Abstract
Introduction
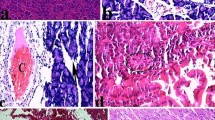
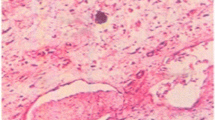
The present analysis deals with the biochemical and histopathological effects of l-carnitine in mice with l-asparaginase (ASNase)-induced experimental acute pancreatic injury (API).
Methods
A total of 32 male Balb/c mice were divided into four groups as follows. Group I (control) was injected with single saline via the intraperitoneal route. Group II received 500 mg/kg of l-carnitine daily with the injected volume of 62.5–75 μl for 25–30 g mice using a Hamilton microinjector applied for 5 days. Group III received a single 10,000 IU Escherichia coli ASNase/kg body weight dose of ASNase at a dose of 500 mg/kg. Group IV received 500 mg/kg of l-carnitine daily and a single dose of 500 mg/kg of ASNase and were decapitated on the fifth day following the injection. Blood and pancreatic tissue samples were obtained for evaluation of histopathological structure and levels of malondialdehyde (MDA), reduced glutathione (GSH), total sialic acid (TSA), glucose, amylase and triglyceride.
Results
In group III, compared to group IV and group I it was determined that levels of GSH and amylase were significantly lower while levels of MDA, TSA, glucose and triglyceride were higher. Levels of GSH, MDA, TSA, glucose, triglyceride and amylase, especially in group IV, approached that of group I. As a result, l-carnitine for ASNase-induced API mice may be protective against pancreatic tissue degeneration and oxidative stress or lipid peroxidation.

Similar content being viewed by others
References
Priest J, Ramsay N, Latchaw R, et al. Thrombotic and hemorrhagic strokes complicating early therapy for childhood acute lymphoblastic leukemia. Cancer. 1980;46:1548–1554.
Lee A, Levine M. The thrombophilic state induced by therapeutic agents in cancer patients. Semin Thromb Hemost. 1999;25:137–145.
Muller HJ, Boos J. Use of l-asparaginase in childhood ALL. Crit Rev Oncol Hematol. 1998;28:97–113.
Nakamura CT, Wilkinson R, Woodruff K. Pancreatitis and parotitis following therapy with l-asparaginase. Int Pediatr. 1999;14:25–27.
Cremer P, Lakomek M, Beck W, Prindull G. The effect of lasparaginase on lipid metabolism during induction chemotherapy of childhood lymphoblastic leukemia. Eur J Pediatr. 1988;147:64–67.
Alvarez OA, Zimmerman G. Pegaspargase-induced pancreatitis. Med Pediatr Oncol. 2000;34:200–205.
Knoderer HM, Robarge J, Flockhart DA. Predicting asparaginase-associated pancreatitis. Pediatr Blood Cancer. 2007;49:634–639.
Ortega JA, Nesbit ME, Donaldson MH, et al. l-Asparaginase, vincristine, and prednisone for induction of first remission in acute lymphocytic leukemia. Cancer Res. 1977;37:535–540.
Kelly GSL. Therapeutic applications of a conditionally-essential amino acid. Altern Med Rev. 1998;3:345–360.
Brass EM. Supplemental carnitine and exercise. Am J Clin Nutr. 2000;72:618–623.
Rani PJ, Panneerselvam C. Effect of l-carnitine on brain lipid peroxidation and antioxidant enzymes in old rats. J Gerontol A Biol Sci Med Sci. 2002;57:134–137.
Kalaiselvi T, Panneersalvam C. Effect of l-carnitine on the status of lipid peroxidation and antioxidants in aging rats. J Nutr Biochem. 1998;9:575–581.
Sillanaukee P, Pönniö M, Jääskeläinen IP. Occurrence of sialic acids in healthy humans and different disorders. Eur J Clin Invest. 1999;29:413–425.
Wang B, Brand-Miller J. The role and potential of sialic acid in human nutrition. Eur J Clin Nutr. 2003;57:1351–1369.
Draper HH, Hadley M. Malondialdehyde determination as index of lipid peroxidation. Methods Enzymol. 1990;186:421–431.
Kaya I, Karapehlivan M, Yilmaz M, Ersan Y, Koc E. Investigation of effects on plasma nitric oxide, malondialdehyde and total sialic acid levels of glyphosate in Kars creek transcaucasian barb (Capoeta capoeta [Guldenstaedt, 1773]) in Turkey. Fresenius Environ Bull. 2012;21:123–126.
Zaidi SM, Al-Qirim TM, Banu N. Effects of antioxidant vitamins on glutathione depletion and lipid peroxidation induced by restraint stress in the rat liver. Drugs R D. 2005;6:157–165.
