Abstract
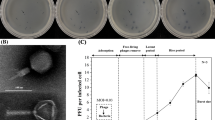
Bacteriophage isolated from the semi-intensive culture of Pacific white leg shrimp Litopenaeus vannamei infects the luminous bacteria Vibrio harveyi. Lytic activity and lytic spectrum results revealed that the isolated phage had strong lytic activity in V. harveyi, V. parahaemolyticus, and V. vulnificus. Biofilm inhibition activity was performed against different pathogenic vibrios on high-density polyethylene (HDPE) template and the result revealed that the phage effectively inhibited the biofilm formation in V. harveyi. Spectrophotometric assay performed for lytic activity of the isolated phage in V. harveyi liquid culture showed that the phage significantly decreased the V. harveyi cell densities at different time intervals (P < 0.05). To study the stability of phage at different temperature and pH revealed that the phage withstands the temperature ranged between 40 and 70 °C and the pH of 4 and 9 at a significant level (P < 0.001). One-step growth curve depicted that the burst size gradually increased to a significant level and reached the maximum of 90% at 180 min (P < 0.05). This study concluded that the isolated phage had specific activity against pathogenic V. harveyi infections.




Similar content being viewed by others
Data availability
Nil.
Code availability
Not applicable.
References
Adam MH (1959) Bacteriophages. Interscience Publishers Inc., New York, p 450
Ahiwale S, Tambol N, Thorat K, Kulkarni R, Ackermann H, Kapadnis B (2011) In vitro management of hospital Pseudomonas aeruginosa biofilm using indigenous T7-like lytic phage. Curr Microbiol 62:335–340
Alagappan K, Karuppiah V, Deivasigamani B (2016) Protective effect of phages on experimental V. parahaemolyticus infection and immune response in shrimp (Fabricius, 1798). Aquaculture 453:86–92
Almeida A, Cunha A, Gomes NC, Alves E, Costa L, Faustino AFM (2009) Phage therapy and photodynamic therapy: low environmental impact approaches to inactivate microorganisms in fish farming plants. Mar Drugs 7:268–313. https://doi.org/10.3390/md7030268
Anon (2020) Shrimp market report 2020: global industry size analysis by value chain, SWOT analysis, drivers and opportunities by region and forecast to 2024
Baer A, Kehn-Hall K (2014) Viral concentration determination through plaque assays: using traditional and novel overlay systems. J vis Exp 93:e52065
Carson L, Gilmore GSP, BF, (2010) The use of lytic bacteriophages in the prevention and eradication of biofilms of Proteus mirabilis and Escherichia coli. FEMS Immunol Med Microbiol 59:447–455
Chittem PB, Kunda SB (2018) Socio-economic condition of the Litopenaeusvannamei farmers with implementation of better management practices (BMP’s) in Andhra Pradesh, India. International Journal of Fisheries and Aquatic Studies 6(6):325–331
Chopin MC, Chopin A, Roux C (1976) Definition of bacteriophage groups according to their lytic action on mesophilic lactic Streptococci. Applied and Environmental Microbiology, pp.741–746.
Chrisolite B, Thiyagarajan S, Alavandi SV, Abhilash EC, Kalaimani N, Vijayan KK, Santiago TC (2008) Distribution of luminescent Vibrio harveyi and their bacteriophages in a commercial shrimp hatchery in South India. Aquaculture 275:13–19
Clarke T (2003) Drug companies snub antibiotics as pipeline threatens to run dry. Nature 425:225
Corbian BD, Mclean RJC, Aron GM (2001) Bacteriophage T4 multiplication in a glucose limited E. coli biofilm. Can J Microbiol 47:680–684
Defoirdt T, Sorgeloos P, Bossier P (2011) Alternatives to antibiotics for the control of bacterial disease in aquaculture. Curr Opin Microbiol 14:251–258
Doolittle MM, Cooney JJ, Cladwell DE (1995) Lytic infection of Escherichia coli biofilms by bacteriophage T4. Can J Microbiol 41:12–18
Gutierrez D, Martinez B, Rodriguez A, Garcia P (2010) Isolation and characterization of bacteriophages infecting Staphylococcus epidermidis. Curr Microbiol 61:601–608
Jensen KC, Hair BB, Wienclaw TM, Murdock MH, Hatch JB, Trent AT, White TD, Haskell KJ, Berges BK (2015) Isolation and host range of bacteriophage with lytic activity against methicillin-resistant Staphylococcus aureus and potential use as a fomite decontaminant. PLoS ONE 10:7
Jonczyk E, Kłak M, Międzybrodzki R, Gorski A (2011) The influence of external factors on bacteriophages-review. Folia Microbiol 56:191–200
Karunasagar I, Pai R, Malathi GR, Karunasagar I (1994) Mass mortality of Penaeus monodon larvae due to antibiotic resistant Vibrio harveyi infection. Aquaculture 128:203–209
Karunasagar I, Otta SK, Karunasagar I (1996) Biofilm formation by Vibrio harveyi on various surfaces. Aquaculture 140:241–245
Karunasagar I, Shivu MM, Girisha SK, Krohne G, Karunasagar I (2007) Biocontrol of pathogens in shrimp hatcheries using bacteriophages. Aquaculture 268:288–292
Karunasagar I, Vinod M, Kennedy B, Vijay A, Deepanjali A, Umesh K, Karunasagar I (2005) Biocontrol of bacterial pathogens in aquaculture with emphasis on phage therapy. In: Walker, P., Lester, R., Bondad-Reantaso, M.G. (Eds.), Diseases in Asian aquaculture V.: Fish health section. Asian Fisheries Society, Manila, pp. 535–542.
