Abstract
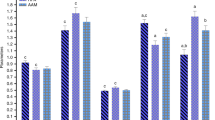
Mental retardation, which occurs in phenylketonuric patients, is associated with increased levels of phenylalanine, increased oxidative stress, and an imbalance of amino acids in the brain. Recent studies have shown that oxidative stress plays a role in the pathogenesis of phenylketonuria. In this work, we aimed to compare the influence of blood phenylalanine levels on oxidative stress parameters in phenylketonuric patients who divided patients into groups according to blood Phe levels during follow-up visits and compared these groups with healthy controls. Results showed significant differences in glutathione peroxidase (GSHPx), coenzyme Q10 (Q10), Q10/cholesterol, and l-carnitine levels in phenylketonuria patients and the control group. GSHPx, Q10, and Q10/cholesterol levels were significantly lower in poor adherence patients than in the control groups. l-carnitine levels were significantly increased in good adherence patients than poor adherence patients and decreased in poor adherence patients than healthy controls. No correlations were observed between phenylalanine and l-carnitine concentrations in poor adherence group. No significant differences were observed in paraoxonase 1 (PON1), total antioxidant status (TAS), total oxidant status (TOS) and oxidative stress index (OSI) levels. As a result, in this work, poor adherence patients are prone to oxidative stress. Although the patients may have the same diagnosis, patients have different clinical characteristics and different prognosis. Antioxidants can be used as an adjuvant therapy in order to avoid neurological damage in these patients.
Similar content being viewed by others
References
Artuch R et al (1999) Decreased serum ubiquinone-10 concentrations in phenylketonuria. Am J Clin Nutr 70:892–895
Artuch R et al (2001) Plasma phenylalanine is associated with decreased serum ubiquinone-10 concentrations in phenylketonuria. J Inherit Metab Dis 24(3):359–366
Artuch R et al (2004) A longitudinal study of antioxidant status in phenylketonuric patients. Clin Biochem 37:198–203
Barschak AG et al (2008) Oxidative stress in plasma from maple syrup urine disease patients during treatment. Metab Brain Dis 23:71–80
Camp KM et al (2014) Phenylketonuria scientific review conference: state of the science and future research needs. Mol Genet Metab 112(2):87–122
Castillo M, Zafra MF, Garcia-Peregrin E (1988) Inhibition of brain and liver 3-hydroxy-3-methylglutaryl-CoA reductase and mevalonate-5-pyrophosphate decarboxylase in experimental hyperphenylalaninemia. Neurochem Res 13(6):551–555
Colome C et al (2003) Lipophilic antioxidants in patients with phenylketonuria. Am J Clin Nutr 77(1):185–188
Deon M et al (2015) Urinary biomarkers of oxidative stress and plasmatic inflammatory profile in phenylketonuric treated patients. Int J Dev Neurosci 47:259–265
Derin N, Izgut-Uysal VN, Agac A, Aliciguzel Y, Demir N (2004) l-carnitine protects gastric mucosa by decreasing ischemia-reperfusion induced lipid peroxidation. J Physiol Pharmacol 55(3):595–606
Erel O (2004) A novel automated direct measurement method for total antioxidant capacity using a new generation, more stable ABTS radical cation. Clin Biochem 37(4):277–285
Erel O (2005) A new automated colorimetric method for measuring total oxidant status. Clin Biochem 38(12):1103–11111
Fischer GM et al (2000) Metabolism of carnitine in phenylacetic acid-treated rats and in patients with phenylketonuria. Biochim et Biophys Acta Mol Basis Dis 1501:200–210
Hagen K, Martine E et al (2002) Experimental hyperphenylalaninemia provokes oxidative stress in rat brain. Biochim et Biophys Acta Mol Basis Dis 1586:344–352
Halliwell B, Gutteridge JMC (2007) Free radicals in biology and medicine. Oxford University Press, Oxford
Hargreaves IP (2007) Coenzyme Q10 in phenylketonuria and mevalonic aciduria. Mitochondrion 7:175–180
Işık A, Selek S (2007) Total antioxidant response and oxidative stress in patients with rheumatoid arthritis. F Ü Sağ Bil Tıp Derg 21(2):67–73
Lu J, Holmgren A (2009) Selenoproteins. J Biol Chem 284(2):723–727
