Abstract
Purpose
The Breast Cancer Surveillance Consortium (BCSC) model is a widely used risk model that predicts 5- and 10-year risk of developing invasive breast cancer for healthy women aged 35–74 years. Women with high BCSC risk may also be at elevated risk to develop interval cancers, which present symptomatically in the year following a normal screening mammogram. We examined the association between high BCSC risk (defined as the top 2.5% by age) and breast cancers presenting as interval cancers.
Methods
We conducted a case-case analysis among women with breast cancer in which we compared the mode of detection and tumor characteristics of patients in the top 2.5% BCSC risk by age with age-matched (1:2) patients in the lower 97.5% risk. We constructed logistic regression models to estimate the odds ratio (OR) of presenting with interval cancers, and poor prognosis tumor features, between women from the top 2.5% and bottom 97.5% of BCSC risk.
Results
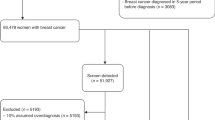
Our analysis included 113 breast cancer patients in the top 2.5% of risk for their age and 226 breast cancer patients in the lower 97.5% of risk. High-risk patients were more likely to have presented with an interval cancer within one year of a normal screening, OR 6.62 (95% CI 3.28–13.4, p < 0.001). These interval cancers were also more likely to be larger, node positive, and higher stage than the screen-detected cancers.
Conclusion
Breast cancer patients in the top 2.5% of BCSC risk for their age were more likely to present with interval cancers. The BCSC model could be used to identify healthy women who may benefit from intensified screening.

Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Code availability
Code used in this analysis will be made available from the corresponding author on reasonable request.
References
Kirsh VA, Chiarelli AM, Edwards SA, O’Malley FP, Shumak RS, Yaffe MJ, Boyd NF (2011) Tumor characteristics associated with mammographic detection of breast cancer in the Ontario breast screening program. J Natl Cancer Inst 103:942–950. https://doi.org/10.1093/jnci/djr138
Holm J, Humphreys K, Li J, Ploner A, Cheddad A, Eriksson M, Törnberg S, Hall P, Czene K (2015) Risk factors and tumor characteristics of interval cancers by mammographic density. J Clin Oncol. https://doi.org/10.1200/JCO.2014.58.9986
Henderson LM, Miglioretti DL, Kerlikowske K, Wernli KJ, Sprague BL, Lehman CD (2015) Breast cancer characteristics associated with digital versus film-screen mammography for screen-detected and interval cancers. AJR Am J Roentgenol 205:676–684. https://doi.org/10.2214/AJR.14.13904
PAM50 and risk of recurrence scores for interval breast cancers (2018). https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5984721/. Accessed 18 Sept 2019
Tice JA, Miglioretti DL, Li C-S, Vachon CM, Gard CC, Kerlikowske K (2015) Breast density and benign breast disease: risk assessment to identify women at high risk of breast cancer. J Clin Oncol 33:3137–3143. https://doi.org/10.1200/JCO.2015.60.8869
Sickles EA, Drsi CJ, Bassett LW, ACR BI-RADS Atlas (2013) Breast imaging reporting and data system. American College of Radiology, Reston
Kerlikowske K, Zhu W, Tosteson ANA, Sprague BL, Tice JA, Lehman CD, Miglioretti DL (2015) Identifying women with dense breasts at highi risk of interval cancers. Ann Intern Med 162:673–681. https://doi.org/10.7326/M14-1465
Long-term outcome in young women with breast cancer: a population-based study (2016). https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5050247/. Accessed 18 Sept 2019
Young adult breast cancer patients have a poor prognosis independent of prognostic clinicopathological factors: a study from the Japanese Breast Cancer Registry (2016). https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5050233/. Accessed 18 Sept 2019
Huilgol YS, Keane H, Shieh Y, Tice JA, Ziv E, Madlensky L, Sabacan L, Acerbi I, Che M, Fiscalini AS, Anton-Culver H, Borowsky A, Hunt S, Naiem A, Parker B, Van’t Veer LJ (2019) Athena Investigators and Advocate Partners, Esserman LJ. Breast cancer risk thresholds as a predictor of chemoprevention uptake in the athena breast health network. In: San Antonio Breast Cancer Symposium (SABCS).
