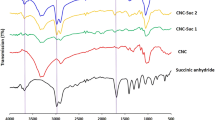
Abstract
In this work, the carboxylate-functionalized cellulose (CFC) from waste cotton fabrics (WCF) was studied for removal of cationic dyes from aqueous solutions. Instead of using conventional process for esterifying cellulose, which uses sulfuric or perchloric acid as catalysts, and methylene chloride as solvent, a novel mechanochemical approach which requires no catalyst or solvent in the reaction mixture was used. The prepared CFC with high concentrations of carboxyl groups ranging from 1.34 to 2.14 mmol/g was tested as adsorbent for the removal of methylene blue (MB) and rhodamine 6G (R6G) from aqueous solutions. The modified cellulose showed much higher adsorption capacity than that of unmodified WCF. The effect of operating variables such as concentration of carboxyl groups, solution pH, adsorbent dosage, temperature, and initial dye concentration on the adsorption capacity were investigated. The maximum adsorption capacity (185.63 mg/g for MB and 118.21 mg/g for R6G) was obtained at pH = 10 and temperature of 298 K. The experimental equilibrium data were analyzed by using Langmuir and Freundlich isotherm models and the results indicated that the adsorption behavior of both dyes could be described well by Langmuir isotherm. The value of standard enthalpy changes was −26.48 kJ/mol for MB and −42.16 kJ/mol for R6G, suggesting that the adsorption process is exothermic in nature. The results revealed that solvent-free mechanochemistry is an environmentally friendly method to prepare novel efficient absorbent for the removal of dyes from wastewater.
Graphical Abstract








Similar content being viewed by others
References
Al-Degs YS, El-Barghouthi MI, Issa AA, Khraisheh MA, Walker GM (2006) Sorption of Zn (II), Pb(II), and Co (II) using natural sorbents: equilibrium and kinetic studies. Water Res 40:2645–2658
Annadurai G, Juang R, Lee D (2002) Use of cellulose-based wastes for adsorption of dyes from aqueous solutions. J Hazard Mater B92:263–274
Biswas A, Selling G, Appell M, Woods KK, Willett JL, Buchanan CM (2007) Iodine catalyzed esterification of cellulose using reduced levels of solvent. Carbohydr Polym 68:555–560
Bulut Y, Aydin HA (2006) Kinetics and thermodynamics study of methylene blue adsorption on wheat shells. Desalination 194:259–267
Chao AC, Shyu SS, Lin YC, Mi FL (2004) Enzymatic grafting of carboxyl groups on to chitosan—to confer on chitosan the property of a cationic dye adsorbent. Bioresour Technol 91:157–162
De DS, Basu JK (1998) Adsorption of methylene blue on to a low cost adsorbent developed from sawdust. Ind J Environ Protec 19:416–421
Demirbas A (2009) Agricultural based activated carbons for the removal of dyes from aqueous solutions: a review. J Hazard Mater 167:1–9
Deniz F, Saygideger SD (2011) Removal of a hazardous azo dye (Basic Red 46) from aqueous solution by princess tree leaf. Desalination 268:6–11
Fernandes AN, Almeida CAP, Debacher NA, Sierra MMDS (2010) Isotherm and thermodynamic data of adsorption of methylene blue from aqueous solution onto peat. J Mol Struct 982:62–65
French AD (2013) Idealized powder diffraction patterns for cellulose polymorphs. Cellulose. doi:10.1007/s10570-013-0030-4
French AD, Santiago Cintrón M (2013) Cellulose polymorphy, crystallite size, and the Segal crystallinity index. Cellulose 20:583–588
Freundlich H (1936) Adsorptionstechnik. J Phys Chem 40:857–858
