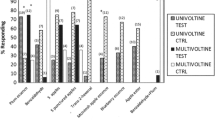
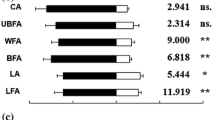
Abstract
The larval parasitoid Cotesia urabae Austin and Allen (Hymenoptera: Braconidae) is known to be attracted to odours of its target host Uraba lugens Walker (Lepidoptera: Nolidae), host plant (Eucalyptus species), and target plant-host complex. Cotesia urabae females were tested in both a Y-tube and four-arm olfactometer to further investigate these attractions as well as their attraction to three non-target Lepidoptera (two in the family Erebidae and one in the family Geometridae), and their corresponding host plants and plant-host complexes. In a Y-tube olfactometer, wasps were attracted to the odours of the non-target Erebidae larvae when tested on their own and when feeding on their host plants, but not to their non-target host plants alone, suggesting some rare circumstances in the field these non-targets could be attacked by C. urabae. The multiple-comparison bioassay conducted in a four-arm olfactometer indicates that target plant-host complex odours invariably produced the strongest attraction compared with any other of the non-target plant-host complex odours tested. Cotesia urabae females that were given prior exposure and the opportunity to oviposit within either non-target species were not subsequently more attracted to the Erebidae odours, suggesting that associative learning is unlikely to increase non-target attack. Such olfactometer assays could be a very useful addition to the host specificity testing methods able to be conducted within quarantine facilities, prior to the release of candidate biological control agents. We urge other biocontrol scientists to undertake similar assays to assist with non-target risk assessments.

Similar content being viewed by others
References
Agelopoulos N, Keller M (1994) Plant- natural enemy association in tritrophic system, Cotesia rubecula–Pieris rapae–Brassicaceae (Cruciferae). III: collection and identification of plant and frass volatiles. J Chem Ecol 20:1955–1967
Allen GR (1990) Uraba lugens Walker (Lepidoptera: Noctuidae): Larval survival and parasitoid biology in the field in South Australia. J Aust Entomol Soc 29:301–312
Avila GA, Berndt LA, Holwell GI (2013) First releases and monitoring of the biological control agent Cotesia urabae Austin and Allen (Hymenoptera: Braconidae). N Z Entomol 36:65–72
Avila GA, Withers TM, Holwell GI (2016) Olfactory cues used in host-habitat location and host location by the parasitoid Cotesia urabae (Hymenoptera: Braconidae). Entomol Exp Appl. doi:10.1111/eea.12393
Avila GA, Withers TM, Holwell GI (2015) Host testing of the parasitoid Cotesia urabae (Austin & Allen, 1989) (Hymenoptera: Braconidae) to assess the risk posed to the New Zealand nolid moth Celama parvitis (Howes, 1917) (Lepidoptera: Nolidae): do host deprivation and experience influence acceptance of non-target hosts? Aust Entomol 54:270–277
Bai S-X, Wang Z-Y, He K-L, Im D-J (2011) Olfactory response of Trichogramma ostriniae (Hymenoptera: Trichogrammatidae) to volatiles emitted by mungbean plants. Agric Sci China 10:560–565
Berndt L, Brockerhoff EG, Jactel H, Weiss T, Beaton J (2004) Biology and rearing of Pseudocoremia suavis, an endemic looper (Lepidoptera: Geometridae) with a history of outbreaks on exotic conifers. N Z Entomol 27:73–82
Berndt LA, Allen GR (2010) Biology and pest status of Uraba lugens Walker (Lepidoptera: Nolidae) in Australia and New Zealand. Aust J Entomol 49:268–277
Berndt LA, Sharpe A, Withers TM, Kimberley M, Gresham B (2010) Evaluation & review report for the release of Cotesia urabae for the biological control of gum leaf skeletoniser. Application ER-AF-NOR-1-2 09/05. Appendix 2: risks to non-target species from potential biological control agent Cotesia urabae against Uraba lugens in New Zealand. http://www.epa.govt.nz/search-databases/HSNO%20Application%20Register%20Documents/Application%20Appendix%202.pdf. Accessed 05 Sept 2015
Berndt LA, Mansfield S, Withers TM (2007) A method for host range testing of a biological control agent for Uraba lugens. N Z Plant Prot 60:286–290
Berndt LA, Withers TM, Mansfield S, Hoare RJB (2009) Non-target species selection for host range testing of Cotesia urabae. N Z Plant Prot 62:168–173
Berry JA, Walker GP (2004) Meteorus pulchricornis (Wesmael) (Hymenoptera: Braconidae: Euphorinae): An exotic polyphagous parasitoid in New Zealand. N Z J Zool 31:33–44
Cameron E (1935) A study of the natural control of ragwort (Senecio jacobaea L.). J Ecol 23:265–322
