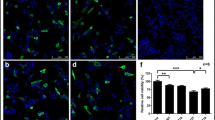
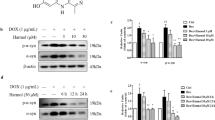
Abstract
Alpha-synuclein (α-syn) is associated to Parkinson’s disease (PD). The aggregated form of α-syn has potential neurotoxicity. Thus, the clearance of α-syn aggregation is a plausible strategy to delay disease progression of PD. In our study, we found that the treatment of Ginkgolide B (GB) and Ginkgolide K (GK) reduced cell death, and enhanced cell proliferation in SH-SY5Y cells, which overexpressed A53T mutant α-syn. Surprisingly, GK, but not GB, promoted the clearance of A53T α-syn, which can be abolished by autophagy inhibitor 3-methyladenine, indicating that GK-induced autophagy intervened in the clearance of A53T α-syn. However, GK did not affect the NEDD4 that belongs to the ubiquitin ligase in the endosomal–lysosomal pathway. Furthermore, GK treatment inhibited the p-NF-kB/p65 and induced the PI3K, BDNF, and PSD-95. Taken together, GK increased the clearance of α-syn, reduced cell death, and triggered complex crosstalk between different signaling pathways. Although our results show a potentially new therapeutic candidate for PD, the details of this mechanism need to be further identified.




Similar content being viewed by others
References
Autry AE, Monteggia LM. Brain-derived neurotrophic factor and neuropsychiatric disorders. Pharmacol Rev. 2012;64:238–58. https://doi.org/10.1124/pr.111.005108.
Bisaglia M, Mammi S, Bubacco L. Structural insights on physiological functions and pathological effects of alpha-synuclein. FASEB J. 2009;23:329–40. https://doi.org/10.1096/fj.08-119784.
Bonini NM, Giasson BI. Snaring the function of alpha-synuclein. Cell. 2005;123:359–61. https://doi.org/10.1016/j.cell.2005.10.017.
Bramham CR, Messaoudi E. BDNF function in adult synaptic plasticity: the synaptic consolidation hypothesis. Prog Neurobiol. 2005;76:99–125. https://doi.org/10.1016/j.pneurobio.2005.06.003.
Burre J, Sharma M, Sudhof TC. Alpha-synuclein assembles into higher-order multimers upon membrane binding to promote SNARE complex formation. Proc Natl Acad Sci U S A. 2014;111:E4274–83. https://doi.org/10.1073/pnas.1416598111.
Busch DJ, Oliphint PA, Walsh RB, Banks SM, Woods WS, George JM, et al. Acute increase of alpha-synuclein inhibits synaptic vesicle recycling evoked during intense stimulation. Mol Biol Cell. 2014;25:3926–41. https://doi.org/10.1091/mbc.E14-02-0708.
Chartier-Harlin MC, et al. Alpha-synuclein locus duplication as a cause of familial Parkinson’s disease. Lancet (London, England). 2004;364:1167–9. https://doi.org/10.1016/s0140-6736(04)17103-1.
Chen A, Xiong LJ, Tong Y, Mao M. Neuroprotective effect of brain-derived neurotrophic factor mediated by autophagy through the PI3K/Akt/mTOR pathway. Mol Med Rep. 2013;8:1011–6. https://doi.org/10.3892/mmr.2013.1628.
Clark J, Clore EL, Zheng K, Adame A, Masliah E, Simon DK. Oral N-acetyl-cysteine attenuates loss of dopaminergic terminals in alpha-synuclein overexpressing mice. PLoS One. 2010;5:e12333. https://doi.org/10.1371/journal.pone.0012333.
Cuervo AM, Stefanis L, Fredenburg R, Lansbury PT, Sulzer D. Impaired degradation of mutant alpha-synuclein by chaperone-mediated autophagy. Science (New York, NY). 2004;305:1292–5. https://doi.org/10.1126/science.1101738.
