Abstract
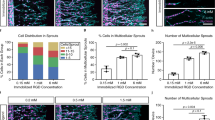
Induction of an inter-connected microvessel network in a tissue-engineered construct prior to implantion may be an alternative to improve the success rate of cell/tissue survival and wound integration. Conditions of endothelial cell-seeding density and distribution were investigated in two 3-D angiogenesis culture systems. Endothelial cells were either seeded dispersed in a fibrin gel, or subconfluent on micro-beads (Cytodex®) prior to being embedded in fibrin. Human fibroblasts and growth factors were introduced to optimize angiogenesis. A density higher than 4 × 104 cells/ml of fibrin was necessary to induce angiogenic-like structures (i.e., sprouting, cord-, lumen-like structures) by 14 days in the dispersed cell model. Endothelial cells on micro-beads also exhibited angiogenic-like structures that were inter-connected to those on neighboring beads. The sizes of the angiogenic-like structures were larger on beads compared to those found in the dispersed cell model. High cell density was needed in angiogenesis when cells were seeded separately, whereas the association of endothelial cells on bead surfaces significantly reduced the cell density used. Moreover, increasing bead density was not necessary to facilitate further angiogenic formation. Micro-spheres may represent a potential support for endothelial cells in microvessel networking, with subsequent applications in the pre-vascularization of bio-implants.







Similar content being viewed by others
References
Carmeliet P (2000) Mechanism of angiogenesis and arteriogenesis. Nat Med 6:389–395
Bergers G, Benjamin LE (2003) Tumorigenesis and the angiogenic switch. Nat Rev 3:401–410
Kannan RY, Salacinski HJ, Sales K et al (2005) The roles of tissue engineering and vascularisation in the development of micro-vascular networks: a review. Biomaterials 26:1857–1875
Ito K, Yamada Y, Nagasaka T et al (2005) Osteogenic potential of injectable tissue-engineered bone: a comparison among autogenous bone, bone substitute (Bio-oss), platelet-rich plasma, and tissue-engineered bone with respect to their mechanical properties and histological findings. J Biomed Mater Res A 73:63–72
Richardson TP, Peters MC, Ennett AB (2001) Polymeric system for dual growth factor delivery. Nat Biotechnol 19:1029–1034
Soejima N, Negishi N, Nozaki M et al (1998) Effect of cultured endothelial cells on angiogenesis in vivo. Plast Reconstr Surg 101:1552–1560
Schechner JS, Crane SK, Wang F et al (2003) Engraftment of a vascularised human skin equivalent. FASEB J 17:2250–2256
Breier G, Risau W (1996) The role of vascular endothelial growth factor in blood vessel formation. Trends Cell Biol 6:454–456
Folkman J, Haudenschild C (1980) Angiogenesis in vitro. Nature 288:551–556
Haralabopoulos GC, Grant DS, Kleinman HK et al (1997) Thrombin promotes endothelial cell alignment in Matrigel in vitro and angiogenesis in vivo. Am J Physiol 273:C239–C245
Vailhé B, Vittet D, Feige JJ (2001) In vitro models of vasculogenesis and angiogenesis. Lab Invest 81:439–452
Davis GE, Black SM, Bayless KJ (2000) Capillary morphogenesis during human endothelial cell invasion of three-dimensional collagen matrices. In vitro Cell Dev Biol Anim 36:513–519
Gagnon E, Cattaruzzi P, Griffith M et al (2002) Human vascular endothelial cells with life spans: in vitro cell response, protein expression, and angiogenesis. Angiogenesis 5:21–33
Nehls V, Drenckhahn D (1995) A novel, microcarrier-based in vitro assay for rapid and reliable quantification of three-dimensional cell migration and angiogenesis. Microvasc Res 50:311–322
