in the setting of metastatic malignancy. Most cases in the published literature are found in metastatic breast cancer [1, 4, 6, 26, 54], but cases have been reported in other primary malignancies, including pancreatic [43], colorectal [44, 45], gastric [46,47,48], esophageal [49], lung [50, 51], ovarian [52], and medullary thyroid cancers [53].
In this series, approximately half of the patients with pseudocirrhosis had metastatic breast cancer and of those the majority were hormone receptor positive, which is similar to what has been reported by others [1, 32, 33, 54, 55]. Patients with non-breast malignancies included several primary malignancies that have not previously been described as causing pseudocirrhosis, viz., renal cell carcinoma, appendiceal carcinoid, cholangiocarcinoma, and melanoma.
The prevalence of portal hypertension among patients with pseudocirrhosis has not been established. Engelman et al. identified portal hypertension in 40% of their series of 48 patients [26], Gopalakrishnan et al. reported 36% prevalence among 86 patients [27], while Oliai et al. reported its prevalence to be 11% among 37 patients [28]. Qayyum et al. analyzed serial CT scans of 91 women with breast cancer with hepatic metastases and found 75% of cases had hepatic contour abnormalities and only 10% of those patients developed portal hypertension [4]. The differences in prevalence arise from how radiographic portal hypertension was defined in the various studies. For instance, Qayyum et al. required the presence of at least two of findings, ascites, splenomegaly, or portosystemic collaterals [4], whereas Oliai et al. considered portal vein size, but not ascites [25]. Like Engelman and Gopalakrishnan, we defined portal hypertension by the presence of splenomegaly, abdominopelvic collaterals, or ascites with proven serum ascites albumin gradient > 1.1 g/dL [26, 27]. Among the 14 breast cancer patients in this series, 4 (29%) had at least one sign and 2 of these had 2 or more radiographic signs of portal hypertension. Furthermore, our study included only those patients with a diagnosis of pseudocirrhosis and did not include patients with hepatic metastases without the radiographic appearance of pseudocirrhosis.
Several studies have characterized pseudocirrhosis in metastatic breast cancer, but to our knowledge, none have compared findings with non-breast malignancies. Interestingly, the prevalence of radiographic portal hypertension among patients with non-breast malignancies was significantly greater than in breast cancer patients in our series. The highest prevalence of portal hypertension was seen in our patients with colorectal cancer. However, given the relatively small number of patients, it is not possible to draw definitive conclusions. It is unclear if this difference is due to sampling error or to differences in tumor characteristics or chemotherapeutic exposures.
The median time from cancer diagnosis to development of pseudocirrhosis was significantly longer for patients with non-breast primary malignancies. This difference may arise from earlier detection of breast cancer through screening, and the fact that liver metastasis in breast cancer more commonly occurs in recurrence [35]. A study using the Surveillance, Epidemiology, and End Results (SEER) database found that at the time of cancer diagnosis, 5.14% of patients with any cancer are found to have liver metastasis, in comparison to 1.4% of breast cancer patients [34]. On the other hand, liver metastases are seen in over 50% of patients with late recurrence of breast cancer [35, 36].
Our patients’ laboratory studies illustrate an overall trend toward progression of hepatic decompensation and cholestatic injury from the patients’ baseline studies after the diagnosis of pseudocirrhosis to the final values available in the chart. Serum albumin, bilirubin, platelet count, and coagulopathy progressively worsened. These laboratory changes may be confounded in some cases by chemotherapy or by systemic illness related to malignancy alone. Subgroup analysis of laboratory changes between breast and non-breast primary malignancies were not significantly different.
The pathogenesis of pseudocirrhosis remains poorly understood. Pseudocirrhosis has been infrequently described in chemotherapy-naive patients due to metastatic involvement of the liver parenchyma [56], but most of the published literature describes patients who have received treatment. A study of metastatic breast cancer patients demonstrated higher incidence in patients who had previously received multiple rounds of systemic chemotherapy [27]. Within our case series, two (7%) of our patients were chemotherapy naive at the time of diagnosis.