Dijkhuizen PA, Ghosh A. BDNF regulates primary dendrite formation in cortical neurons via the PI3-kinase and MAP kinase signaling pathways. J Neurobiol. 2005;62:278–88. https://doi.org/10.1002/neu.20100.
Ebrahimi-Fakhari D, et al. Distinct roles in vivo for the ubiquitin-proteasome system and the autophagy-lysosomal pathway in the degradation of alpha-synuclein. J Neurosci. 2011;31:14508–20. https://doi.org/10.1523/jneurosci.1560-11.2011.
Emamzadeh FN. Alpha-synuclein structure, functions, and interactions. J Res Med Sci. 2016;21:29. https://doi.org/10.4103/1735-1995.181989.
Feany MB, Bender WW. A Drosophila model of Parkinson’s disease. Nature. 2000;404:394–8. https://doi.org/10.1038/35006074.
Gasparini L, Netzer WJ, Greengard P, Xu H. Does insulin dysfunction play a role in Alzheimer’s disease? Trends Pharmacol Sci. 2002;23:288–93.
George S, Brundin P. Immunotherapy in Parkinson’s disease: micromanaging alpha-synuclein aggregation. J Parkinson's Dis. 2015;5:413–24. https://doi.org/10.3233/jpd-150630.
Hernandez-Vargas R, Fonseca-Ornelas L, Lopez-Gonzalez I, Riesgo-Escovar J, Zurita M, Reynaud E. Synphilin suppresses alpha-synuclein neurotoxicity in a Parkinson's disease Drosophila model. Genesis (New York, NY: 2000). 2011;49:392–402. https://doi.org/10.1002/dvg.20740.
Kanei-Ishii C, Nomura T, Tanikawa J, Ichikawa-Iwata E, Ishii S. Differential sensitivity of v-Myb and c-Myb to Wnt-1-induced protein degradation. J Biol Chem. 2004;279:44582–9. https://doi.org/10.1074/jbc.M407831200.
Kirik D, et al. Parkinson-like neurodegeneration induced by targeted overexpression of alpha-synuclein in the nigrostriatal system. J Neurosci. 2002;22:2780–91.
Kruger R, et al. Ala30Pro mutation in the gene encoding alpha-synuclein in Parkinson’s disease. Nat Genet. 1998;18:106–8. https://doi.org/10.1038/ng0298-106.
Lamberto GR, Binolfi A, Orcellet ML, Bertoncini CW, Zweckstetter M, Griesinger C, et al. Structural and mechanistic basis behind the inhibitory interaction of PcTS on alpha-synuclein amyloid fibril formation. Proc Natl Acad Sci U S A. 2009;106:21057–62. https://doi.org/10.1073/pnas.0902603106.
Lan DM, et al. Effect of trehalose on PC12 cells overexpressing wild-type or A53T mutant alpha-synuclein. Neurochem Res. 2012;37:2025–32. https://doi.org/10.1007/s11064-012-0823-0.
Lashuel HA, Overk CR, Oueslati A, Masliah E. The many faces of alpha-synuclein: from structure and toxicity to therapeutic target. Nat Rev Neurosci. 2013;14:38–48. https://doi.org/10.1038/nrn3406.
Lee HJ, Khoshaghideh F, Lee S, Lee SJ. Impairment of microtubule-dependent trafficking by overexpression of alpha-synuclein. Eur J Neurosci. 2006;24:3153–62. https://doi.org/10.1111/j.1460-9568.2006.05210.x.
Liu FT, et al. Involvement of mortalin/GRP75/mthsp70 in the mitochondrial impairments induced by A53T mutant alpha-synuclein. Brain Res. 2015;1604:52–61. https://doi.org/10.1016/j.brainres.2015.01.050.
Liu FT, et al. Fasudil, a Rho kinase inhibitor, promotes the autophagic degradation of A53T alpha-synuclein by activating the JNK 1/Bcl-2/beclin 1 pathway. Brain Res. 2016;1632:9–18. https://doi.org/10.1016/j.brainres.2015.12.002.