Elson SL, Hiatt RA, Anton-Culver H, Howell LP, Naeim A, Parker BA, Van’t Veer LJ, Hogarth M, Pierce JP, Duwors RJ, Hajopoulos K, Esserman LJ, Athena Breast Health Network (2013) The Athena breast health network: developing a rapid learning system in breast cancer prevention, screening, treatment, and care. Breast Cancer Res Treat 140:417–425. https://doi.org/10.1007/s10549-013-2612-0
Wong EC, Kaplan CP, Dreher N, Hwang J, Van’t Veer L, Melisko ME (2018) Integration of Health Questionnaire Systems to facilitate supportive care services for patients at an academic breast care center. JCO Clin Cancer Inform 2:1–13. https://doi.org/10.1200/CCI.18.00018
Lokate M, Stellato RK, Veldhuis WB, Peeters PHM, van Gils CH (2013) Age-related changes in mammographic density and breast cancer risk. Am J Epidemiol 178:101–109. https://doi.org/10.1093/aje/kws446
Risk Factors Dataset: BCSC (2016). https://www.bcsc-research.org/data/rf. Accessed 18 Sept 2019
Chien AJ, Duralde E, Hwang R, Tsung K, Kao C-N, Rugo HS, Melisko ME, Esserman LJ, Munster PN, Cedars M, Kerlikowske K, McCulloch CE, Rosen MP (2015) Association of tamoxifen use and ovarian function in patients with invasive or pre-invasive breast cancer. Breast Cancer Res Treat 153:173–181. https://doi.org/10.1007/s10549-015-3511-3
Engmann NJ, Scott CG, Jensen MR, Winham S, Miglioretti DL, Ma L, Brandt K, Mahmoudzadeh A, Whaley DH, Hruska C, Wu F, Norman AD, Hiatt RA, Heine J, Shepherd J, Pankratz VS, Vachon CM, Kerlikowske K (2019) Combined effect of volumetric breast density and body mass index on breast cancer risk. Breast Cancer Res Treat 177:165–173. https://doi.org/10.1007/s10549-019-05283-z
Sundquist M, Thorstenson S, Brudin L, Wingren S, Nordenskjöld B (2002) Incidence and prognosis in early onset breast cancer. Breast 11:30–35. https://doi.org/10.1054/brst.2001.0358
Syed BM, Green AR, Paish EC, Soria D, Garibaldi J, Morgan L, Morgan DAL, Ellis IO, Cheung KL (2013) Biology of primary breast cancer in older women treated by surgery: with correlation with long-term clinical outcome and comparison with their younger counterparts. Br J Cancer 108:1042–1051. https://doi.org/10.1038/bjc.2012.601
Shieh Y, Eklund M, Madlensky L, Sawyer SD, Thompson CK, Stover Fiscalini A, Ziv E, Van’t Veer LJ, Esserman LJ, Tice JA, Athena Breast Health Network Investigators (2017) Breast cancer screening in the precision medicine era: risk-based screening in a population-based trial. J Natl Cancer Inst. https://doi.org/10.1093/jnci/djw290
Esserman LJ (2017) The WISDOM Study: breaking the deadlock in the breast cancer screening debate. NPJ Breast Cancer 3:1–7. https://doi.org/10.1038/s41523-017-0035-5
Ozanne EM, Esserman LJ (2004) Evaluation of breast cancer risk assessment techniques: a cost-effectiveness analysis. Cancer Epidemiol Biomark Prev Publ Am Assoc Cancer Res Cosponsored Am Soc Prev Oncol 13:2043–2052
Kerlikowske K, Sprague BL, Tosteson ANA, Wernli KJ, Rauscher GH, Johnson D, Buist DSM, Onega T, Henderson LM, O’Meara ES, Miglioretti DL (2019) Strategies to identify women at high risk of advanced breast cancer during routine screening for discussion of supplemental imaging. JAMA Intern Med. https://doi.org/10.1001/jamainternmed.2019.1758
Acknowledgements
We are extremely grateful to Karla Kerlikowske and her team at the San Francisco Mammography Registry (SFMR) for their guidance contextualizing this research and their willingness to collaborate. The SFMR provided access to data that were not ultimately used in this study. We would also like to thank Ann Griffin from the UCSF Cancer Registry and Patrick Wang from the UCSF Breast Care Center Internship Program. Data collection and sharing was supported by the National Cancer Institute-funded BCSC (HHSN261201100031C). You can learn more about the BCSC at: http://www.bcsc-research.org/. Yiwey Shieh was supported by funding from the National Cancer Institute (1K08CA237829) and the MCL consortium. Dr. Esserman is supported by funding from the NCI MCL consortium (U01CA196406). We would also like to thank the dedicated Athena investigators and advocates for their continued work and support.
Funding
Yiwey Shieh was supported by funding from the National Cancer Institute (1K08CA237829) and the MCL consortium. Laura Esserman is supported by funding from the NCI MCL consortium (U01CA196406).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by ND, MM, and EH. The first draft of the manuscript was written by ND and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare no potential conflicts of interest.
Ethical approval
This work was approved by the UCSF Institutional Review Board and the study was performed in accordance with the ethical standards as laid down in the 1964 Declaration of Helsinki and its later amendments.
Consent to participate
All participants consented to have their data used for research that may result in publication.
Consent for publication
All participants consented to have their data used for research that may result in publication.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Dreher, N., Matthys, M., Hadeler, E. et al. A case-case analysis of women with breast cancer: predictors of interval vs screen-detected cancer. Breast Cancer Res Treat 191, 623–629 (2022). https://doi.org/10.1007/s10549-021-06451-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-021-06451-w