Abstract
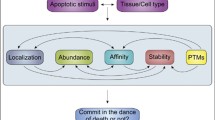
Permeabilization of the outer mitochondrial membrane is the point of no return in most programmed cell deaths. This critical step is mainly regulated by the various protein-protein and protein-membrane interactions of the Bcl-2 family proteins. The two main models for regulation of mitochondrial outer membrane permeabilization, direct activation and displacement do not account for all of the experimental data and both largely neglect the importance of the membrane. We propose the embedding together model to emphasize the critical importance of Bcl-2 family protein interactions with and within membranes. The embedding together model proposes that both pro- and anti-apoptotic Bcl-2 family proteins engage in similar dynamic interactions that are governed by membrane dependent conformational changes and culminate in either aborted or productive membrane permeabilization depending on the final oligomeric state of pro-apoptotic Bax and/or Bak.
Article PDF
Similar content being viewed by others
References
Letai A, Bassik MC, Walensky LD et al (2002) Distinct BH3 domains either sensitize or activate mitochondrial apoptosis, serving as prototype cancer therapeutics. Cancer Cell 2:183–192
Kuwana T, Bouchier-Hayes L, Chipuk JE et al (2005) BH3 domains of BH3-only proteins differentially regulate Bax-mediated mitochondrial membrane permeabilization both directly and indirectly. Mol Cell 17:525–535
Certo M, Del Gaizo Moore V, Nishino N et al (2006) Mitochondria primed by death signals determine cellular addiction to antiapoptotic BCL-2 family members. Cancer Cell 9:351–365
Wei MC, Zong WX, Cheng EH et al (2001) Proapoptotic BAX and BAK: a requisite gateway to mitochondrial dysfunction and death. Science 292:727–730
Cartron PF, Gallenne T, Bougras B et al (2004) The first alpha helix of Bax plays a necessary role in its ligand-induced activation by the BH3-only proteins Bid and PUMA. Mol Cell 16:807– 818
Walensky LD, Kung AL, Escher I et al (2004) Activation of apoptosis in vivo by a hydrocarbon-stapled BH3 helix. Science 305:1466–1470
Kuwana T, Mackey MR, Perkins G et al (2002) Bid, Bax, and lipids cooperate to form supramolecular openings in the outer mitochondrial membrane. Cell 111:331–342
Cartron PF, Arokium H, Oliver L et al (2005) Distinct domains control the addressing and the insertion of Bax into mitochondria. J Biol Chem 280:10587–10598
Terrones O, Antonsonn B, Yamaguchi H et al (2004) Lipidic pore formation by the concerted action of proapoptotic BAX and tBID. J Biol Chem 279:30081–30091
Annis MG, Soucie EL, Dlugosz PJ et al (2005) Bax forms multispanning monomers that oligomerize to permeabilize membranes during apoptosis. EMBO J 24:2096–2103
Dlugosz PJ, Billen LP, Annis MG et al (2006) Bcl-2 changes conformation to inhibit Bax oligomerization. EMBO J 25:2287–2296
Yethon JA, Epand R, Leber B et al (2003) Interaction with a membrane surface triggers a reversible conformational change in Bax normally associated with induction of apoptosis. J Biol Chem 278:48935–48941
Desagher S, Osen-Sand A, Nichols A et al (1999) Bid-induced conformational change of Bax is responsible for mitochondrial cytochrome c release during apoptosis. J Cell Biol 144:891– 901
Eskes R, Desagher S, Antonsonn B et al (2000) Bid induces the oligomerization and insertion of Bax into the outer mitochondrial membrane. Mol Cell Biol 20:929–935
Wei MC, Lindsten T, Mootha VK et al (2000) tBID, a membrane-targeted death ligand, oligomerizes BAK to release cytochrome c. Genes Dev 14:2060–2071
Cheng EH, Wei MC, Weiler S et al (2001) BCL-2, BCL-X(L) sequester BH3 domain-only molecules preventing BAX- and BAK-mediated mitochondrial apoptosis. Mol Cell 8:705–711
