Abstract
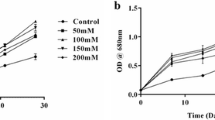
In this study, Dolichospermum affine was cultivated under starvation and limitation conditions, using six different concentrations of nitrate (NaNO3) and phosphate (K2HPO4) in BG-11 medium. The biomass, pigments, proteins, carbohydrates, lipids, triacylglycerol-TAG, and fatty acid responses to different modified media were examined in terms of cell density, dry biomass, optical density, growth rate, chlorophyll-a, carotene content, and lipid productivity. Molecular characterization of D. affine isolates was performed using Fourier transform–infrared spectroscopy (FT-IR) and polymerase chain reaction (PCR). In the 20-day batch culture period, one of the modified media, %50 N and P limitation (N-PLimitation), exhibited higher values than the others in terms of cell density (1.41 × 107 cells/mL), dry biomass (1.68 gL−1), optical density (1.26), growth rate (0.48 day−1), chlorophyll-a (3.76 μg L−1), and total carotenoid (12.99 μg L−1). The maximum percentage lipid content of D. affine was obtained in the control group at 10.67%. Fatty acid profiles have been investigated in six conditions and consisted of 10 fatty acids. The basic saturated fatty acid palmitic acid was (C16:0) for all conditions, and its rate was 40.61% for starvation of N and P (N-PDeprivation) conditions. Oleic acid (C18:1) rate was detected in all groups, but the highest rate was found in the control group with 33.72%. In all conditions, TAG values were found similar, but the highest value was determined at 50% P and 100% N (PLimitation-NControl) (27.5%) modified media. Carbohydrate content (20.37%) was found to be high under N and P starvation (N-PDeprivation), while protein content (66.92%) was high under 50% N and 100% P (NLimitation-PControl) conditions. The results obtained from this study showed that nitrogen and phosphorus starvation and limitation affecting the biomass, valuable secondary metabolites and lipid accumulation and affected the fatty acid composition of D. affine cultures.






Similar content being viewed by others
References
Abed RMM, Dobretsov S, Sudesh K (2009) Applications of cyanobacteria in biotechnology. J Appl Microbiol 106:1–12
Alonso DL, Belarbi EH, Fernández-Sevilla JM, Rodríguez-Ruiz J, Grima EM (2000) Acyl lipid composition variation related to culture age and nitrogen concentration in continuous culture of the microalga Phaeodactylum tricornutum. Phytochemistry 54:461–471
AOAC (1990) Official methods of analysis of the association of official analytical chemists. Arlington
Battah MG, El-Ayoty YM, Esmael AE, Abd El-Ghany SE (2014) Effect of different concentrations of sodium nitrate, sodium chloride, and ferrous sulphate on the growth and lipid content of Chlorella vulgaris. J Agric Technol 10(2):339–353
Becker EW (2007) Micro-algae as a source of protein. Biotechnol Adv 25:207–210
Benning LG, Phoenix VR, Yee N, Tobin MJ (2004) Molecular characterization of cyanobacterial silification using synchrotron infrared micro-spectroscopy. Geocim Cosmochim Acta 68:729–741
Bhamawat PM (2010) Growth of Chlamydomonas reinhardtii under nutrient-limited conditions in steady-state bioreactors. Dissertation, Faculty of the Graduate School, of Cornell University.
Bojović BM, Stojanović J (2005) Chlorophyll and carotenoid content in wheat cultivars as a function of mineral nutrition. Arch Biol Sci 57:283–290
Castenholz RW (2001) Phylum BX. Cyanobacteria. Oxygenic photosynthetic bacteria. In: Garrity G, Boone DR, Castenholz RW (eds) Bergey’s manual of systematic bacteriology. Springer-Verlag, New York, pp 474–487
Chia MA, Lombardi AT, Melao MGG (2013) Growth and biochemical composition of Chlorella vulgaris in different growth media. Ann Braz Acad Sci 85(4):1427–1438
Chisti Y (2008) Biodiesel from microalgae beats bioethanol. Trends Biotech 26:126–131
Dahms HU, Xu Y, Pfeiffer C (2006) Antifouling potential of cyanobacteria: a mini-review. Biofouling 22:317–327
Das K, Sarma GC (2015) Optimization of culture media for the growth of Anabaena spiroides and Nostoc punctiformae of Jorhat district, Assam. IOSR J Pharm Biol Sci 10(2):37–41
Dean AP, Martin MC, Sigee DC (2007) Resolution of codominant phytoplankton species in a eutrophic lake using synchrotron-based Fourier transform infrared spectroscopy. Phycologia 46(2):151–159
Dutta D, De D, Chaudhuri S, Bhattacharya SK (2005) Hydrogen production by cyanobacteria. Microb Cell Factories. https://doi.org/10.1186/1475-2859-4-36
