Abstract
The conventional production of nanocellulose from biomass demands multiple steps involving numerous chemicals like corrosive acids, bases, halogenated bleaching agents, and mechanical energy. This research aims to find a greener approach to producing nanocellulose particles from biomass. In nature, the brown rot fungi utilize iron complex and hydrogen peroxide to overcome the lignin boundary and approach cellulose in biomass. This fact encouraged us to use the same oxidative catalytic system on biomass in a one-pot process. Pinewood sawdust was taken as starting material, and the iron oxide, hydrogen peroxide oxidation system was applied with varying the concentration of hydrogen peroxide and the reaction time. In 6 h, a one-pot system obtained a 95% pure cellulose with an aspect ratio of 1–15 from pinewood sawdust. The yield of the process was very high, i.e., from the total cellulose content in pinewood, 85% of cellulose was recovered in this process. The degree of polymerization of the obtained cellulose product was 100 ± 20, with an aspect ratio of 1–20. The particles were sphere and ellipsoidal-shaped, with the majority having a diameter of 120–180 nm. It had a crystallinity of 81%, an increase of 26% crystallinity was observed compared to the untreated pinewood sawdust. These nanocellulose particles would be a promising material for different derivatizing reactions, with their high surface area (15.0 ± 0.5 m2/g) and low degree of polymerization. This developed one-pot process has avoided using hazardous chemicals to produce nanocellulose particles.
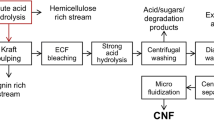
Graphical abstract





Similar content being viewed by others
Availability of data and materials
The data supporting this study's findings are available in this article's supporting information. In addition, other raw experimental data is available on request.
Abbreviations
- NC:
-
Nanocellulose
- Fe2O3/H2O2 :
-
Iron oxide-hydrogen peroxide catalytic system
- BEK cellulose:
-
Bleached eucalyptus kraft cellulose
- RB flask:
-
Round-bottomed flask
- DI water:
-
Deionized water
- NREL:
-
National renewable energy laboratory
- SEM:
-
Scanning electron microscopy
- DP:
-
Degree of polymerization
- CED:
-
Cupriethylenediamine solution
- XRD:
-
X-ray diffraction
References
Agbor VB, Cicek N, Sparling R, Berlin A, Levin DB (2011) Biomass pretreatment: fundamentals toward application. Biotechnol Adv 29:675–685. https://doi.org/10.1016/j.biotechadv.2011.05.005
Alonso-Lerma B, Larraza I, Barandiaran L, Ugarte L, Saralegi A, Corcuera MA, Perez-Jimenez R, Eceiza A (2021) Enzymatically produced cellulose nanocrystals as reinforcement for waterborne polyurethane and its applications. Carbohydr Polym 254:117478. https://doi.org/10.1016/j.carbpol.2020.117478
Arantes V, Jellison J, Goodell B (2012) Peculiarities of brown-rot fungi and biochemical Fenton reaction with regard to their potential as a model for bioprocessing biomass. Appl Microbiol Biotechnol 94:323–338. https://doi.org/10.1007/s00253-012-3954-y
Barana D, Salanti A, Orlandi M, Ali DS, Zoia L (2016) Biorefinery process for the simultaneous recovery of lignin, hemicelluloses, cellulose nanocrystals and silica from rice husk and Arundo donax. Ind Crops Prod 86:31–39. https://doi.org/10.1016/j.indcrop.2016.03.029
Behera S, Arora R, Nandhagopal N, Kumar S (2014) Importance of chemical pretreatment for bioconversion of lignocellulosic biomass. Renew Sust Energy Rev 36:91–106. https://doi.org/10.1016/j.rser.2014.04.047
Carvalho JPF, Silva ACQ, Silvestre AJD, Freire CSR, Vilela C (2021) Spherical cellulose micro and nanoparticles: a review of recent developments and applications. Nanomaterials 11:2744. https://doi.org/10.3390/nano11102744