Cameron PJ, Walker GP (1997) Host specificity of Cotesia rubecula and Cotesia plutellae, parasitoids of white butterfly and diamondback moth. In: Proceedings of the 50th New Zealand plant protection conference, Lincoln University, Canterbury, New Zealand, pp 236–241
Cameron PJ, Walker GP, Keller MA (1997) Clearwater JR Host specificity assessments of Cotesia plutellae, a parasitoid of diamondback moth. In: Sivapragasam A, Loke WH, Hussan AK, Lim GS (eds) The management of diamondback moth and other crucifer pests: Proceedings of the third international workshop. Kuala Lumpur, Malaysia, pp 85–89
Chinwada P, Schulthess F, Overholt W, Jowah P, Omwega C (2008) Release and establishment of Cotesia flavipes for biological control of maize stemborers in Zimbabwe. Phytoparasitica 36:160–167
Conover WJ (1999) Practical nonparametric statistics, 3rd edn. Wiley, New York
Conti E, Salerno G, Bin F, Bradleigh Vinson S (2004) The role of host semiochemicals in parasitoid specificity: a case study with Trissolcus brochymenae and Trissolcus simoni on pentatomid bugs. Biol Control 29:435–444
Cugala D, Overholt W, Santos L, Giga D (2001) Release of Cotesia flavipes Cameron for biological control of cereal stemborers in two ecological zones in Mozambique. Insect Sci Appl 21:303–310
Dicke M, Baarlen P, Wessels R, Dijkman H (1993) Herbivory induces systemic production of plant volatiles that attract predators of the herbivore: extraction of endogenous elicitor. J Chem Ecol 19:581–599
Drost YC, Carde RT (1992) Host switching in Brachymeria intermedia (Hymenoptera: Chalcididae), a pupal endoparasitoid of Lymantria dispar (Lepidoptera: Lymantriidae). Environ Entomol 21:760–766
El-Wakeil N, Farghaly H, Ragab Z (2010) Efficacy of inundative releases of Trichogramma evanescens in controlling Lobesia botrana in vineyards in Egypt. J Pest Sci 83:379
Godfray HCJ (1994) Parasitoids: behavioral and evolutionary ecology. Princeton University Press, Princeton
Jembere B, Ngi-Song AJ, Overholt W (2003) Olfactory responses of Cotesia flavipes (Hymenoptera: Braconidae) to target and non- target Lepidoptera and their host plants. Biol Control 28:360–367
Martin NA (2009) Plant-SyNZ™: an invertebrate herbivore biodiversity assessment tool. Landcare Research. http://plant-synz.landcareresearch.co.nz/DetailsForm.aspx?Type=H&RecordId=2463&LSID=NAM52459. Accessed 05 June 2015
Munro VMW, Henderson IM (2002) Nontarget effect of entomophagous biocontrol: shared parasitism between native lepidopteran parasitoids and the biocontrol agent Trigonospila brevifacies (Diptera: Tachinidae) in forest habitats. Environ Entomol 31:388–396
Ngi-Song A, Overholt W, Njagi P, Dicke M, Ayertey J, Lwande W (1996) Volatile infochemicals used in host and host habitat location by Cotesia flavipes (Cameron) and Cotesia sesamiae (Cameron) (Hymenoptera: Braconidae), larval parasitoids of stemborers on graminae. J Chem Ecol 22:307–323
Ngi-Song AJ, Overholt WA (1997) Host location and acceptance by Cotesia flavipes Cameron and Cotesia sesamiae (Cameron) (Hymenoptera: Braconidae), parasitoids of african gramineous stemborers: role of frass and other host cues. Biol Control 9:136–142
Nordlund DA, Lewis WJ, Altieri MA (1988) Influence of plant produced allelochemicals on host-prey selection behavior of entomophagous insects. In: Barbosa P, Letourneau DK (eds) Novel aspects of insect-plant interaction. Wiley, New York, pp 65–90
Obonyo M, Schulthess F, Le Ru B, van den Berg J, Silvain J-F, Calatayud P-A (2010) Importance of contact chemical cues in host recognition and acceptance by the braconid larval endoparasitoids Cotesia sesamiae and Cotesia flavipes. Biol Control 54:270–275
Orr DB, Garcia-Salazar C, Landis DA (2000) Trichogramma nontarget impacts: a method for biological control risk assessment. In: Follett PA, Duan JJ (eds) Nontarget effects of biological control. Kluwer Academic Publishers, Boston, pp 111–125
Perfecto I, Vet L (2003) Effect of a nonhost plant on the location behavior of two parasitoids: the tritrophic system of Cotesia spp. (Hymenoptera: Braconidae), Pieris rapae (Lepidoptera: Pieridae), and Brassica oleraceae. Environ Entomol 32:163–174
Péter A (2014) Solomon Coder (version beta 14.10.04): A simple solution and free solution for behavior coding. http://www.solomoncoder.com/. Accessed 09 Nov 2014
Quinn GP, Keough MJ (2002) Experimental design and data analysis for biologists. Cambridge University Press, Cambridge
Romeis J, Shanower T, Zebitz C (1997) Volatile plant infochemicals mediate plant preference of Trichogramma chilonis. J Chem Ecol 23:2455–2465
Roux O, Gers C, Tene-Ghomsi J, Arvanitakis L, Bordat D, Legal L (2007) Chemical characterization of contact semiochemicals for host-recognition and host-acceptance by the specialist parasitoid Cotesia plutellae (Kurdjumov). Chemoecology 17:13–18