Lavilla-Pitogo CR, Baticados MCL, Cruz-Lacierda ER, de la Pena LD (1990) Occurrence of luminous bacterial disease of Penaeusmonodon larvae in the Philippines. Aquaculture 91:1–13
Luu QH, Nguyen TBT, Nguyen TLA (2021) Antibiotics use in fish and shrimp farms in Vietnam. Aquac Reports 20:100711
Martínez-Díaz SF, Hipólito-Morales A (2013) Efficacy of phage therapy to prevent mortality during the vibriosis of brine shrimp. Aquaculture 400–401:120–124
Mateus L, Costa L, Silva YJ, Pereira C, Cunha A, Almeida A (2014) Efficiency of phage cocktails in the inactivation of Vibrio in aquaculture. Aquaculture 424–425:167–173
Matsuzaki S, Tanaka S, Koga T, Kawata T (1992) A broad-host-range vibrio phage KVP40 isolated from sea water. Microbiol Immunol 36:93–97
Matsuzaki S, Rashel M, Uchiyama J, Sakurai S, Ujihara T, Kuroda M, Ikeuchi M, Tani T, Fujieda M, Wakiguchi H, Imai S (2005) Bacteriophage therapy: a revitalized therapy against bacterial infectious diseases. J Infect Chemother 11(5):211–219
Miller ES, Heidelberg JF, Eisen JA, Nelson WC, Durkin AS, Ciecko A, Feldblyum TV, White O, Paulsen IT, Nierman WC, Lee J, Szczypinski B, Fraser CM (2003) Complete genome sequence of the broad-host-range vibriophage KVP40: comparative genomics of a T4-related bacteriophage. J Bacteriol 185:5220–5233
Mullan WMA (2001) Isolation and purification of bacteriophages. [On-line] UK. Available at http://www.dairyscience.info/isolation-and-purification-ofbacteriophages.html
Nakai T, Park SC (2002) Bacteriophage therapy of infectious diseases in aquaculture. Res Microbiol 153:13–18. https://doi.org/10.1016/S0923-2508(01)01280-3
Nakai T, Sugimoto R, Park KH, Matsuoka S, Mori K, Nishioka T, Maruyama K (1999) Protective effects of bacteriophage on experimental Lactococcusgarvieae infection in yellowtail. Dis Aquat Organ 37:33–41
Oliveira J, Castilho F, Cunha A, Pereira MJ (2012) Bacteriophage therapy as a bacterial control strategy in aquaculture. Aquac Int 20:879–910
Orndorff SA, Colwell RR (1980) Distribution and identification of luminous bacteria from the Sargasso Sea. Appl Environ Microbiol 39:983–987
Pajunen M, Kiljunen S, Skurnik M (2000) Bacteriophage fYeO3-12, specific for Yersinia enterocolitica serotype O:3, is related to coliphages T3 and T7. J Bacteriol 182:5114–5120
Pasharawipas T, Thaikua S, Sriurairatana S, Ruangpan L, Direkbusarakum S, Manopvisetcharean J, Flegel TW (2005) Partial characterization of a novel bacteriophage of Vibrio harveyi isolated from shrimp culture ponds in Thailand. Virus Res 114:63–69
Pasharawipas T, Manopvisetcharean J, Flegel TW (2011) Phage treatment of Vibrio harveyi: a concept of protection against bacterial infection. Res J Microbiol 6(6):560–567
Patil PK (2016) Background for the workshop. In: Patil PK, Alavandi SV, Otta SK, Avunje S, Raja RA (Eds.), Proceedings of the workshop on Responsible Use of Antimicrobials in Indian Aquaculture : Opportunities and Challenges, ICAR - Central Institute of Brackishwater Aquaculture, Chennai, Tamil Nadu, India, pp. 44.