Chirayil CJ, Mathew L, Thomas S (2014) Review of recent research in nono cellulose preparation from different lignocellulosic fibres. Rev Adv Mater Sci 37:20–28
Coelho AA (2018) TOPAS and TOPAS-academic: an optimization program integrating computer algebra and crystallographic objects written in C++. J Appl Cryst 51:210–218. https://doi.org/10.1107/S1600576718000183
Coelho A, Cheary RW (1992) A fundamental parameters approach to X-ray line-profile fitting. J Appl Cryst 25:109–121. https://doi.org/10.1107/S0021889891010804
Driemeier C, Calligaris GA (2011) Theoretical and experimental developments for accurate determination of crystallinity of cellulose I materials. J Appl Crystallogr 44:184–192. https://doi.org/10.1107/S0021889810043955
Dutra ED, Santos FA, Alencar BRA, Reis ALS, de Souza Rd FR, da Silva Aquino KA, Morais MA Jr, Menezes RSC (2018) Alkaline hydrogen peroxide pretreatment of lignocellulosic biomass: status and perspectives. Biomass Convers Biorefin 8:225–234. https://doi.org/10.1007/s13399-017-0277-3
El-Sakhawy M, Hassan ML (2007) Physical and mechanical properties of microcrystalline cellulose prepared from agricultural residues. Carbohydr Polym 67:1–10. https://doi.org/10.1016/j.carbpol.2006.04.009
Fodil Cherif M, Trache D, Brosse N, Benaliouche F, Tarchoun AF (2020) Comparison of the physicochemical properties and thermal stability of organosolv and kraft lignins from hardwood and softwood biomass for their potential valorization. Waste Biomass Valor 11:6541–6553. https://doi.org/10.1007/s12649-020-00955-0
French AD (2014) Idealized power diffraction patterns for cellulose polymorphs. Cellulose 21:885–896. https://doi.org/10.1007/s10570-013-0030-4
French AD (2020) Increment in evolution of cellulose crystallinity analysis. Cellulose 27:5445–5448. https://doi.org/10.1007/s10570-020-03172-z
French AD, Santiago Cintrón M (2013) Cellulose polymorphy, crystallite size, and the segal crystallinity index. Cellulose 20:583–588. https://doi.org/10.1007/s10570-012-9833-y
Garcia-Segura S, Bellotindos LM, Huang Y-H, Brillas E, Lu MC (2016) Fluidized-bed Fenton process as alternative wastewater treatment technology—a review. J Taiwan Inst Chem Eng. 67:211–225. https://doi.org/10.1016/j.jtice.2016.07.021
Goyal R, Singh O, Agrawal A, Samanta C, Sarkar B (2022) Advantages and limitations of catalytic oxidation with hydrogen peroxide: from bulk chemicals to lab scale process. Catal Rev Sci Eng 64:229–285. https://doi.org/10.1080/01614940.2020.1796190
Guo J, Catchmark JM (2012) Surface area and porosity of acid hydrolyzed cellulose nanowhiskers and cellulose produced by Gluconacetobacter xylinus. Carbohydr Polym 87:1026–1037. https://doi.org/10.1016/j.carbpol.2011.07.060
Hafid HS, Omar FN, Zhu J, Wakisaka M (2021) Enhanced crystallinity and thermal properties of cellulose from rice husk using acid hydrolysis treatment. Carbohydr Polym 260:117789. https://doi.org/10.1016/j.carbpol.2021.117789
Haldar D, Purkait MK (2020) Micro and nanocrystalline cellulose derivatives of lignocellulosic biomass: a review on synthesis, applications and advancements. Carbohydr Polym 250:116937. https://doi.org/10.1016/j.carbpol.2020.116937
Heinze T, El Seoud OA, Koschella A (2018) Cellulose derivatives: synthesis, structure, and properties; Springer, 2018
Isogai A, Saito T, Fukuzumi H (2011) TEMPO-oxidized cellulose nanofibers. Nanoscale 3:71–85. https://doi.org/10.1039/C0NR00583E
Jia Y, Bai Y, Chang J, Zhai Y, Zhang T, Ren K, Hong J (2022) Life cycle assessment of hydrogen peroxide produced from mainstream hydrogen sources in China. J Clean Prod 352:131655. https://doi.org/10.1016/j.jclepro.2022.131655