Ma S, et al. Neuroprotective effect of ginkgolide K on glutamate-induced cytotoxicity in PC 12 cells via inhibition of ROS generation and Ca(2+) influx. Neurotoxicology. 2012a;33:59–69. https://doi.org/10.1016/j.neuro.2011.11.003.
Ma S, Yin H, Chen L, Liu H, Zhao M, Zhang X. Neuroprotective effect of Ginkgolide K against acute ischemic stroke on middle cerebral ischemia occlusion in rats. J Nat Med. 2012b;66:25–31. https://doi.org/10.1007/s11418-011-0545-7.
Ma S, Liu X, Xun Q, Zhang X. Neuroprotective effect of Ginkgolide K against H2O2-induced PC12 cell cytotoxicity by ameliorating mitochondrial dysfunction and oxidative stress. Biol Pharm Bull. 2014;37:217–25.
Masliah E, et al. Dopaminergic loss and inclusion body formation in alpha-synuclein mice: implications for neurodegenerative disorders. Science (New York, NY). 2000;287:1265–9.
Mazzulli JR, Zunke F, Tsunemi T. Activation of beta-glucocerebrosidase reduces pathological alpha-synuclein and restores lysosomal function in Parkinson’s patient midbrain neurons. J Neurosci. 2016;36:7693–706. https://doi.org/10.1523/jneurosci.0628-16.2016.
Mizuno M, Yamada K, Olariu A, Nawa H, Nabeshima T. Involvement of brain-derived neurotrophic factor in spatial memory formation and maintenance in a radial arm maze test in rats. J Neurosci. 2000;20:7116–21.
Mizushima N, Yoshimori T, Levine B. Methods in mammalian autophagy research. Cell. 2010;140:313–26. https://doi.org/10.1016/j.cell.2010.01.028.
Murer MG, Yan Q, Raisman-Vozari R. Brain-derived neurotrophic factor in the control human brain, and in Alzheimer’s disease and Parkinson's disease. Prog Neurobiol. 2001;63:71–124.
Murphy DD, Rueter SM, Trojanowski JQ, Lee VM. Synucleins are developmentally expressed, and alpha-synuclein regulates the size of the presynaptic vesicular pool in primary hippocampal neurons. J Neurosci. 2000;20:3214–20.
Olanow CW, McNaught K. Parkinson’s disease, proteins, and prions: milestones. Mov Disord. 2011;26:1056–71. https://doi.org/10.1002/mds.23767.
Olzmann JA, Li L, Chin LS. Aggresome formation and neurodegenerative diseases: therapeutic implications. Curr Med Chem. 2008;15:47–60.
Outeiro TF, Lindquist S. Yeast cells provide insight into alpha-synuclein biology and pathobiology. Science (New York, NY). 2003;302:1772–5. https://doi.org/10.1126/science.1090439.
Polymeropoulos MH, et al. Mutation in the alpha-synuclein gene identified in families with Parkinson’s disease. Science (New York, NY). 1997;276:2045–7.
Postuma RB, Aarsland D, Barone P, Burn DJ, Hawkes CH, Oertel W, et al. Identifying prodromal Parkinson’s disease: pre-motor disorders in Parkinson’s disease. Mov Disord. 2012;27:617–26. https://doi.org/10.1002/mds.24996.
Prabhakaran K, Chapman GD, Gunasekar PG. Alpha-Synuclein overexpression enhances manganese-induced neurotoxicity through the NF-kappaB-mediated pathway. Toxicol Mech Methods. 2011;21:435–43. https://doi.org/10.3109/15376516.2011.560210.
Rannikko EH, Weber SS, Kahle PJ. Exogenous alpha-synuclein induces toll-like receptor 4 dependent inflammatory responses in astrocytes. BMC Neurosci. 2015;16:57. https://doi.org/10.1186/s12868-015-0192-0.
Rothaug M, et al. LIMP-2 expression is critical for beta-glucocerebrosidase activity and alpha-synuclein clearance. Proc Natl Acad Sci U S A. 2014;111:15573–8. https://doi.org/10.1073/pnas.1405700111.
