Abstract
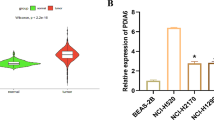
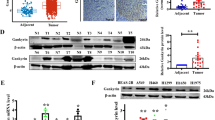
To investigate the role of SLITRK6 in lung adenocarcinoma (LUAD) and the underlying mechanism in it, clinical tissues and tissue microarray of LUAD were used to detect the expression of SLITRK6. In vitro cell viability assay and colony formation assay in LUAD cells were conducted to investigate SLITRK6 related biological functions. In vivo subcutaneous model was used to determine the role of SLITRK6 in LUAD growth. It was found that the expression of SLITRK6 was significantly upregulated in LUAD tissues compared with that in para-cancerous tissues. Knockdown of SLITRK6 suppressed the proliferation and colony formation of LUAD cells in vitro. Meanwhile, the growth of LUAD cells was also inhibited by SLITRK6 knockdown in vivo. Furthermore, we found that SLITRK6 knockdown could suppress the glycolysis of LUAD cells by regulating the phosphorylation of AKT and mTOR. All results suggest that SLITRK6 promotes LUAD cell proliferation and colony formation by regulating PI3K/AKT/mTOR signaling and Warburg effect. SLITRK6 may serve as a potential therapeutic target for LUAD in future.





Similar content being viewed by others
Data Availability
The datasets used or analysed during the current study are available from the corresponding author on reasonable request.
References
Sung H, Ferlay J, Siegel RL et al (2021) Global Cancer Statistics 2020: GLOBOCAN estimates of incidence and Mortality Worldwide for 36 cancers in 185 countries. CA Cancer J Clin 71(3):209–249. https://doi.org/10.3322/caac.21660
Melosky B, Juergens R, McLeod D et al (2019) Immune checkpoint-inhibitors and chemoradiation in stage III unresectable non-small cell lung cancer. Lung Cancer 134:259–267. https://doi.org/10.1016/j.lungcan.2019.05.027
Pinto R, Petriella D, Lacalamita R et al (2019) KRAS-Driven Lung Adenocarcinoma and B Cell Infiltration: Novel Insights for Immunotherapy. Cancers (Basel) 11(8). https://doi.org/10.3390/cancers11081145
Dziadziuszko R, Krebs MG, De Braud F et al (2021) Updated Integrated Analysis of the efficacy and safety of Entrectinib in locally Advanced or Metastatic ROS1 Fusion-Positive non-small-cell Lung Cancer. J Clin Oncol 39(11):1253–1263. https://doi.org/10.1200/jco.20.03025
König D, Savic Prince S, Rothschild SI (2021) Targeted therapy in Advanced and Metastatic Non-Small Cell Lung Cancer. An update on treatment of the most important actionable oncogenic driver alterations. Cancers (Basel) 13(4). https://doi.org/10.3390/cancers13040804
Nie M, Yao K, Zhu X et al (2021) Evolutionary metabolic landscape from preneoplasia to invasive lung adenocarcinoma. Nat Commun 12(1):6479. https://doi.org/10.1038/s41467-021-26685-y
Aruga J, Yokota N, Mikoshiba K (2003) Human SLITRK family genes: genomic organization and expression profiling in normal brain and brain tumor tissue. Gene 315:87–94. https://doi.org/10.1016/s0378-1119(03)00715-7
Aruga J (2003) Slitrk6 expression profile in the mouse embryo and its relationship to that of Nlrr3. Gene Expr Patterns 3(6):727–733. https://doi.org/10.1016/s1567-133x(03)00141-8
Proenca CC, Gao KP, Shmelkov SV, Rafii S, Lee FS (2011) Slitrks as emerging candidate genes involved in neuropsychiatric disorders. Trends Neurosci 34(3):143–153. https://doi.org/10.1016/j.tins.2011.01.001
Aruga J, Mikoshiba K (2003) Identification and characterization of Slitrk, a novel neuronal transmembrane protein family controlling neurite outgrowth. Mol Cell Neurosci 24(1):117–129. https://doi.org/10.1016/s1044-7431(03)00129-5