Terradillos O, Montessuit S, Huang DC et al (2002) Direct addition of BimL to mitochondria does not lead to cytochrome c release. FEBS Lett 522:29–34
Marani M, Tenev T, Hancock D et al (2002) Identification of novel isoforms of the BH3 domain protein Bim which directly activate Bax to trigger apoptosis. Mol Cell Biol 22:3577–3589
Harada H, Quearry B, Ruiz-Vela A et al (2004) Survival factor-induced extracellular signal-regulated kinase phosphorylates BIM, inhibiting its association with BAX and proapoptotic activity. Proc Natl Acad Sci USA 101:15313–15317
Suzuki M, Youle RJ, Tjandra N (2000) Structure of Bax: coregulation of dimer formation and intracellular localization. Cell 103:645–654
Sattler M, Liang H, Nettesheim D et al (1997) Structure of Bcl-xL-Bak peptide complex: recognition between regulators of apoptosis. Science 275:983–986
Zha J, Wewiler S, Oh KJ et al (2000) Posttranslational N-myristoylation of BID as a molecular switch for targeting mitochondria and apoptosis. Science 290:1761–1765
Hu X, Han Z, Wyche JH et al (2003) Helix 6 of tBid is necessary but not sufficient for mitochondrial binding activity. Apoptosis 8:277–289
Kim TH, Zhao Y, Ding WX et al (2004) Bid-cardiolipin interaction at mitochondrial contact site contributes to mitochondrial cristae reorganization and cytochrome c release. Mol Biol Cell 15:3061–3072
Gong XM, Choi J, Franzin CM et al (2004) Conformation of membrane-associated proapoptotic tBid. J Biol Chem 279:28954–28960
Oh KJ, Barbuto S, Meyer N et al (2005) Conformational changes in BID, a pro-apoptotic BCL-2 family member, upon membrane binding. A site-directed spin labeling study. J Biol Chem 280:753–767
Oh KJ, Barbuto S, Pitter K et al (2006) A membrane-targeted BID BH3 peptide is sufficient for high potency activation of BAX in vitro. J Biol Chem 281:36999–37008
Grinberg M, Sarig R, Zaltsman Y et al (2002) BID Homooligomerizes in the mitochondrial membrane to induce apoptosis. J Biol Chem 277:12237–12245
Chen L, Willis SN, Wei A et al (2005) Differential targeting of prosurvival Bcl-2 proteins by their BH3-only ligands allows complementary apoptotic function. Mol Cell 17:393–403
Wang K, Yin XM, Chao DT et al (1996) BID: a novel BH3 domain-only death agonist. Genes Dev 10:2859–2869
Ruffolo SC, Shore GC (2003) BCL-2 selectively interacts with the BID-induced open conformer of BAK, inhibiting BAK auto-oligomerization. J Biol Chem 278:25039–25045
Tan C, Dlugosz PJ, Peng J et al (2006) Auto-activation of the apoptosis protein Bax increases mitochondrial membrane permeability and is inhibited by Bcl-2. J Biol Chem 281:14764–14775
Yamaguchi H, Wang HG (2002) Bcl-XL protects BimEL-induced Bax conformational change and cytochrome c release independent of interacting with Bax or BimEL. J Biol Chem 277:41604–41612
Mikhailov V, Mikhailova M, Degenhardt K et al (2003) Association of Bax and Bak homo-oligomers in mitochondria. Bax requirement for Bak reorganization and cytochrome c release. J Biol Chem 278:5367–5376
Pierrat B, Simonen M, Cueto M et al (2001) SH3GLB, a new endophilin-related protein family featuring an SH3 domain. Genomics 71:222–234
Takahashi Y, Karbowski M, Yamaguchi H et al (2005) Loss of Bif-1 suppresses Bax/Bak conformational change and mitochondrial apoptosis. Mol Cell Biol 25:9369–9382
Willis SN, Chen L, Dewson G et al (2005) Proapoptotic Bak is sequestered by Mcl-1 and Bcl-xL, but not Bcl-2, until displaced by BH3-only proteins. Genes Dev 19:1294–1305
Wilson-Annan J, O’Reilly LA, Crawford SA et al (2003) Proapoptotic BH3-only proteins trigger membrane integration of prosurvival Bcl-w and neutralize its activity. J Cell Biol 162:877–887
Cuconati A, Mukherjee C, Perez D et al (2003) DNA damage response and MCL-1 destruction initiate apoptosis in adenovirus-infected cells. Genes Dev 17:2922–2932