Duygu DY, Udoh AU, Ozer TB, Akbulut A, Erkaya IA, Yildiz K, Guler D (2012) Fourier transform infrared (FTIR) spectroscopy for identification of Chlorella vulgaris Beijerinck 1890 and Scenedesmus obliquus (Turpin) Kützing 1833. Afr J Biotechnol 11(16):3817–3824
Garcia-Pichel F, Pringault O (2001) Cyanobacteria track water in desert soils. Nature 413:380–381
Giordano M, Kansiz M, Heraud P, Beardall J, Wood B, McNaughton D (2001) Fourier transform infrared spectroscopy as a novel tool to investigate changes in intracellular macromolecular pools in the marine alga Chaetoceros muellerii (Bacillariphyceae). J Phycol 37:271–279
Godoy-Hernández G, Vázquez-Flota FA (2006) Growth measurements: estimation of cell division and cell expansion. In: Loyola-Vargas VM, Vázquez-Flota F (eds) Plant cell culture protocols. Humana Press Inc, New Jersey, pp 51–58
Gouveia L, Oliveira AC (2009) Microalgae as a raw material for biofuels production. J Ind Microbiol Biotechnol 36:269–274
Guiry MD, Guiry GM (2018) AlgaeBase. World-wide electronic publication, National University of Ireland, Galway. http://www.algaebase.org. Cited 21 October 2018
Guschina IA, Harwood JL (2006) Lipids and lipid metabolism in eukaryotic algae. Prog Lipid Res 45:160–186
Hannon M, Gimpel J, Tran M, Rasala B, Mayfield S (2010) Biofuels from algae: challenges and potential. Biofuels 1(5):763–784
Hempel N, Petric I, Behrendt F (2012) Biomass productivity and productivity of fatty acids and amino acids of microalgae strains as key characteristics of suitability for biodiesel production. J Appl Phycol 24:1407–1418
Hsieh CH, Wu WT (2009) Cultivation of microalgae for oil production with a cultivation strategy of urea limitation. Bioresour Technol 100:3921–3926
Hu H, Gao K (2005) Response of growth and fatty acid compositions of Nannochloropsis sp. to environmental factors under elevated CO2 concentration. Biotechnol Lett 28:987–992
Hu Q, Sommerfeld M, Jarvis E, Ghirardi M, Posewitz M, Seibert M, Darzins A (2008) Microalgal triacylglycerols as feedstocks for biofuel production: perspectives and advances. Plant J 54(4):621–639
Ilavarasi A, Mubarakali D, Praveenkumar R, Baldev E, Thajuddin N (2011) Optimization of various growth media to freshwater microalgae for biomass production. Biotechnology 10(6):540–545
John DM, Witton BA, Brook AJ (2002) The freshwater algal Flora of the British Isles. Cambridge
Johnson X, Alric J (2013) Central carbon metabolism and electron transport in Chlamydomonas reinhardtii: metabolic constraints for carbon partitioning between oil and starch. Eukaryot Cell 12:776–793
Kaplan D, Richman A, Boussiba S, Vonshak A, Cohen Z, Abeliovich A (1986) Development of an outdoor system for production of lipid-rich halotolerant microalgae: effect of light on biomass production in two halotolerant microalgae Nannochloropsis salina and Isochrysis galbana. Solar Energy Research Institute, Golden
Klinthong W, Yang YH, Huang CH, Tan CH (2015) A review: microalgae and their applications in CO2 capture and renewable energy. Aerosol Air Qual Res 15:712–742
Liu J, Huang J, Sun Z, Zhong Y, Jiang Y, Chen F (2011) Differential lipid and fatty acid profiles of photoautotrophic and heterotrophic Chlorella zofingiensis: assessment of algal oils for biodiesel production. Bioresour Technol 102:106–110
Metcalf LD, Schmitz AA, Pelka JR (1966) Rapid preparation of fatty acid esters from lipids for gas chromatographic. Anal Chem 38:514–515
Mujtaba G, Choi W, Lee CG, Lee K (2012) Lipid production by Chlorella vulgaris after a shift from nutrient-rich to nitrogen starvation conditions. Bioresour Technol 123:279–283
Nalewajko C, Murphy TP (2001) Effects of temperature, and availability of nitrogen and phosphorus on the abundance of Anabaena and Microcystis in Lake Biwa, Japan: an experimental approach. Limnology 2:45–48
Neilan BA, Jacobs D, del Dot T, Blackall LL, Hawkins PR, Cox PT, Goodman AE (1997) rRNA sequences and evolutionary relationships among toxic and nontoxic cyanobacteria of the genus Microcystis. Int J Syst Bacteriol 47:693–697
Nigam S, Rai MP, Sharma R (2011) Effect of nitrogen on growth and lipid content of Chlorella pyrenoidosa. Am J Biochem Biotechnol 7(3):124–129
Parsons TR, Strickland JDH (1963) Discussion of spectrophotometric determination of marine plant pigments, with revised equations for ascertaining chlorophylls and carotenoids. J Mar Res 21(3):115–163
Parvin M, Zannat MN, Habib MAB (2007) Two important technique for isolation of microalgae. Asian Fish Sci 20:117–124