Phumkhachorn P, Rattanachaikunsopon P (2010) Isolation and partial characterization of a bacteriophage infecting the shrimp pathogen Vibrio harveyi. Afr J Microbiol 4:1794–1800
Prada-Peñarandaa C, Salazarb M, Güizab L, Pérezc MI, Leidyc C, Vives-Floreza MJ (2018) Phage preparation FBL1 prevents Bacillus licheniformis biofilm, bacterium responsible for the mortality of the Pacific White Shrimp Litopenaeusvannamei. Aquaculture 484:160–167
Richards GP (2014) Bacteriophage remediation of bacterial pathogens in aquaculture: a review of the technology. Bacteriophage 4:e975540
Ruby EG, Nealson KH (1978) Seasonal changes in the species composition of luminous bacteria in nearshore seawater. Limnol Oceanogr 23:530–533
Sasikala D, Srinivasan P (2016) Characterization of potential lytic bacteriophage against Vibrio alginolyticus and its therapeutic implications on biofilm dispersal. Microb Pathog 101:24–35
Shende RK, Hirpurkar SD, Sannat C, Rawat N, Pandey V (2017) Isolation and characterization of bacteriophages with lytic activity against common bacterial pathogens. Veterinary World 10:973–978
Shivu MM, Rajeeva BC, Girisha SK, Karunasagar I, Krohne G, Karunasagar I (2007) Molecular characterization of Vibrio harveyi bacteriophages isolated from aquaculture environments along the coast of India. Environ Microbiol 9(2):322–331
Srinivasan P, Ramasamy P (2017) Morphological characterization and biocontrol effects of Vibrio vulnificus phages against vibriosis in the shrimp aquaculture environment. Microb Pathog 111:472–480
Sunaryanto A, Mariam A (1986) Occurrence of a pathogenic bacteria causing luminescence in penaeid larvae in Indonesian hatcheries. Bulletin of Brackishwater Aquaculture Developmental Center 8(64):70
Teplitski M, Wright A, Lorca G (2009) Biological approaches for controlling shellfish-associated pathogens. Curr Opin Biotechnol 20(2):185–190
Vinod MG, Shivu MM, Umesha KR, Rajeeva BC, Krohne G, Karunasagar I, Karunasagar I (2006) Isolation of Vibrio harveyi bacteriophage with a potential for biocontrol of luminous vibriosis in hatchery environments. Aquaculture 255:117–124
Wu JL, Lin HM, Jan L, Hsu YL, Chang LH (1981) Biological control of fish bacterial pathogen Aeromonashydrophila by bacteriophage. Fish Pathol 15:271–276
Yele AB, Thawal ND, Sahu PK, Chopade BA (2012) Novel lytic bacteriophage AB7-IBB1 of Acinetobacterbaumannii: isolation, characterization and its effect on biofilm. Arch Virol 157:1441–1450
Zahid MS, Waise Z, Kamruzzaman M, Ghosh AN, Nair GB, Khairul Bashar SA, Mekalanos JJ, Faruque SM (2011) An experimental study of phage mediated bactericidal selection and emergence of the El Tor Vibrio Cholerae. Indian J Med Res 133:218–224
Zar JH (1974) Biostatistical analysis. Prentice Hall, New Jersey
Zhou JF, Fang W, Yang X, Zhou S, Hu L, Li X, Qi X, Su H, Xie L (2012) A nonluminescent and highly virulent Vibrio harveyi strain is associated with bacterial white tail disease of Litopenaeusvannamei shrimp. PLoS ONE 7(2):e29961
Acknowledgements
We thank University Grants Commission (UGC), India.
Funding
This work was financially supported by the Basic Science Research (BSR), University Grants Commission (UGC), New Delhi, Government of India (grant F.25–1/2013–2014 [BSR]/7–374/2012 [BSR] dated 30 May 2014).
Author information
Authors and Affiliations
Contributions
CL, ET, and CA were involved in experimental works. GU and MMB are involved in data analysis. TC is responsible for the conceptualization of this work and overall manuscript preparation.
Corresponding author
Ethics declarations
This is to declare that all authors unanimously submit the manuscript in the journal Aquaculture International.
Ethics approval
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Handling Editor: Gavin Burnel
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Lelin, C., Thirumalaikumar, E., Uma, G. et al. Isolation and partial characterization of bacteriophages infecting Vibrio harveyi from shrimp farm effluent water. Aquacult Int 30, 2081–2094 (2022). https://doi.org/10.1007/s10499-022-00891-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10499-022-00891-x