Several pathophysiologic mechanisms have been proposed, and it is likely that pseudocirrhosis actually represents several heterogeneous histopathologic etiologies that present with similar radiographic findings. One proposed pathophysiologic mechanism that has been described is a desmoplastic reaction that forms around regions of intrasinusoidal hepatic tumor [17, 56,57,58,59,60,61]. Other studies have suggested that a direct hepatotoxic effect of chemotherapy treatment may play a role, and some have implicated specific agents such as paclitaxel, trastuzumab, tamoxifen, regorafenib, and palbociclib [2, 37, 45, 62]. Interruption or pause of chemotherapy may result in resolution of imaging features of pseudocirrhosis [63]. Kang et al. describe a case of pseudocirrhosis in which cessation of chemotherapy resulted in improvement of portal hypertensive complications, lab abnormalities, and near resolution of cirrhotic liver appearance on imaging [43]. Multiple mechanisms for chemotherapeutic injury resulting in the characteristic radiographic appearance of pseudocirrhosis have been described. Certain chemotherapeutic agents may induce hepatic injury and ischemia, resulting in secondary NRH with the absence of significant fibrosis on liver biopsy [16]. In a case series of 22 patients with pseudocirrhosis by Young et al., 7 patients underwent liver biopsy with 6 showing histopathologic findings suggestive of NRH [16]. SOS, formerly known as “hepatic veno-occlusive disease,” is another potential etiology. SOS describes histopathological findings of sinusoidal fibrosis, centrilobular hepatocyte necrosis, and narrowing and fibrosis of central veins occurring after exposure to a drug or toxin [64]. Vuppalanchi et al. describe two cases of pseudocirrhosis with hepatic decompensation that had histologic and clinical findings suggesting SOS induced by palbociclib [37]. Alberti et al. describe five patients with metastatic breast carcinoma resulting in hepatic contour abnormalities and rapid hepatic failure, two of whom underwent percutaneous liver biopsy with findings revealing SOS [65].
To gain a better understanding of the potential causative agents in our patients, we identified all chemotherapeutic agents received that have had reported associations with NRH and SOS. In this series, 79% of the patients who received chemotherapy prior to their diagnosis of pseudocirrhosis had exposure to one or more agents known to cause sinusoidal injury, SOS, or NRH. However, the incidence of portal hypertension among patients receiving SOS/VOD-associated chemotherapy was similar to that of patients who did not receive these agents.
The main limitation to this study is the lack of histological correlation to provide a better understanding of the underlying pathophysiology of pseudocirrhosis. Since the majority of patients received chemotherapy associated with sinusoidal injury, it cannot be discerned whether this is a significant factor in the development of pseudocirrhosis. However, pseudocirrhosis has been reported in untreated patients and prior to the era of these drugs [11, 56].
Conclusion
Pseudocirrhosis is a radiologic diagnosis of hepatic morphologic changes resembling cirrhosis that occurs in the setting of metastatic cancer. It has been most frequently reported in the setting of recurrent metastatic breast cancer but may be seen in other cancers that metastasize to the liver. Our series includes patients with renal cell carcinoma, appendiceal carcinoid, cholangiocarcinoma, and melanoma, primary malignancies that have not yet been described in the literature. Among patients with pseudocirrhosis, portal hypertension is often present radiographically and clinically. In our series, portal hypertension developed more frequently in pseudocirrhosis from non-breast malignancies than those with breast cancer. Most cases of pseudocirrhosis are diagnosed after systemic chemotherapy, and associations have been made between exposure to specific chemotherapeutic agents and its development. Two cases in this series were treatment naïve, but the large majority received at least one chemotherapeutic agent prior to pseudocirrhosis diagnosis. Pseudocirrhosis likely encompasses several heterogeneous histopathologic entities.
Abbreviations
- CT:
-
Computed tomography
- MRI:
-
Magnetic resonance imaging
- NRH:
-
Nodular regenerative hyperplasia
- SOS:
-
Sinusoidal obstruction syndrome
- VOD:
-
Veno-occlusive disease
- HER2-receptor:
-
Human epidermal growth factor receptor 2
- SAAG:
-
Serum ascites albumin gradient
- IQR:
-
Interquartile range
References
Adike A, Karlin N, Menias C, Carey EJ. Pseudocirrhosis: a case series and literature review. Case Rep Gastroenterol. 2016;10:381–391.
Jeong WK, Choi SY, Kim J. Pseudocirrhosis as a complication after chemotherapy for hepatic metastasis from breast cancer. Clin Mol Hepatol. 2013;19:190–194.
Leshchinskiy S, Kanner C, Keating DP. Pseudocirrhosis. Abdom Radiol (NY). 2018;43:3197–3198.