Leu JI, Dumont P, Hafey M et al (2004) Mitochondrial p53 activates Bak and causes disruption of a Bak-Mcl1 complex. Nat Cell Biol 6:443–450
Cheng EH, Sheiko TV, Fisher JK et al (2003) VDAC2 inhibits BAK activation and mitochondrial apoptosis. Science 301:513–517
Oltvai ZN, Milliman CL, Korsmeyer SJ et al (1993) Bcl-2 heterodimerizes in vivo with a conserved homolog, Bax, that accelerates programmed cell death. Cell 74:609–619
Hsu YT, Youle RJ (1997) Nonionic detergents induce dimerization among members of the Bcl-2 family. J Biol Chem 272:13829–13834
Hsu YT, Youle RJ (1998) Bax in murine thymus is a soluble monomeric protein that displays differential detergent-induced conformations. J Biol Chem 273:10777–10783
Antonsson B, Montessuit S, Sanchez B et al (2001) Bax is present as a high molecular weight oligomer/complex in the mitochondrial membrane of apoptotic cells. J Biol Chem 276:11615–11623
Yang E, Zha J, Jockel J et al (1995) Bad, a heterodimeric partner for Bcl-XL and Bcl-2, displaces Bax and promotes cell death. Cell 80:285–291
Letai A, Sorcinelli MD, Beard C et al (2004) Antiapoptotic BCL-2 is required for maintenance of a model leukemia. Cancer Cell 6:241–249
Nijhawan D, Fang M, Traer E et al (2003) Elimination of Mcl-1 is required for the initiation of apoptosis following ultraviolet irradiation. Genes Dev 17:1475–1486
Zhong O, Gao W, Dhu F et al (2005) Mule/ARF-BP1, a BH3-only E3 ubiquitin ligase, catalyzes the polyubiquitination of Mcl-1 and regulates apoptosis. Cell 121:1085–1095
Warr MR, Acoca S, Liu Z et al (2005) BH3-ligand regulates access of MCL-1 to its E3 ligase. FEBS Lett 579:5603–5608
Jeong SY, Gaume B, Lee YJ et al (2004) Bcl-x(L) sequesters its C-terminal membrane anchor in soluble, cytosolic homodimers. EMBO J 23:2146–2155
Wang K, Yin XM, Chao DT et al (1996) BID: a novel BH3 domain-only death agonist. Genes Dev 10:2859–2869
Peng J, Tan C, Roberts GJ et al (2006) tBid elicits a conformational alteration in membrane-bound Bcl-2 such that it inhibits bax pore formation. J Biol Chem 281:35802–35811
Sharpe JC, Arnoult D, Youle RJ (2004) Control of mitochondrial permeability by Bcl-2 family members. Biochim Biophys Acta 1644:107–113
Zhang H, Kim JK, Edwards CA et al (2005) Clusterin inhibits apoptosis by interacting with activated Bax. Nat Cell Biol 7:909–915
Cuddeback SM, Yamaguchi H, Komatsu K et al (2001) Molecular cloning and characterization of Bif-1. A novel Src homology 3 domain-containing protein that associates with Bax. J Biol Chem 276:20559–20565
Schinzel A, Kaufmann T, Borner C (2004) Bcl-2 family members: integrators of survival and death signals in physiology and pathology. Biochim Biophys Acta 1644:95–105
Tan KO, Tan KM, Chan SL et al (2001) MAP-1, a novel proapoptotic protein containing a BH3-like motif that associates with Bax through its Bcl-2 homology domains. J Biol Chem 276:2802–2807
Tan KO, Fu NY, Sukumaran SK et al (2005) MAP-1 is a mitochondrial effector of Bax. Proc Natl Acad Sci USA 102:14623–14628
Ohtsuka T, Ryu H, Minamishima YA et al (2004) ASC is a Bax adaptor and regulates the p53-Bax mitochondrial apoptosis pathway. Nat Cell Biol 6:121–128
Dumont P, Leu JI, Della Pietra AC et al (2003) The codon 72 polymorphic variants of p53 have markedly different apoptotic potential. Nat Genet 33:357–365
Chipuk JE, Kuwana T, Bouchier-Hayes L et al (2004) Direct activation of Bax by p53 mediates mitochondrial membrane permeabilization and apoptosis. Science 303:1010–1014
Leu JI, Dumont P, Hafey M et al (2004) Mitochondrial p53 activates Bak and causes disruption of a Bak-Mcl1 complex. Nat Cell Biol 6:443–450
Perfettini JL, Kroemer RT, Kroemer G (2004) Fatal liaisons of p53 with Bax and Bak. Nat Cell Biol 6:386–388