Jiao Y, Wan C, Qiang T, Li J (2016) Synthesis of superhydrophobic ultralight aerogels from nanofibrillated cellulose isolated from natural reed for high-performance adsorbents. Appl Phys A 122:1–10. https://doi.org/10.1007/s00339-016-0194-5
Kaffashsaie E, Yousefi H, Nishino T, Matsumoto T, Mashkour M, Madhoushi M, Kawaguchi H (2021) Direct conversion of raw wood to tempo-oxidized cellulose nanofibers. Carbohydr Polym 262:117938. https://doi.org/10.1016/j.carbpol.2021.117938
Kamali M, Khodaparast Z (2015) Review on recent developments on pulp and paper mill wastewater treatment. Ecotoxicol Environ Saf 114:326–342. https://doi.org/10.1016/j.ecoenv.2014.05.005
Kohli K, Katuwal S, Biswas A, Sharma BK (2020) Effective delignification of lignocellulosic biomass by microwave assisted deep eutectic solvents. Bioresour Technol 303:122897. https://doi.org/10.1016/j.biortech.2020.122897
Kumar V, Pathak P, Bhardwaj NK (2020) Waste paper: an underutilized but promising source for nanocellulose mining. Waste Manag 102:281–303. https://doi.org/10.1016/j.wasman.2019.10.041
Liimatainen H, Visanko M, Sirvio JA, Hormi OE, Niinimaki J (2012) Enhancement of the nanofibrillation of wood cellulose through sequential periodate–chlorite oxidation. Biomacromolecules 13(5):1592–1597
Macrae CF, Bruno IJ, Chisholm JA, Edgington PR, McCabe P, Pidcock E, Rodriguez-Monge L, Taylor R, van de Streek J, Wood PA (2008) Mercury CSD 2.0-new features for the visualization and investigation of crystal structures. J Appl Crystallogr 41:466–470. https://doi.org/10.1107/S0021889807067908
Mahmud MM, Perveen A, Jahan RA, Matin MA, Wong SY, Li X, Arafat MT (2019) Preparation of different polymorphs of cellulose from different acid hydrolysis medium. Int J Biol Macromol 130:969–976. https://doi.org/10.1016/j.ijbiomac.2019.03.027
Meiland M, Liebert T, Heinze T (2011) Tailoring the degree of polymerization of low molecular weight cellulose. Macromol Mater Eng 296:802–809. https://doi.org/10.1002/mame.201000424
Mishra RK, Mohanty K (2018) Pyrolysis kinetics and thermal behavior of waste sawdust biomass using thermogravimetric analysis. Bioresour Technol 251:63–74. https://doi.org/10.1016/j.biortech.2017.12.029
Nagarajan K, Ramanujam N, Sanjay M, Siengchin S, Surya Rajan B, Sathick Basha K, Madhu P, Raghav GA (2021) Comprehensive review on cellulose nanocrystals and cellulose nanofibers: pretreatment, preparation, and characterization. Polym Compos 42:1588–1630. https://doi.org/10.1002/pc.25929
Nishiyama Y, Langan P, Chanzy H (2002) Crystal structure and hydrogen-bonding system in cellulose 1β from synchrotron X-ray and neutron fibre diffraction. J Am Chem Soc 124(31):9074–9082. https://doi.org/10.1021/ja0257319
Ong HC, Chen W-H, Singh Y, Gan YY, Chen C-Y, Show PL (2020) A state-of-the-art review on thermochemical conversion of biomass for biofuel production: a TG-FTIR approach. Energy Convers Manag 209:112634. https://doi.org/10.1016/j.enconman.2020.112634
Park S, Baker JO, Himmel ME, Parilla PA, Johnson DK (2010) Cellulose crystallinity index: measurement techniques and their impact on interpreting cellulase performance. Biotechnol Biofuels 3:1–10. https://doi.org/10.1186/1754-6834-3-10
Peltola H, Immonen K, Johansson LS, Virkajärvi J, Sandquist D (2019) Influence of pulp bleaching and compatibilizer selection on performance of pulp fiber reinforced PLA biocomposites. J Appl Polym Sci 136:47955. https://doi.org/10.1002/app.47955
Presley GN, Panisko E, Purvine SO, Schilling JS (2018) Coupling secretomics with enzyme activities to compare the temporal processes of wood metabolism among white and brown rot fungi. Appl Environ Microbiol 84:e00159-e218. https://doi.org/10.1128/AEM.00159-18