Qayyum A, Lee GK, Yeh BM, Allen JN, Venook AP, Coakley FV. Frequency of hepatic contour abnormalities and signs of portal hypertension at CT in patients receiving chemotherapy for breast cancer metastatic to the liver. Clin Imaging. 2007;31:6–10.
Busni NA. Hepar lobatum carcinomatosum. Virchows Arch Path Anat. 1924;252:727–733.
Villani R, Di Cosimo F, Sangineto M, Romano AD, Serviddio G. Pseudocirrhosis and portal hypertension in patients with metastatic cancers: a systematic review and meta-analysis. Sci Rep. 2022;12:19865.
Honma K. Hepar lobatum carcinomatosum due to metastatic breast carcinoma. Virchows Arch A Pathol Anat Histopathol. 1987;410:465–469.
Qizilbash A, Kontozoglou T, Sianos J, Scully K. Hepar lobatum associated with chemotherapy and metastatic breast cancer. Arch Pathol Lab Med. 1987;111:58–61.
Chin NW, Chapman I, Jimenez FA. Complete chemotherapeutic regression of hepatic metastases with resultant hepar lobatum. Am J Gastroenterol. 1987;82:149–151.
Watson AJ. Diffuse intra-sinusoidal metastatic carcinoma of the liver. J Pathol Bacteriol. 1955;69:207–217.
Micolonghi T, Pineda E, Stanley MM. Metastatic carcinomatous cirrhosis of the liver; report of a case in which death followed hemorrhage from esophageal varices and hepatic coma. AMA Arch Pathol. 1958;65:56–62.
Borja ER, Hori JM, Pugh RP. Metastatic carcinomatosis of the liver mimicking cirrhosis: case report and review of the literature. Cancer. 1975;35:445–449.
Knouse P, Hancock C, Iwaz S, Kaiser P. Metastatic carcinomatosis cirrhosis: a rare pattern of metastasis. Cureus. 2019;11:e3876.
Gill AW, Craig T. Pericarditic pseudo-cirrhosis of the liver, or pick’s disease. Br Med J. 1929;1:241–242.
Strong GF. Pick’s disease (Mediastino-pericarditic Pseudo-cirrhosis of the Liver): a case, with pericardial resection and recovery. Can Med Assoc J. 1938;39:247–249.
Young ST, Paulson EK, Washington K, Gulliver DJ, Vredenburgh JJ, Baker ME. CT of the liver in patients with metastatic breast carcinoma treated by chemotherapy: findings simulating cirrhosis. Am J Roentgenol. 1994;163:1385–1388.
Jüngst C, Krämer J, Schneider G, Lammert F, Zimmer V. Subacute liver failure by pseudocirrhotic metastatic breast cancer infiltration. Ann Hepatol. 2013;12:834–836.
Harbin WP, Robert NJ, Ferrucci JT. Diagnosis of cirrhosis based on regional changes in hepatic morphology: a radiological and pathological analysis. Radiology. 1980;135:273–283.
Torres WE, Whitmire LF, Gedgaudas-McClees K, Bernardino ME. Computed tomography of hepatic morphologic changes in cirrhosis of the liver. J Comput Assist Tomogr. 1986;10:47–50.
Kudo M, Zheng RQ, Kim SR et al. Diagnostic accuracy of imaging for liver cirrhosis compared to histologically proven liver cirrhosis. A multicenter collaborative study. Intervirology. 2008;51 Suppl 1:17–26.
Yeom SK, Lee CH, Cha SH, Park CM. Prediction of liver cirrhosis, using diagnostic imaging tools. World J Hepatol. 2015;7:2069–2079.
Cerný E, Bucek J. Tumor-related partial pseudocirrhosis of the liver. Cesk Patol. 1992;28:53–56.
Shirkhoda A, Baird S. Morphologic changes of the liver following chemotherapy for metastatic breast carcinoma: CT findings. Abdom Imaging. 1994;19:39–42.
Dachman AH, Ros PR, Goodman ZD, Olmsted WW, Ishak KG. Nodular regenerative hyperplasia of the liver: clinical and radiologic observations. Am J Roentgenol. 1987;148:717–722.
Oliai C, Douek ML, Rhoane C et al. Clinical features of pseudocirrhosis in metastatic breast cancer. Breast Cancer Res Treat. 2019;177:409–417.
Engelman D, Moreau M, Lepida A, Zaouak Y, Paesmans M, Awada A. Metastatic breast cancer and pseudocirrhosis: an unknown clinical entity. ESMO Open. 2020;5:e000695.