Nakatsu MN, Sainson RCA, Aoto JN et al (2003) Angiogenic sprouting and capillary lumen formation modeled by human umbilical vein endothelial cells (HUVECs) in fibrin gels: the role of fibroblasts and Angiopoietin-1. Micro Res 66:102–112
Jaffe EA, Nachman RL, Becker CG et al (1973) Culture of human endothelial cells derived from umbilical veins. Identification by morphologic and immunologic criteria. J Clin Invest 52:2745–2756
Griffith CK, Miller C, Sainson RC et al (2005) Diffusion limits of an in vitro thick prevascularized tissue. Tissue Eng 11:257–266
Kunz-Schughart LA, Schroeder JA, Wondrak M et al (2006) Potential of fibroblasts to regulate the formation of three-dimensional vessel-like structures from endothelial cells in vitro. Am J Physiol Cell Physiol 290:C1385–C1398
Wenger A, Kowalewski N, Stahl A et al (2005) Development and characterization of a spheroidal coculture model of endothelial cells and fibroblasts for improving angiogenesis in tissue engineering. Cells Tissues Organs 181: 80–88
Pinney E, Liu K, Sheeman B, Mansbridg J (2000) Human three-dimensional fibroblast cultures express angiogenic activity. J Cell Physiol 183:74–82
Korff T, Kimmina S, Martiny-Baron G et al (2001) Blood vessel maturation in a 3-dimensional spheroidal coculture model: direct contact with smooth muscle cells regulates endothelial cell quiescence and abrogates VEGF responsiveness. FASEB J 15:447–457
Seghezzi G, Patel S, Ren CJ et al (1998) Fibroblast growth factor-2 (FGF-2) induces vascular endothelial growth factor (VEGF) expression in the endothelial cells of forming capillaries: an autocrine mechanism contributing to angiogenesis. J Cell Biol 141:1659–1673
Villaschi S, Nicosia RF (1993) Angiogenic role of endogenous basic fibroblast growth factor released by rat aorta after injury. Am J Pathol 143:181–190
Xue L, Greisler HP (2002) Angiogenic effect of fibroblast growth factor-1 and vascular endothelial growth factor and their synergism in a novel in vitro quantitative fibrin-based 3-dimensional angiogenesis system. Surgery 132:259–267
Collen A, Koolwijk P, Kroon M, van Hinsbergh VW (1998) Influence of fibrin structure on the formation and maintenance of capillary-like tubules by human microvascular endothelial cells. Angiogenesis 2:153–165
Davis GE, Camarillo CW (1996) An alpha 2 beta 1 integrin-dependent pinocytic mechanism involving intracellular vacuole formation and coalescence regulates capillary lumen and tube formation in three-dimensional collagen matrix. Exp Cell Res 224:39–51
Ilan N, Mahooti S, Madri JA (1998) Distinct signal transduction pathways are utilized during the tube formation and survival phases of in vitro angiogenesis. J Cell Sci 111:3621–3631
Sun MB, Jiang YJ, Li WD, Li PZ, Li GL, Jiang SD, Liao GY (2004) A novel process for production of hepatitis A virus in Vero cells grown on microcarriers in bioreactor. World J Gastroenterol 10: 2571–2573
Finney MR, Greco NJ, Haynesworth SE et al (2006) Direct comparison of umbilical cord blood versus bone marrow-derived endothelial precursor cells in mediating neovascularization in response to vascular ischemia. Biol Blood Marrow Transplant 12:585–593
Acknowledgments
We are thankful to Mrs. Renée Paradis for technical assistance and Catherine Gérard for scientific advice. LM is the recipient of a studentship from the Fonds de recherche sur la nature et les technologies (FQRNT). This work was supported by the Natural Sciences and Engineering Research Council of Canada (NSERC, strategic project; CJD).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Martineau, L., Doillon, C.J. Angiogenic response of endothelial cells seeded dispersed versus on beads in fibrin gels. Angiogenesis 10, 269–277 (2007). https://doi.org/10.1007/s10456-007-9079-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10456-007-9079-8