Scherrer P (1918) Bestimmung der Größe und der inneren Struktur von Kolloidteilchen mittels Röntgenstrahlen: Nachrichten von der Gesellschaft der Wissenschaften zu Göttingen. Mathematisch-Physikalische Klasse, Berlin, pp 98–100. http://eudml.org/doc/59018
Sharma N, Bhardwaj NK, Singh RBP (2020) Environmental issues of pulp bleaching and prospects of peracetic acid pulp bleaching: a Review. J Clean Prod 256:120338. https://doi.org/10.1016/j.jclepro.2020.120338
Sluiter JB, Ruiz RO, Scarlata CJ, Sluiter AD, Templeton DW (2010) Compositional analysis of Lignocellulosic feedstocks. 1. Review and description of methods. J Agric Food Chem 58:9043–9053. https://doi.org/10.1021/jf1008023
Tarchoun AF, Trache D, Klapötke TM, Derradji M, Bessa W (2019) Ecofriendly isolation and characterization of microcrystalline cellulose from giant reed using various acidic media. Cellulose 26:7635–7651. https://doi.org/10.1007/s10570-019-02672-x
Tarchoun AF, Trache D, Klapötke TM, Krumm B, Khimeche K, Mezroua AA (2020) Promising energetic biopolymer based on azide-functionalized microcrystalline cellulose: synthesis and characterization. Carbohydr Polym 249:116820
Trache D, Hussin MH, Haafiz MKM, Thakur VK (2017) Recent progress in cellulose nanocrystals: sources and production. Nanoscale 9(5):1763–1786. https://doi.org/10.1039/C6NR09494E
Vijay P, Batchelor W, Saito K (2022) One-pot treatment of cellulose using iron oxide catalysts to produce nanocellulose and water-soluble oxidized cellulose. Carbohydr Polym 282:119060. https://doi.org/10.1016/j.carbpol.2021.119060
Wada M, Okano T, Sugiyama J (2001) Allomorphs of native crystalline cellulose I evaluated by two equatorial d-spacings. J Wood Sci 47:124–128. https://doi.org/10.1007/BF00780560
Wu L, Tian F, Sun J (2021) On the use of cellulose nanowhisker as reinforcement in all-cellulose composite membrane from corn stalk. J Appl Polym Sci 138:50206. https://doi.org/10.1002/app.50206
Yarbrough JM, Zhang R, Mittal A, Vander Wall T, Bomble YJ, Decker SR, Himmel ME, Ciesielski PN (2017) Multifunctional cellulolytic enzymes outperform processive fungal cellulases for coproduction of nanocellulose and biofuels. ACS Nano 11:3101–3109. https://doi.org/10.1021/acsnano.7b00086
Zimmermann T, Bordeanu N, Strub E (2010) Properties of nanofibrillated cellulose from different raw materials and its reinforcement potential. Carbohydr Polym 79:1086–1093. https://doi.org/10.1016/j.carbpol.2009.10.045
Acknowledgements
The authors gratefully acknowledge the funding of the Australian Research Council (ARC) and Processing Advance Lignocellulosics (PALS) [grant number- IH170100020]. The authors also acknowledge the Monash school of chemistry, Monash X-ray Platform, and Monash Centre for Electron Microscopy to use facilities.
Funding
This work was supported by funding of the Australian Research Council (ARC) and Processing Advance Lignocellulosics (PALS) [grant number- IH170100020].
Author information
Authors and Affiliations
Contributions
All authors contributed to the study's conception and design. Material preparation, data collection and analysis were performed by PV, VR, JM. The first draft of the manuscript was written by PV and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Ethics approval
There was no ethical content in preparing this manuscript.
Consent to participate
All authors consented to participate in this work.
Consent for publication
All authors consented to publish this work.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Vijay, P., Raghuwanshi, V.S., Ma, J. et al. Fenton-like oxidation of pinewood to produce cellulose nanoparticles in one pot treatment. Cellulose 31, 953–967 (2024). https://doi.org/10.1007/s10570-023-05573-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-023-05573-2