Abstract
Objectives
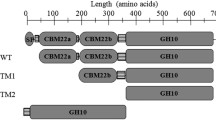
To develop a novel multi-catalytic domain (CD) xylanase Xyn2083 from Clostridium clariflavum by expression of its truncated forms in Escherichia coli and cooperation of xylanase with cellulase in the hydrolysis of waste lignocellulosic resources.
Results
Xyn2083 has two glycoside hydrolase family (GH) domains GH11 and GH10. These two catalytic domains functioned synergistically in xylan hydrolysis. The recombinant protein with GH11 domain, Xyn2083GH11, had the highest xylanase activity among three constructed truncated forms. The deletion of N-terminal extra amino acid residues of Xyn2083GH11 decreased catalytic activity as well as the stability of the enzyme. The hydrolysis rates of cellulose and xylan in the pretreated corn cobs were 90.56% and 72.80% with the addition of Xyn2083GH11 and cellulase, whereas those were 67.95% and 34.45% using sole cellulase respectively. The structural analysis of substrates indicated that the addition of Xyn2083GH11 led to a looser structure and more exposure of crystal cellulose for cellulase to approach.
Conclusions
Since the native multi-CDs’ xylanases are rare, the thermostable Xyn2083 provides a good source for functional studies of two CDs coexisted in one xylanase and for potential applications after modification.





Similar content being viewed by others
References
Biely P, Vršanská M, Tenkanen M, Kluepfel D (1997) Endo-β-1, 4-xylanase families: differences in catalytic properties. J Biotechnol 57:151–166
Chang S, Guo Y, Wu B, He B (2017) Extracellular expression of alkali tolerant xylanase from Bacillus subtilis Lucky9 in E. coli and application for xylo oligosaccharides production from agro-industrial waste. Int J Biol Macromol 96:249–256
Feng H, Sun Y, Zhi Y, Mao L, Luo Y, Xu L, Wang L, Zhou P (2015) Expression and characterization of a novel endo-1,4-β-xylanase produced by Streptomyces griseorubens JSD-1 isolated from compost-treated soil. Ann Microbiol 65:1771–1779
Geng A, Wang H, Wu J, Xie R, Sun J (2017) Characterization of a β-xylosidase from Clostridium clariflavum and its application in xylan hydrolysis. BioResources 12:9253–9262
Goncalves GA, Takasugi Y, Jia L, Noda S, Tanaka T, Ichinose H, Kamiya N (2015) Synergistic effect and application of xylanases as accessory enzymes to enhance the hydrolysis of pretreated bagasse. Enzyme Microb Technol 72:16–24
Hu J, Arantes V, Pribowo A, Saddler JN (2013) The synergistic action of accessory enzymes enhances the hydrolytic potential of a “cellulase mixture” but is highly substrate specific. Biotechnol Biofuels 6:112
Izquierdo JA, Goodwin L, Davenport KW et al (2012) Complete genome sequence of Clostridium clariflavum DSM 19732. Stand Genomic Sci 6:104–115
Juturu V, Wu JC (2012) Microbial xylanases: engineering, production and industrial applications. Biotechnol Adv 30:1219–1227
Maitan-Alfenas GP, Visser EM, Guimarães VM (2015) Enzymatic hydrolysis of lignocellulosic biomass: converting food waste in valuable products. Curr Opin Food Sci 1:44–49
Melo-Silveira RF, Fidelis GP, Viana RLS, Soeiro VC, da Silva RG, Machado D, Costa LS, Ferreira CV, Rocha HAO (2014) Antioxidant and antiproliferative activities of methanolic extract from a neglected agricultural products: corn cobs. Molecules 19:5360–5378
Shiratori H, Sasaya K, Ohiwa H, Ayame S, Miya A, Beppu T, Ueda K (2009) Clostridium clariflavum sp. nov. and Clostridium caenicola sp. nov., moderately thermophilic, cellulose-/cellobiose-digesting bacteria isolated from methanogenic sludge. Int J Syst Evol Microbiol 59:1764–1770
Wang G, Ren Y, Ng TB, Streit WR, Ye X (2019) High-throughput amplicon sequencing demonstrates extensive diversity of xylanase genes in the sediment of soda lake Dabusu. Biotechnol Lett 41:409–418
Zhang Q, He J, Tian M, Mao Z, Tang L, Zhang J, Zhang H (2011) Enhancement of methane production from cassava residues by biological pretreatment using a constructed microbial consortium. Bioresour Technol 102:8899–8906
Zhang Q, Li H, Zhu X, Lai F, Zhai Z, Wang Y (2016) Exploration of the key functional proteins from an efficient cellulolytic microbial consortium using dilution-to-extinction approach. J Environ Sci (China) 43:199–207
Acknowledgements
This work was supported by the Program of Introducing Talents of Discipline to Universities (111-2-06), the National First-class Discipline Program of Light Industry Technology and Engineering (LITE2018-27), and Top-notch Academic Programs Project of Jiangsu Higher Education Institutions (PPZY2015A056).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
10529_2019_2721_MOESM1_ESM.docx
Supplementary Table 1—Primers used for amplification of the genes of Xyn2083 derivatives. Supplementary Fig. 1—Xyn2083GH11and its truncated forms. Supplementary material 1 (DOCX 40 kb)
Rights and permissions
About this article
Cite this article
Liu, Y., Sun, Y., Wang, H. et al. Characterization of a novel multi-domain xylanase from Clostridium clariflavum with application in hydrolysis of corn cobs. Biotechnol Lett 41, 1177–1186 (2019). https://doi.org/10.1007/s10529-019-02721-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-019-02721-2