Park BS, Song YS, Yee SB et al (2005) Phospho-ser 15-p53 translocates into mitochondria and interacts with Bcl-2 and Bcl-xL in eugenol-induced apoptosis. Apoptosis 10:193–200
Roucou X, Rostotseva T, Montessuit S et al (2002) Bid induces cytochrome c-impermeable Bax channels in liposomes. Biochem J 363:547–552
Goldmacher VS, Bartle LM, Skaletskaya A et al (1999) A cytomegalovirus-encoded mitochondria-localized inhibitor of apoptosis structurally unrelated to Bcl-2. Proc Natl Acad Sci USA 96:12536–12541
Poncet D, Pauleau AL, Szabadkai G et al (2006) Cytopathic effects of the cytomegalovirus-encoded apoptosis inhibitory protein vMIA. J Cell Biol 174:985–996
Arnoult D, Bartle LM, Skaletskaya A et al (2004) Cytomegalovirus cell death suppressor vMIA blocks Bax- but not Bak-mediated apoptosis by binding and sequestering Bax at mitochondria. Proc Natl Acad Sci USA 101:7988–7993
Wasilenko ST, Banadyga L, Bond D et al (2005) The vaccinia virus F1L protein interacts with the proapoptotic protein Bak and inhibits Bak activation. J Virol 79:14031–14043
Postigo A, Cross JR, Downward J et al (2006) Interaction of F1L with the BH3 domain of Bak is responsible for inhibiting vaccinia-induced apoptosis. Cell Death Differ 13:1651–1662
Kim PK, Annis MG, Dlugosz PJ et al (2004) During apoptosis Bcl-2 changes membrane topology at both the endoplasmic reticulum and mitochondria. Mol Cell 14:523–529
Korsmeyer SJ, Gross A, Harada H et al (1999) Death and survival signals determine active/inactive conformations of pro-apoptotic BAX, BAD, and BID molecules. Cold Spring Harb Symp Quant Biol 64:343–350
Schendel SL, Montal M, Redd JC (1998) Bcl-2 family proteins as ion-channels. Cell Death Differ 5:372–380
Cheng EH, Kirsch DG, Clem RJ et al (1997) Conversion of Bcl-2 to a Bax-like death effector by caspases. Science 278:1966–1968
Clem RJ, Cheng EH, Karp CL et al (1998) Modulation of cell death by Bcl-XL through caspase interaction. Proc Natl Acad Sci USA 95:554–559
Lin B, Kolluri SK, Lin F et al (2004) Conversion of Bcl-2 from protector to killer by interaction with nuclear orphan receptor Nur77/TR3. Cell 116:527–540
Delavani P, Adrain C, Taylor RC et al (2006) Role for CED-9 and Egl-1 as regulators of mitochondrial fission and fusion dynamics. Mol Cell 21:761–773
Aouacheria A, Brunet F, Gouy M (2005) Phylogenomics of life-or-death switches in multicellular animals: Bcl-2, BH3-Only, and BNip families of apoptotic regulators. Mol Biol Evol 22:2395–2416
Fiebig A, Zhu W, Hollerbach C et al (2006) Bcl-XL is qualitatively different from and ten times more effective than Bcl-2 when expressed in a breast cancer cell line. BMC Cancer 6: 213
Goldsmith KC, Liu X, Dam V et al (2006) BH3 peptidomimetics potently activate apoptosis and demonstrate single agent efficacy in neuroblastoma. Oncogene 25:4525–4533
Zhang Z, Lapolla SM, Annis MG et al (2004) Bcl-2 homodimerization involves two distinct binding surfaces, a topographic arrangement that provides an effective mechanism for Bcl-2 to capture activated Bax. J Biol Chem 279:43920–43928
Lazebnik Y (2002) Can a biologist fix a radio?–Or, what I learned while studying apoptosis. Cancer Cell 2:179–182
Kerr JF, Wylie AH, Currie AR (1972) Apoptosis: a basic biological phenomenon with wide-ranging implications in tissue kinetics. Br J Cancer 26:239–257
Janes KA, Albeck JG, Gaudet S et al (2005) A systems model of signaling identifies a molecular basis set for cytokine-induced apoptosis. Science 310:1646–1653
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Leber, B., Lin, J. & Andrews, D.W. Embedded together: The life and death consequences of interaction of the Bcl-2 family with membranes. Apoptosis 12, 897–911 (2007). https://doi.org/10.1007/s10495-007-0746-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10495-007-0746-4