Gopalakrishnan D, Shajihan A, Purysko AS, Abraham J. Pseudocirrhosis in breast cancer—experience from an academic cancer center. Front Oncol. 2021;11:679163.
Hyun BH, Singer EP, Sharrett RH. Esophageal varices and metastatic carcinoma of liver. A report of three cases and review of the literature. Arch Pathol. 1964;77:292–298.
Shreve LA, O’Leary C, Clark TWI, Stavropoulos SW, Soulen MC. Transjugular intrahepatic portosystemic shunt for the management of symptomatic malignant pseudocirrhosis. J Gastrointest Oncol. 2022;13:279–287.
Geeroms B, De Hertogh G, Vanslembrouck R, Wildiers H, Nevens F, Maleux G. Transjugular intrahepatic portosystemic shunt for the treatment of portal hypertension-induced refractory ascites due to metastatic carcinomatous liver disease. J Vasc Interv Radiol. 2018;29:1713–1716.
Kashyap R, Reddy R, Voona MK. Pseudocirrhosis of the liver in setting of metastatic carcinoma breast: an ominous sign to be remembered. Indian J Nucl Med. 2018;33:86–87.
Ma WL, Chang DY, Lin CH et al. Clinical outcomes of metastatic breast cancer in patients having imaging liver pseudocirrhosis with or without evident varices. Oncologist. 2022;27:1008–1015.
Huppert LA, Walker Z, Li M et al. Clinical characteristics and outcomes in patients with metastatic breast cancer and pseudocirrhosis: a single center retrospective cohort study. Breast Cancer Res Treat. 2023;197:137–148.
Horn SR, Stoltzfus KC, Lehrer EJ et al. Epidemiology of liver metastases. Cancer Epidemiol. 2020;67:101760.
Rashid NS, Grible JM, Clevenger CV, Harrell JC. Breast cancer liver metastasis: current and future treatment approaches. Clin Exp Metastasis. 2021;38:263–277.
Cummings MC, Simpson PT, Reid LE et al. Metastatic progression of breast cancer: insights from 50 years of autopsies. J Pathol. 2014;232:23–31.
Vuppalanchi R, Saxena R, Storniolo AMV, Chalasani N. Pseudocirrhosis and liver failure in patients with metastatic breast cancer after treatment with palbociclib. Hepatology. 2017;65:1762–1764.
Aoyagi T, Takabe K, Tamanuki T, Matsubara H, Matsuzaki H. Pseudocirrhosis after chemotherapy in breast cancer, case reports. Breast Cancer. 2018;25:614–618.
Zeina AR, Abu-Mouch S, Mari A. Pseudocirrhosis in metastatic breast cancer. Isr Med Assoc J. 2017;19:328.
Liu CH, Chao TY. Education and imaging. Hepatobiliary and pancreatic: pseudocirrhosis after chemotherapy. J Gastroenterol Hepatol. 2011;26:788.
Chandrakar V, Isaacs C. Breast cancer-related pseudocirrhosis and esophageal varices. Breast J. 2005;11:301–302.
Fournier C, Tisman G, Kleinman R, Park Y, Macdonald WD. Clinical evidence for overcoming capecitabine resistance in a woman with breast cancer terminating in radiologically occult micronodular pseudo-cirrhosis with portal hypertension: a case report. J Med Case Rep. 2010;4:112.
Kang SP, Taddei T, McLennan B, Lacy J. Pseudocirrhosis in a pancreatic cancer patient with liver metastases: a case report of complete resolution of pseudocirrhosis with an early recognition and management. World J Gastroenterol. 2008;14:1622–1624.
Battisti S, Guida FM, Pagliara E, Tonini G, Zobel BB, Santini D. Pseudocirrhosis after anti-EGFR-based neoadjuvant therapy for hepatic metastasis from colon cancer: a different point of view. Clin Colorectal Cancer. 2014;13:e13-15.
Kumamoto K, Endo S, Isohata N et al. Pseudocirrhosis caused by regorafenib in an advanced rectal cancer patient with multiple liver metastases. Mol Clin Oncol. 2017;6:63–66.
Mitani S, Kadowaki S, Taniguchi H, Muto H, Muro K. Pseudocirrhosis in gastric cancer with diffuse liver metastases after a dramatic response to chemotherapy. Case Rep Oncol. 2016;9:106–111.