Giménez-Martín E, López-Andrade M, Ontiveros-Ortega A, Espinosa-Jiménez M (2009) Adsorption of chlorhexidine onto cellulosic fibers. Cellulose 16:467–479
Gong R, Sun Y, Chen J, Liu H, Yang C (2005) Effect of chemical modification on dye adsorption capacity of peanut hull. Dyes Pigm 67:175–181
Gu X, Yang CQ (1998) FT-IR and FT-raman spectroscopy study of the cyclic anhydride intermediates for esterification of cellulose: I. Formation of anhydrides without a catalyst. Res Chem Intermed 24:979–996
Gurgel LVA, Júnior OK, Gil RPDF, Gil LF (2008) Adsorption of Cu(II), Cd(II), and Pb(II) from aqueous single metal solutions by cellulose and mercerized cellulose chemically modified with succinic anhydride. Bioresour Technol 99:3077–3083
Hameed BH, Ahmad AA (2009) Batch adsorption of methylene blue from aqueous solution by garlic peel, an agricultural waste biomass. J Hazard Mater 164:870–875
Janaki V, Vijayaraghavan K, Oh BT, Ramasamy AK, Kamala-Kannan S (2013) Synthesis, characterization and application of cellulose/polyaniline nanocomposite for the treatment of simulated textile effluent. Cellulose. doi:10.1007/s10570-013-9910-x
Jayakumar R, Balaji R, Nanjundan S (2000) Studies on copolymers of 2-(Nphthalimido)ethyl methacrylate with methyl methacrylate. Eur Polym J 36:1659–1666
Júnior OK, Gurgel LVA, Melo JCP, Botaro VR, Melo TMS, Gil RPDF, Gil LF (2007) Adsorption of heavy metal ion from aqueous single metal solution by chemically modified sugarcane bagasse. Bioresour Technol 98:1291–1297
Khan AA, Singh RP (1987) Adsorption thermodynamics of carbofuran on Sn (IV) arsenosilicate in H+ , Na+ and Ca2+ forms. Colloids Surf 24:33–42
Langmuir I (1918) The adsorption of gases on plan surfaces of glass, mica and platinum. J Am Chem Soc 40:1361–1403
Li WY, Jin AX, Liu CF, Sun RC, Zhang AP, Kennedy JF (2009) Homogeneous modification of cellulose with succinic anhydride in ionic liquid using 4-dimethylaminopyridine as a catalyst. Carbohyd Polym 78:389–395
Mane VS, Mall ID, Srivastava VC (2007) Use of bagasse fly ash as an adsorbent for the removal of brilliant green dye from aqueous solution. Dyes Pigm 73:269–278
Mao YH, Guan Y, Zheng QK, Feng XN, Wang XX (2011) Adsorption thermodynamic and kinetic of disperse dye on cotton fiber modified with tolylene diisocyanate derivative. Cellulose 18:271–279
Miranda R, Sosa-Blanco C, Bustos-Martinez D, Vasile C (2007) Pyrolysis oftextile wastes: I. Kinetics and yields. J Anal Appl Pyrol 80:489–495
Mui ELK, Cheung WH, Valix M, McKay G (2010) Dye adsorption onto char from bamboo. J Hazard Mater 177:1001–1005
Nandi BK, Goswami A, Purkait MK (2009) Removal of cationic dyes from aqueous solutions by kaolin: kinetic and equilibrium studies. Appl Clay Sci 42:583–590
Nishiyama Y, Langan P, Chanzy H (2002) Crystal structure and hydrogen-bonding system in cellulose Iβ from synchrotron X-ray and neutronriber diffraction. J Am Chem Soc 124:9074–9082
Ponnusami V, Vikram S, Srivastava SN (2008) Guava (Psidium guajava) leaf powder: novel adsorbent for removal of methylene blue from aqueous solutions. J Hazard Mater 152:276–286
Rafatullaha M, Sulaimana O, Hashima R, Ahmad A (2010) Adsorption of methylene blue on low-cost adsorbents: a review. J Hazard Mater 177:70–80
Saliba R, Gauthier H, Gauthier R, Petit-Ramel M (2002) The use of amidoximated cellulose for the removal of metal ions and dyes from waste waters. Cellulose 9:183–191