Sawamura M, Uemura N, Takahashi R. Innate immunity regulates alpha-synuclein clearance. Mov Disord. 2016;31:638. https://doi.org/10.1002/mds.26571.
Schinder AF, Poo M. The neurotrophin hypothesis for synaptic plasticity. Trends Neurosci. 2000;23:639–45.
Shen DN, Zhang LH, Wei EQ, Yang Y. Autophagy in synaptic development, function, and pathology. Neurosci Bull. 2015;31:416–26. https://doi.org/10.1007/s12264-015-1536-6.
Song JX, et al. HMGB1 is involved in autophagy inhibition caused by SNCA/alpha-synuclein overexpression: a process modulated by the natural autophagy inducer corynoxine B. Autophagy. 2014;10:144–54. https://doi.org/10.4161/auto.26751.
Spillantini MG, Schmidt ML, Lee VM, Trojanowski JQ, Jakes R, Goedert M. Alpha-synuclein in Lewy bodies. Nature. 1997;388:839–40. https://doi.org/10.1038/42166.
Surguchov A. Molecular and cellular biology of synucleins. Int Rev Cell Mol Biol. 2008;270:225–317. https://doi.org/10.1016/s1937-6448(08)01406-8.
Surguchov A. Intracellular dynamics of synucleins: “here, there and everywhere”. Int Rev Cell Mol Biol. 2015;320:103–69. https://doi.org/10.1016/bs.ircmb.2015.07.007.
Tao W, et al. Liquiritigenin reverses depression-like behavior in unpredictable chronic mild stress-induced mice by regulating PI3K/Akt/mTOR mediated BDNF/TrkB pathway. Behav Brain Res. 2016;308:177–86. https://doi.org/10.1016/j.bbr.2016.04.039.
Tofaris GK, Kim HT, Hourez R, Jung JW, Kim KP, Goldberg AL. Ubiquitin ligase Nedd4 promotes alpha-synuclein degradation by the endosomal-lysosomal pathway. Proc Natl Acad Sci U S A. 2011;108:17004–9. https://doi.org/10.1073/pnas.1109356108.
Vilar E, Salazar R, Perez-Garcia J, Cortes J, Oberg K, Tabernero J. Chemotherapy and role of the proliferation marker Ki-67 in digestive neuroendocrine tumors. Endocr Relat Cancer. 2007;14:221–32. https://doi.org/10.1677/erc-06-0074.
Wang S, et al. Ginkgolide K protects the heart against endoplasmic reticulum stress injury by activating the inositol-requiring enzyme 1alpha/X box-binding protein-1 pathway. Br J Pharmacol. 2016;173:2402–18. https://doi.org/10.1111/bph.13516.
Xiong R, Zhou W, Siegel D, Kitson RR, Freed CR, Moody CJ, et al. A novel Hsp90 inhibitor activates compensatory heat shock protein responses and autophagy and alleviates mutant A53T alpha-synuclein toxicity. Mol Pharmacol. 2015;88:1045–54. https://doi.org/10.1124/mol.115.101451.
Yoshii A, Constantine-Paton M. BDNF induces transport of PSD-95 to dendrites through PI3K-AKT signaling after NMDA receptor activation. Nat Neurosci. 2007;10:702–11. https://doi.org/10.1038/nn1903.
Yoshii A, Murata Y, Kim J, Zhang C, Shokat KM, Constantine-Paton M. TrkB and protein kinase Mzeta regulate synaptic localization of PSD-95 in developing cortex. J Neurosci. 2011;31:11894–904. https://doi.org/10.1523/jneurosci.2190-11.2011.
Funding
This work was supported by grants from the National Natural Science Foundation of China (No. 81371414) and National Major Scientific and Technological Special Project for significant new drug development (2013ZX09402203).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yu, W., Chen, S., Cao, L. et al. Ginkgolide K promotes the clearance of A53T mutation alpha-synuclein in SH-SY5Y cells. Cell Biol Toxicol 34, 291–303 (2018). https://doi.org/10.1007/s10565-017-9419-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10565-017-9419-4