Basinger JM, Tucci J, Kapp ME. A fatal case of metastatic gastric adenocarcinoma mimicking cirrhosis. Autops Case Rep. 2021;11:e2021325.
Shinoda T, Tanahashi T, Sakuratani T et al. Pseudocirrhosis after chemotherapy for gastric cancer with diffuse liver metastases: a case report. Mol Clin Oncol. 2022;16:11.
Kobashigawa C, Nakamoto M, Hokama A, Hirata T, Kinjo F, Fujita J. Pseudocirrhosis in metastatic esophageal cancer. South Med J. 2010;103:488–489.
Shijubou N, Sumi T, Keira Y et al. Pseudocirrhosis due to liver metastasis from lung adenocarcinoma. Thorac Cancer. 2021;12:2407–2410.
Nakano S, Suzuki T, Takase Y et al. Pseudocirrhosis caused by lung adenocarcinoma with diffuse liver metastasis: an autopsy case report. Thorac Cancer. 2021;12:2046–2049.
Zanazanian A, Kalani A, Tabibian JH. Pseudocirrhosis in a patient with new-onset dysphagia and history of ovarian carcinoma. ACG Case Rep J. 2018;5:e29.
Harry BL, Smith ML, Burton JR, Dasari A, Eckhardt SG, Diamond JR. Medullary thyroid cancer and pseudocirrhosis: case report and literature review. Curr Oncol. 2012;19:e36-41.
Adler M, Tang I, Gach MW, MacFaul G. Recurrent metastatic breast cancer presenting with portal hypertension and pseudocirrhosis. BMJ Case Rep. 2019;12:e231044.
Sonnenblick A, Appelbaum L, Peretz T. Liver failure on the background of pseudocirrhosis in patients with liver metastasis from breast cancer, who responded to treatment. Onkologie. 2011;34:199–201.
Nascimento AB, Mitchell DG, Rubin R, Weaver E. Diffuse desmoplastic breast carcinoma metastases to the liver simulating cirrhosis at MR imaging: report of two cases. Radiology. 2001;221:117–121.
Viguier J, De Muret A, Bacq Y. Ascites due to portal hypertension from breast cancer-related metastatic liver infiltration. Gastroenterol Clin Biol. 2006;30:903–905.
Sass DA, Clark K, Grzybicki D, Rabinovitz M, Shaw-Stiffel TA. Diffuse desmoplastic metastatic breast cancer simulating cirrhosis with severe portal hypertension: a case of “pseudocirrhosis.” Dig Dis Sci. 2007;52:749–752. https://doi.org/10.1007/s10620-006-9332-9.
Lee SL, Chang ED, Na SJ et al. Pseudocirrhosis of breast cancer metastases to the liver treated by chemotherapy. Cancer Res Treat. 2014;46:98–103.
Hidalgo-Blanco A, Aguirresarobe-Gil de San Vicente M, Aresti S, de Miguel E, Cabriada-Nuno JL. Pseudocirrhosis in metastatic breast cancer. Gastroenterol Hepatol. 2018;41:111–113.
Takata K, Mogi A, Yamauchi R, Shakado S, Hirai F. Pseudocirrhosis due to desmoplastic response to chemotherapy in breast cancer liver metastases. Cureus. 2022;14:e25321.
Schreiner SA, Gorman B, Stephens DH. Chemotherapy-related hepatotoxicity causing imaging findings resembling cirrhosis. Mayo Clin Proc. 1998;73:780–783.
Calistri L, Rastrelli V, Nardi C et al. Imaging of the chemotherapy-induced hepatic damage: yellow liver, blue liver, and pseudocirrhosis. World J Gastroenterol. 2021;27:7866–7893.
DeLeve LD, Valla DC, Garcia-Tsao G. American Association for the Study Liver Diseases. Vascular disorders of the liver. Hepatology. 2009;49:1729–1764.
Alberti N, Bechade D, Dupuis F et al. Hepar lobatum carcinomatosum associated with liver metastases from breast cancer: report of five cases. Diagn Interv Imaging. 2015;96:73–78.
Acknowledgments
None.
Funding
Open access funding provided by SCELC, Statewide California Electronic Library Consortium.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Sadlik, G., Anderson, RC., Lei, X. et al. Pseudocirrhosis: A Case Series with Clinical and Radiographic Correlation and Review of the Literature. Dig Dis Sci 69, 1004–1014 (2024). https://doi.org/10.1007/s10620-023-08226-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-023-08226-3