Matalon R et al (2007) Double blind placebo control trial of large neutral amino acids in treatment of PKU: effect on blood phenylalanine. J Inherit Metab Dis 30(2):153–158
Moraes TB et al (2010) Lipoic acid prevents oxidative stress in vitro and in vivo by an acute hyperphenylalaninemia chemically-induced in rat brain. J Neurol Sci 292(1–2):89–95
Ribas GS, Sitta A, Wajner M, Vargas CR (2011) Oxidative stress in phenylketonuria: what is the evidence? Cell Mol Neurobiol 31:653–662
Rocha JC, Martel F (2009) Large neutral amino acids supplementation in phenylketonuric patients. J Inherit Metab Dis 32:472–480
Rocha JC, Martins MJ (2012) Oxidative stress in phenylketonuria: future directions. J Inherit Metab Dis 35:381–398
Sanayama Y et al (2011) Experimental evidence that phenylalanine is strongly associated to oxidative stress in adolescents and adults with phenylketonuria. Mol Genet Metab 103(3):220–225
Schulpis KH, Tsakiris S, Karikas GA, Moukas M, Behrakis P (2003) Effect of diet on plasma total antioxidant status in phenylketonuric patients. Eur J Clin Nutr 57(2):383–387
Schulpis KH, Tsakiris S, Traeger-Synodinos J, Papassotiriou I (2005) Low total antioxidant status is implicated with high 8-hydroxy-2-deoxyguanosine serum concentrations in phenylketonuria. Clin Biochem 38(3):239–242
Schulpis KH, Bartzeliotou A, Tsakiris S, Gounaris A, Papassotiriou I (2007) Serum paraoxonase/arylesterase activities in phenylketonuric patients on diet. Eur J Clin Nutr 61:803–808
Scriver CR, Kaufman S (2001) Hyperphenylalaninemia: phenylalanine hydroxylase deficiency. In: The metabolic and molecular bases of inherited disease, pp 1667–1724
Selek S, Aslan M, Horoz M, Gur M, Erel O (2007) Oxidative status and serum PON1 activity in beta-thalassemia minor. Clin Biochem 40(5–6):287–291
Sierra C et al (1998) Antioxidant status in hyperphenylalaninemia. Clin Chim Acta 276:1–9
Sirtori LR et al (2005) Oxidative stress in patients with phenylketonuria. Biochem et Biophys Acta 1740:68–73
Sitta A et al (2006) Investigation of oxidative stress parameters in treated phenylketonuric patients. Metab Brain Dis 21:287–296
Sitta A et al (2009) l-carnitine blood levels and oxidative stress in treated phenylketonuric patients. Cell Mol Neurobiol 29:211–218
Sitta A et al (2011) Evidence that l-carnitine and selenium supplementation reduces oxidative stress in phenylketonuric patients. Cell Mol Neurobiol 31:429–436
Uysal S, Akyol S, Hasgül R, Armutcu F, Yiğitoğlu F (2011) Paraoxonase: a multifaceted enzyme. Yeni Tıp Dergisi 28(3):136–141
Van Bakel MME, Printzen G, Wermuth B, Wiesmann UN (2000) Antioxidant and thyroid hormone status in selenium-deficient phenylketonuric and hyperphenylalaninemic patients. Am J Clin Nutr 72(4):976–981
Vockley J et al (2014) Phenylalanine hydroxylase deficiency: diagnosis and management guideline. Genet Med 16(2):188–200
Weigel C et al (2008) Carnitine status in early-treated children, adolescents and young adults with phenylketonuria on low phenylalanine diets. Ann Nutr Metab 53:91–95
Acknowledgements
This study was supported by Gaziantep University Scientific Research Projects Governing Unit.
Author information
Authors and Affiliations
Contributions
BK, DSK, and BOH conceived the study design. BK, DSK, BOH, and HC performed the experiment. BK and DSK participated in the data analysis and interpretation. BOH helped to draft and revise the manuscript. All authors read and approved the final manuscripts.
Corresponding author
Ethics declarations
Conflict of interest
The authors (Burcu Kumru, Davut Sinan Kaplan, Burcu Oztürk Hismi, Hakim Celik) declare that there are no financial interests and/or no conflict of interest disclosure associated with this manuscript.
Rights and permissions
About this article
Cite this article
Kumru, B., Kaplan, D.S., Oztürk Hismi, B. et al. Effect of Blood Phenylalanine Levels on Oxidative Stress in Classical Phenylketonuric Patients. Cell Mol Neurobiol 38, 1033–1038 (2018). https://doi.org/10.1007/s10571-017-0573-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-017-0573-2