Beaubien F, Cloutier JF (2009) Differential expression of Slitrk family members in the mouse nervous system. Dev Dyn 238(12):3285–3296. https://doi.org/10.1002/dvdy.22160
Liu X, Liu Y, Liu Z et al (2021) Identification of SLITRK6 as a Novel Biomarker in hepatocellular carcinoma by comprehensive bioinformatic analysis. Biochem Biophys Rep 28:101157. https://doi.org/10.1016/j.bbrep.2021.101157
Morrison K, Challita-Eid PM, Raitano A et al (2016) Development of ASG-15ME, a novel antibody-drug Conjugate Targeting SLITRK6, a New Urothelial Cancer Biomarker. Mol Cancer Ther 15(6):1301–1310. https://doi.org/10.1158/1535-7163.Mct-15-0570
Akram M (2013) Mini-review on glycolysis and cancer. J Cancer Educ 28(3):454–457. https://doi.org/10.1007/s13187-013-0486-9
Ganapathy-Kanniappan S, Geschwind JF (2013) Tumor glycolysis as a target for cancer therapy: progress and prospects. Mol Cancer 12:152. https://doi.org/10.1186/1476-4598-12-152
Amoêdo ND, Valencia JP, Rodrigues MF, Galina A, Rumjanek FD (2013) How does the metabolism of tumour cells differ from that of normal cells. Biosci Rep 33(6). https://doi.org/10.1042/bsr20130066
Lu H, Forbes RA, Verma A (2002) Hypoxia-inducible factor 1 activation by aerobic glycolysis implicates the Warburg effect in carcinogenesis. J Biol Chem 277(26):23111–23115. https://doi.org/10.1074/jbc.M202487200
Schwickert G, Walenta S, Sundfør K, Rofstad EK, Mueller-Klieser W (1995) Correlation of high lactate levels in human cervical cancer with incidence of metastasis. Cancer Res 55(21):4757–4759
Xiao Y, Peng H, Hong C et al (2017) PDGF promotes the Warburg Effect in Pulmonary arterial smooth muscle cells via activation of the PI3K/AKT/mTOR/HIF-1α signaling pathway. Cell Physiol Biochem 42(4):1603–1613. https://doi.org/10.1159/000479401
Charbonnier LM, Cui Y, Stephen-Victor E et al (2019) Functional reprogramming of regulatory T cells in the absence of Foxp3. Nat Immunol 20(9):1208–1219. https://doi.org/10.1038/s41590-019-0442-x
Mossmann D, Park S, Hall MN (2018) mTOR signalling and cellular metabolism are mutual determinants in cancer. Nat Rev Cancer 18(12):744–757. https://doi.org/10.1038/s41568-018-0074-8
Patterson RE, Sears DD (2017) Metabolic Effects of Intermittent Fasting. Annu Rev Nutr 37:371–393. https://doi.org/10.1146/annurev-nutr-071816-064634
Weng ML, Chen WK, Chen XY et al (2020) Fasting inhibits aerobic glycolysis and proliferation in colorectal cancer via the Fdft1-mediated AKT/mTOR/HIF1α pathway suppression. Nat Commun 11(1):1869. https://doi.org/10.1038/s41467-020-15795-8
Acknowledgements
None.
Funding
None.
Author information
Authors and Affiliations
Contributions
F.Y.Y. and M.M. designed the study. F.Y.Y. and X.Y.Z. performed the experiments and analysis of the data. M.T.L. participated in parts of the experiments. M.M. wrote the paper. All authors read and approved the final manuscript. The authors declare that all data were generated in-house and that no paper mill was used.
Corresponding author
Ethics declarations
Ethical approval and consent to participate
The study was approved by the Ethics Committee of the Shandong First Medical University. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. All human samples were obtained with informed consent.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yu, F., Zhao, X., Li, M. et al. SLITRK6 promotes the progression of lung adenocarcinoma by regulating PI3K/AKT/mTOR signaling and Warburg effect. Apoptosis 28, 1216–1225 (2023). https://doi.org/10.1007/s10495-023-01838-0
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10495-023-01838-0