Abstract
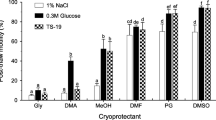
The aim of the present study was to determine the effect of various cryoprotectants on post-thaw sperm quality and fertilizing capacity of cryopreserved scaly carp (Cyprinus carpio) semen. The present study focused on freezing of scaly carp sperm utilizing a practical and inexpensive protocol for aquaculture. Semen was diluted with Kurokura’s extender composing 3.6 g/l NaCl, 10 g/l KCl, 0.22 g/l CaCl2, 0.08 g/l MgCl2 and 0.2 g/l NaHCO3. The extender contained three different cryoprotectants (DMSO, DMA and egg yolk) at ratios of 5, 10 and 15 %. Semen was placed into 0.25-ml straws and exposed to liquid nitrogen vapor (−120 °C) using an insulated box with an adjustable tray for 10 min and then plunged into liquid nitrogen (−196 °C) tank. The thawing process was performed in a water bath at 40 °C for 10 s. The results indicated that type of cryoprotectants and their concentrations are rather effective in scaly carp sperm cryopreservation on post-thaw sperm quality, while they are very important in order to obtain high fertilization rates. The highest fertilization rate was determined as 96.4 ± 0.15 % with 15 % egg yolk, while the highest hatching rate was determined as 99.3 ± 0.80 with 15 % DMA. In conclusion, the applied cryopreservation method for scaly carp sperm is suitable to fertilize high amounts of eggs.
Similar content being viewed by others
References
Alderson R, Macneil AJ (1984) Preliminary investigations of cryopreservation of milt of Atlantic salmon Salmo salar and its application to commercial farming. Aquaculture 43:351–354
Billard R, Cosson J, Perchec G, Linhart O (1995) Biology of sperm and artificial reproduction in carp. Aquaculture 129:95–112
Bozkurt Y, Yavas I, Ogretmen F, Sivaslıgil B, Karaca F (2011) Effect of glycerol on fertility of cryopreserved grass carp (Ctenopharyngodon idella) sperm. Isr J Aquacult-Bamid IIC 63(2011):635
Bozkurt Y, Yavas I, Karaca F (2012) Cryopreservation of brown trout (Salmo trutta macrostigma) and ornamental koi carp (Cyprinus carpio) sperm. In: Katkov I (ed) Current frontiers in cryopreservation. Intech, pp. 293–304
Cabrita E, Robles V, Alvarez R, Herraez MP (2001) Cryopreservation of rainbow trout sperm in large volume straws: application to large scale fertilization. Aquaculture 201:301–304
Christensen JM, Tiersch TR (1996) Cryopreservation of channel catfish sperm: effect of cryoprotectant, straw size and extender formulation. Theriogenology 47:639–645
Dreanno C, Cosson J, Suquet M, Seguin F, Dorange G, Billard R (1999) Nucleotides content, oxidative phosphorylation, morphology and fertilizing capacity of turbot (Psetta maxima) spermatozoa during the motility period. Mol Reprod Develop 53:230–243
Fresneda A, Lenis G, Agudelo E, Olivera-Angel M (2004) Espermiacio′n inducida y crioconservacio′n de semen de cachama blanca (Piaractus brachypomus). Rev Col Cienc Pec 17:46–52
Jamieson BGM (1991) Fish evolution and systematics from sperm. Cambridge University Press, Cambridge
Kurokura H, Hirano R, Tomita M, Iwahashi M (1984) Cryopreservation of carp sperm. Aquaculture 37:267–273
Labbe C, Crowe LM, Crowe JH (1997) Stability of the lipid component of trout sperm plasma membrane during Freeze–Thawing. Cryobiology 34:176–182
Lahnsteiner F, Berger B, Horvarth A, Urbanyi B, Weismann T (2000) Cryopreservation of spermatozoa in Cyprinid fishes. Theriogenology 54:1477–1498
Leung LKP (1991) Principles of biological cryopreservation. In: Jamieson BMG (ed) Fish evolution and systematics: evidence from spermatozoa. Cambridge University Press, Cambridge, pp. 231–244
Linhart O, Rodina M, Cosson J (2000) Cryopreservation of sperm in common carp Cyprinus carpio: sperm motility and hatching success of embryos. Cryobiology 41(3):241–250
Lubzens E, Daube N, Pekarsky I, Magnus Y, Cohen A, Yusefovich F, Feigin P (1997) Carp (Cyprinus carpio L.) spermatozoa cryobanks. Strategies in research and application. Aquaculture 155:13–30
Quinn PJ, Chow PY, White IG (1980) Evidence that phospholipid protects ram spermatozoa from cold shock at a plasma membrane site. J Reprod Fertil 60:403–407
Routray P, Verma DK, Sarkar SK, Sarangi N (2007) Recent advances in carp seed production and milt cryopreservation. Fish Physiol Biochem 33:413–427
Routray P, Dash SN, Dash C, Swain P, Sarkar SK, Sarangi N (2008) Cryopreservation of silver barb Puntius gonionotus (Bleeker) spermatozoa: effect of extender composition, cryoprotective agents and freezing rate on their postthawing fertilization ability. Aquacult Res 39:1597–1605
Rurangwa E, Roelants I, Huyskens G, Ebrahimi M, Kime DE, Ollevier F (1998) The minimum effective spermatozoa: egg ratio for artificial insemination and the effects of mercury on sperm motility and fertilisation ability inClarias gariepinus. J Fish Biol 53:402–413
Rurangwa E, Volckaert FAM, Huyskens G, Kime DE, Ollevier F (2001) Quality control of refrigerated and cryopreserved semen using computer-assisted sperm analysis (CASA), viable staining and standardized fertilisation in African catfish (Clarias gariepinus). Theriogenology 55:751–769
Scott AP, Baynes SM (1980) A review of the biology, handling and storage of salmonid spermatozoa. J Fish Biol 17:707–739
Suquet M, Dreanno C, Fauvel C, Cosson I, Billard R (2000) Cryopreservation of sperm in marine fish. Aquacult Res 31:231–243
Tekin N, Secer S, Akçay E, Bozkurt Y (2003) Cryopreservation of rainbow trout (Oncorhynchus mykiss) semen. Isr J Aquacult-Bamid 55(3):208–212
Tekin N, Secer S, Akcay E, Bozkurt Y, Kayam S (2007) Effects of glycerol additions on post-thaw fertility of frozen rainbow trout sperm, with an emphasis on interaction between extender and cryoprotectant. J Appl Ichthyol 23(1):60–63
Vuthiphandchai V, Zohar Y (1999) Age-related sperm quality of captive striped bass (Morone saxatilis). J World Aquacult Soc 30:65–72
Yavas I, Bozkurt Y (2011) Effect of different thawing rates on motility and fertilizing capacity of cryopreserved grass carp (Ctenopharyngodon idella) sperm. Biotechnol Biotechnol Equip 25(1):2254–2257
Acknowledgments
This research was funded by a grant from Scientific Research Institute of Mustafa Kemal University (MKU-08-E-0206). The authors would like to thank the staff of the State Hydraulic Works (SHW) Fish Production Station in Adana (Turkey) for their technical assistance. Also, the valuable comments and suggestions from anonymous reviewers are deeply thanked.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yavaş, İ., Bozkurt, Y. & Yıldız, C. Cryopreservation of scaly carp (Cyprinus carpio) sperm: effect of different cryoprotectant concentrations on post-thaw motility, fertilization and hatching success of embryos. Aquacult Int 22, 141–148 (2014). https://doi.org/10.1007/s10499-013-9698-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10499-013-9698-6