Salleh MAM, Mahmouda DK, Karima WAWA, Idris A (2011) Cationic and anionic dye adsorption by agricultural solid wastes: a comprehensive review. Desalination 280:1–13
Segal L, Creely JJ, Martin AE, Conrad CM (1959) An empirical method for estimating the degree of crystallinity of native cellulose using the X-ray diffractometer. Textile Res J 29:786–794
Sun X, Lu C, Zhang W, Tian D, Zhang X (2013) Acetone-soluble cellulose acetate extracted from waste blended fabrics via ionic liquid catalyzed acetylation. Carbohydr Polym 98:405–411
Sun X, Lu C, Liu Y, Zhang W, Zhang X (2014) Melt-processed poly (vinyl alcohol) composites filled with microcrystalline cellulose from waste cotton fabrics. Carbohydr Polym 101:642–649
Uddin MT, Islam MA, Mahmud S, Rukanuzzaman M (2009) Adsorptive removal of methylene blue by tea waste. J Hazard Mater 164:53–60
Vadivelan V, Kumar KV (2005) Equilibrium, kinetics, mechanism, and process design for the sorption of methylene blue onto rice husk. J Colloid Interf Sci 286:90–100
Wang M, Zhang X, Zhang W, Tian D, Lu C (2013) Thermoplastic polyurethane composites prepared from mechanochemically activated waste cotton fabric and reclaimed polyurethane foam. J Appl Polym Sci 128:3555–3563
Xiong R, Zhang X, Tian D, Zhou Z, Lu C (2012) Comparing microcrystalline with spherical nanocrystalline cellulose from waste cotton fabrics. Cellulose 19:1189–1198
Xu X, Gao BY, Yue QY, Zhong QQ (2010) Preparation and utilization of wheat straw bearing amine groups for the sorption of acid and reactive dyes from aqueous solutions. J Hazard Mater 182:1–9
Yang F, Bai X, Xu B, Guo H (2013) Triphenylene-modified chitosan: novel high efficient sorbent for cationic and anionic dyes. Cellulose 20:895–906
Zhang W, Li C, Liang M, Geng Y, Lu C (2010a) Preparation of carboxylate-functionalized cellulose via solvent-free mechanochemistry and its characterization as a biosorbent for removal of Pb2+ from aqueous solution. J Hazard Mater 181:468–473
Zhang Z, Zhang Z, Fernández Y, Menéndez JA, Niu H, Peng J, Guo S (2010b) Adsorption isotherms and kinetics of methylene blue on a low-cost adsorbent recovered from a spent catalyst of vinyl acetate synthesis. Appl Surf Sci 256:2569–2576
Zhang X, Lu C, Zheng Q, Liang M (2011) An environmental friendly approach for recycling of post-vulcanized fluoroelastomer scraps through high-shear mechanical milling. Polym Advan Technol 22:2104–2109
Zhang X, Zhang W, Tian D, Zhou Z, Lu C (2013a) A new application of ionic liquids for heterogeneously catalyzed acetylation of cellulose under solvent-free conditions. RSC Adv 3:7722–7725
Zhang X, Tian D, Zhang W, Zhu J, Lu C (2013b) Morphology, foaming rheology and physical properties of ethylene-propylene diene rubber/ground tyre rubber (GTR) composite foams: effect of mechanochemical devulcanisation of GTR. Prog Rubber Plast Re 29:81–98
Acknowledgments
The authors would like to thank the National Science Foundation of China (51203105) and National High Technology Research and Development Program (863 Program, SS2012AA062613) for financial support.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Tian, D., Zhang, X., Lu, C. et al. Solvent-free synthesis of carboxylate-functionalized cellulose from waste cotton fabrics for the removal of cationic dyes from aqueous solutions. Cellulose 21, 473–484 (2014). https://doi.org/10.1007/s10570-013-0112-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-013-0112-3