Sydow G. A simplified quick method for determination of sialic acid in serum. Biomed Biochem Acta. 1985;44:1721–1723.
Beutler E, Duran O, Kelley BM. Improved method for determination of blood glutathion. J Lab Clin Med. 1963;28:882–888.
Yoshoiko T, Kawada K, Shimada T. Lipid peroxidation in maternal and cord blood and protective mechanism against activated-oxygen toxicity in the blood. Am J Obstet Gynecol. 1979;135:372–376.
Dolowy WC, Henson D, Cornet J, Sellin H. Toxic and antineoplastic effects of l-asparaginase. Study of mice with lymphoma and normal monkeys and report on a child with leukemia. Cancer. 1966;19:1813–1819.
Suzuki M, Shimizu T, Kudo T, Shoji H, Ohtsuka Y, Yamashiro Y. Octreotide prevents l-asparaginase—induced pancreatic injury in rats. Exp Hematol. 2008;36:172–180.
Top PC, Tissing WJ, Kuiper JW, Pieters R, van Eijck CH. l-asparaginase-induced severe necrotizing pancreatitis successfully treated with percutaneous drainage. Pediatr Blood Cancer. 2005;44:95–97.
Schleis SE, Rizzo SA, Phillips JC, LeBlanc AK. Asparaginase-associated pancreatitis in a dog. Can Vet J. 2011;52:1009–1012.
Rogers KS. L-asparaginase for treatment of lymphoid neoplasia in dogs. J Am Vet Med Assoc. 1989;194:1626–1630.
Putter J. Pharmacokinetic behavior of l-asparaginase in men and in animals. Recent Results Cancer Res. 1970;33:64–74.
Vaccaro MI, Grasso D, Ropolo A, Iovanna JL, Cerquetti MC. VMP1 expression correlates with acinar cell cytoplasmic vacuolization in arginine-induced acute pancreatitis. Pancreatology. 2003;3:69–74.
Boquist L. Morphologic effects of ethionine on the pancreas of the Chinese hamster. A light and electron microscopic study of degenerative changes. Acta Pathol Microbiol Scand. 1969;76:91–105.
Saito I, Hashimoto S, Saluja A, Steer ML, Meldolesi J. Intracellular transport of pancreatic zymogens during caerulein supramaximal stimulation. Am J Physiol. 1987;253:517–526.
Czakó L, Takács T, Varga ISZ, et al. Involvement of oxygen-derived free radicals in l-arginine-induced acute pancreatitis. Dig Dis Sci. 1998;43:1770–1777.
Sayed-Ahmed MM, Eissa MA, Kenawy SA, Mostafa N, Calvani M, Osman AM. Progression of cisplatin-induced nephrotoxicity in a carnitine-depleted rat model. Chemotherapy. 2004;50:162–170.
Ashraf V, Franco G, Syed I, Zbigniew B, Syed A. The protective role of l-carnitine against neurotoxicity evoked by drug of abuse, methamphetamine, could be related to mitochondrial dysfunction. Ann NY Acad Sci. 2002;965:225–232.
Dokmeci D, Akpolat M, Aydogdu N, Doganay L, Turan FN. l-carnitine inhibits ethanol-induced gastric mucosal injury in rats. Pharmacol Rep. 2005;57:481–488.
Yilmaz M, Karapehlivan M, Kaya I. Effects of zinc sulphate on transcaucasian barb (Capoeta capoeta [Guldenstaedt, 1773]) plasma nitric oxide, malondialdehyde and total sialic acid levels. Kafkas Univ Vet Fak Derg. 2012;18:61–64.
Kushner I. The acute phase response: an overview. Methods Enzymol. 1988;163:373–383.
Gruszewska E, Chrostek L, Cylwik B, et al. Serum sialic acid as a marker of pancreatic cancers. Clin Lab. 2013;59:781–788.
Gornik O, Gornik I, Gasparović V, Lauc G. Change in transferrin sialylation is a potential prognostic marker for severity of acute pancreatitis. Clin Biochem. 2008;41:504–510.
Chrostek L, Cylwik B, Krawiec A, Korcz W, Szmitkowski M. Relationship between serum sialic acid and sialylated glycoproteins in alcoholics. Alcohol. 2007;42:588–592.
Patel AB. Venkatakrishna Bhatt H. Effect of lead on the blood serum, liver and brain sialoglycoconjugate levels in rats. Hum Exp Toxicol. 1992;11:89–92.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kaya, I., Citil, M., Sozmen, M. et al. Investigation of Protective Effect of l-Carnitine on l-Asparaginase-Induced Acute Pancreatic Injury in Male Balb/c Mice. Dig Dis Sci 60, 1290–1296 (2015). https://doi.org/10.1007/s10620-014-3461-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-014-3461-3