Rowbottom R, Allen G, Walker P, Berndt L (2013) Phenology, synchrony and host range of the Tasmanian population of Cotesia urabae introduced into New Zealand for the biocontrol of Uraba lugens. BioControl 58:625–633
Singh P, Mabbett F (1976) Note on the life history of the magpie moth, Nyctemera annulata (Lepidoptera: Arctiidae). N Z J Zool 3:277–278
Sullivan JJ, Winks CJ, Fowler SV (2008) Novel host associations and habitats for Senecio-specialist herbivorous insects in Auckland. N Z Ecol Soc 32:219–224
Tumlinson JH, Lewis WJ, Vet LEM (1993) Parasitic wasps, chemically guided intelligent foragers. Sci Am 268:100–106
Turlings T, Tumlinson J, Eller F, Lewis W (1991) Larval-damaged plants: source of volatile synomones that guide the parasitoid Cotesia marginiventris to the micro-habitat of its hosts. Entomol Exp Appl 58:75–82
Turlings TC, Tumlinson JH (1992) Systemic release of chemical signals by herbivore-injured corn. PNAS 89:8399–8402
Turlings TCJ, Tumlinson JH, Lewis WJ (1990) Exploitation of herbivore-induced plant odors by host-seeking parasitic wasps. Science 250:1251–1253
Turlings TCJ, Wackers FL (2004) Recruitment of predators and parasitoids by herbivore-injured plants. In: Cardé RT, Millar JG (eds) Advances in insect chemical ecology. Cambridge University Press, Cambridge, pp 21–75
Turlings TCJ, Wackers FL, Vet LEM, Lewis WJ, Tumlinson JH (1993) Learning of host-finding cues by hymenopterous parasitoids. In: Papaj DR, Lewis AC (eds) Insect learning: ecological and evolutionary perspectives, vol 3. Chapman & Hall, New York, pp 51–78
van Driesche R, Nunn C, Kreke N, Goldstein B, Benson J (2003) Laboratory and field host preferences of introduced Cotesia spp. parasitoids (Hymenoptera: Braconidae) between native and invasive Pieris butterflies. Biol Control 28:214–221
van Lenteren JC, Bale J, Bigler F, Hokkanen HMT, Loomans AJM (2006) Assessing risks of releasing exotic biological control agents of arthropod pests. Ann Rev Entomol 51:609–634
van Poecke RMP, Roosjen M, Pumarino L, Dicke M (2003) Attraction of the specialist parasitoid Cotesia rubecula to Arabidopsis thaliana infested by host or non-host herbivore species. Entomol Exp Appl 107:229–236
Vet LEM, Dicke M (1992) Ecology of infochemical use by natural enemies in a tritrophic context. Ann Rev Entomol 37:141–172
Vet LEM, van Lenteren JC, Heymans M, Meelis E (1983) An airflow olfactometer for measuring olfactory responses of hymenopterous parasitoids and other small insects. Physiol Entomol 8:97–106
Vet LEM, Lewis WJ, Carde RT (1995) Parasitoid foraging and learning. In: Carde RT, Bell WJ (eds) Chemical ecology of insects, vol 2. Chapman & Hall, New York, pp 65–101
Vet LEM, Wäckers FL, Dicke M (1991) How to hunt for hiding hosts: the reliability-detectability problem in foraging parasitoids. Neth J Zool 41:202–213
Vinson SB (1984) Parasitoid-host relationship. In: Bell WJ, Cardé RT (eds) Chemical ecology of insects. Chapman & Hall, London, pp 206–233
Vinson SB (1998) The general host selection behavior of parasitoid Hymenoptera and a comparison of initial strategies utilized by larvaphagous and oophagous species. Biol Control 11:79–96
Xiaoyi W, Zhongqi Y (2008) Behavioral mechanisms of parasitic wasps for searching concealed insect hosts. Acta Ecol Sin 28:1257–1269
Yong T-H, Pitcher S, Gardner J, Hoffmann M (2007) Odor specificity testing in the assessment of efficacy and non-target risk for Trichogramma ostriniae (Hymenoptera: Trichogrammatidae). Biocontrol Sci Tech 17:135–153
Zahiri R, Kitching IJ, Lafontaine JD, Mutanen M, Kaila L, Holloway JD, Wahlberg N (2010) A new molecular phylogeny offers hope for a stable family level classification of the Noctuoidea (Lepidoptera). Zool Scr 40:158–173
Acknowledgments
Thanks to Maria Saavedra and Nicky Kerr who assisted with the rearing of the Cotesia urabae colony, and also to Anne Barrington (Plant and Food Research) and Lindsay McIntyre for supplying non-target species larvae for this research project. This work was partly funded by Scion as part of the Better Border Biosecurity (B3) (http://www.b3nz.org) research collaboration.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling Editor: Stefano Colazza.
Rights and permissions
About this article
Cite this article
Avila, G.A., Withers, T.M. & Holwell, G.I. Laboratory odour-specificity testing of Cotesia urabae to assess potential risks to non-target species. BioControl 61, 365–377 (2016). https://doi.org/10.1007/s10526-016-9716-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10526-016-9716-5