Ponnuswamy I, Madhavan S, Shabudeen S (2013) Isolation and characterization of green microalgae for carbon sequestration, waste water treatment and bio-fuel production. Int J Bio Sci Bio Technol 5(2):17–26
Prescott GW (1973) Algae of the western great lakes area. C. Brown Company Publishers, Michigan
Pruvost J, Vooren G, Gouic B, Mossion A, Legrand J (2011) Systematic investigation of biomass and lipid productivity by microalgae in photobioreactors for biodiesel application. Bioresour Technol 102:150–158
Quintana N, Van der Kooy F, Van de Rhee MD, Voshol GP, Verpoorte R (2011) Renewable energy from cyanobacteria: energy production optimization by metabolic pathway engineering. Appl Microbiol Biotechnol 91:471–490
Radakovits R, Jinkerson RE, Darzins A, Posewitz MC (2010) Genetic engineering of algae for enhanced biofuel production. Eukaryot Cell 9:486–501
Řezanka T, Dor I, Prell A, Dembitsky VM (2003) Fatty acid composition of six freshwater wild cyanobacterial species. Folia Microbiol 48(1):71–75
Rippka R, Deruelles J, Waterbury JB, Herdman M, Stainer RY (1979) Generic assignments, strain histories and properties of pure cultures of cyanobacteria. J Gen Microbiol 111:1–61
Sallal AK, Nimer NA, Radwan SS (1990) Lipid and fatty acid composition of freshwater cyanobacteria. J Gen Microbiol 136:2043–2048
Santos-Ballardo DU, Rossi S, Hernández V, Gómez RV, Rendón-Unceta MC, Caro-Corrales J, Valdez-Ortiz A (2015) A simple spectrophotometric method for biomass measurement of important microalgae species in aquaculture. Aquaculture 448:87–92
Schuhmann H, Lim DKY, Schenk PM (2011) Perspectives on metabolic engineering for increased lipid contents in microalgae. Biofuels 3:71–86
Sharathchandra K, Rajashekhar M (2011) Total lipid and fatty acid composition in some freshwater cyanobacteria. J Algal Biomass Utln 2(2):83–97
Sharma KK, Schuhmann H, Schenk PM (2012) High lipid induction in microalgae for biodiesel production. Energies 5:1532–1553
Sigee DC, Dean A, Levado E, Tobin MJ (2002) Fourier-transform infrared spectroscopy of Pediastrum duplex: characterization of a micro-population isolated from a eutrophic lake. Eur J Phycol 37:19–26
Solovchenko A, Khozin-Goldberg I, Didi-Cohen S, Cohen Z, Merzlyak M (2008) Effects of light and nitrogen starvation on the content and composition of carotenoids of the green microalga Parietochloris incisa. Russ J Plant Physiol 55:455–462
Stanier RY, Kunisawa R, Mandel M, Cohen-Bazire G (1971) Purification and properties of unicellular blue-green algae (order Chroococcales). Bacteriol Rev 35:171–205
Tang H, Abunasser N et al (2010) Potential of microalgae oil from Dunaliella tertiolecta as a feedstock for biodiesel. Appl Energy 88:3324–3330
Uslu L (2011) The effects of the changes in the composition of nutrient on lipid content of microalgae. Dissertation, Çukurova University Institute of Natural and Applied Sciences Basic Science Department of Fisheries
Woese CR (1987) Bacterial evolution. Microbiol Rev 51:221–271
Wong YK, Ho YH, Ho KC, Leung HM, Yung KKL (2017) Maximization of cell growth and lipid production of freshwater microalga Chlorella vulgaris by enrichment technique for biodiesel production. Environ Sci Pollut Res 24:9089–9101
Yalçın Duygu D, Erkaya Açıkgöz I, Özer T (2018) Investigating the effect of different growth media on biomass production of Pseudopediastrum boryanum (Turpin) E. Hegewald isolates. J Limnol Freshw Fish Res 4(1):6–12
Yeesang C, Cheirsilp B (2011) Effect of nitrogen, salt, and iron content in the growth medium and light intensity on lipid production by microalgae isolated from freshwater sources in Thailand. Bioresour Technol 102:3034–3040
Zhu CJ, Lee YK, Chao TM (1997) Effects of temperature and growth phase on lipid and biochemical composition of Isochrysis galbana TK1. J Appl Phycol 9:451–457
Acknowledgments
I would like to thank Andrew P. Dean from the University of Manchester for the PCR analysis and Ankara University Faculty of Veterinary Medicine for the nutrient analysis.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The author declares that she no conflict of interest.
Ethical approval
This article does not contain any studies with animals.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yalcin, D. Growth, lipid content, and fatty acid profile of freshwater cyanobacteria Dolichospermum affine (Lemmermann) Wacklin, Hoffmann, & Komárek by using modified nutrient media. Aquacult Int 28, 1371–1388 (2020). https://doi.org/10.1007/s10499-020-00531-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10499-020-00531-2