Summary
Background
We investigated whether a minimal dose of FK506 is sufficient to inhibit the rejection of venous allografts and facilitate allograft adaptation to arterialisation in rats.
Methods
Iliolumbar veins from Brown Norway (BN) rats were transplanted into the abdominal aorta of Lewis (LEW) rats. Recipient animals received daily intramuscular injections of 0.1 mg kg− 1 FK506. Light microscope evaluations of grafts were performed 30 days after transplantation. We investigated the presence of endothelial cells, the intensity of intimal proliferation, the deposition of immunoglobulin G molecules into the medial layer and the degree of adventitial infiltration by Lewis MHC class II-positive, CD4-positive and CD8-positive cells. All results were compared to previously reported data for syngeneic, allogeneic non-immunosuppressed and allogeneic FK506-low-dose immunosuppressed animals.
Results
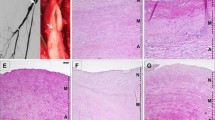
All alloveins under minimal FK506 immunosuppression showed typical histological signs of arterialisation 30 days after transplantation. Intimal thickness (15.1 ± 6.1 mm) and the degree of adventitial infiltration by MHC class II-positive (16.96 ± 10.46), CD8-positive (2.63 ± 2.6), and CD4-positive (7.67 ± 8.29) cells were not significantly different when compared to untreated syngeneic veins and allogeneic veins treated with a low dose of FK506, respectively.
Conclusions
Even the minimal dose of FK506 was sufficient to inhibit the acute rejection of venous allografts and to facilitate allograft arterialisation 30 days after transplantation.


Similar content being viewed by others
References
Management of peripheral arterial disease (PAD). TransAtlantic Inter-Society Consensus (TASC). Eur J Vasc Endovasc Surg. 2000;19 Suppl A:Si-xxviii, S1–250.
Randon C, Jacobs B, De Ryck F, Beele H, Vermassen F. Fifteen years of infrapopliteal arterial reconstructions with cryopreserved venous allografts for limb salvage. J Vasc Surg. 2010;51(4):869–77.
Matia I, Janousek L, Marada T, Adamec M. Cold-stored venous allografts in the treatment of critical limb ischaemia. Eur J Vasc Endovasc Surg. 2007;34(4):424–31.
Brockbank KG, McNally RT, Walsh KA. Cryopreserved vein transplantation. J Card Surg. 1992;7(2):170–6.
Posner MP, Makhoul RG, Altman M, Kimball P, Cohen N, Sobel M, et al. Early results of infrageniculate arterial reconstruction using cryopreserved homograft saphenous conduit (CADVEIN) and combination low-dose systemic immunosuppression. J Am Coll Surg. 1996;183(3):208–16.
Matia I, Lodererova A, Adamec M. Delayed administration of FK 506 is sufficient to suppress acute rejection changes after aortal transplantation in rats. Transpl Int. 2007;20(4):371–80.
Matia I, Varga M, Lodererova A, Adamec M. The positive effect of immunosuppression on adaptation of venous allografts to arterialisation in rats. Eur J Vasc Endovasc Surg. 2010;39(4):478–84.
Azuma N, Sasajima T, Kubo Y. Immunosuppression with FK506 in rat arterial allografts: fate of allogeneic endothelial cells. J Vasc Surg. 1999;29(4):694–702.
Kwei S, Stavrakis G, Takahas M, Taylor G, Folkman MJ, Gimbrone MA Jr, et al. Early adaptive responses of the vascular wall during venous arterialization in mice. Am J Pathol. 2004;164(1):81–9.
Westerband A, Crouse D, Richter LC, Aguirre ML, Wixon CC, James DC, et al. Vein adaptation to arterialization in an experimental model. J Vasc Surg. 2001;33(3):561–9.
Gibbons GH, Dzau VJ. The emerging concept of vascular remodeling. N Engl J Med. 1994;330(20):1431–8.
Axthelm SC, Porter JM, Strickland S, Baur GM. Antigenicity of venous allografts. Ann Surg. 1979;189(3):290–3.
Wagner E, Roy R, Marois Y, Douville Y, Guidoin R. Posttransplant antibodies and fresh venous allograft failure in dogs. Transplantation. 1994;58(5):537–42.
Stevens SL, Tyler JD, Freeman MB, Hopkins F, Lewis T, Bray J, et al. Factors affecting patency of venous allografts in miniature swine. J Vasc Surg. 1990;12(3):361–6.
Wagner E, Roy R, Marois Y, Douville Y, Guidoin R. Fresh venous allografts in peripheral arterial reconstruction in dogs. Effects of histocompatibility and of short-term immunosuppression with cyclosporine A and mycophenolate mofetil. J Thorac Cardiovasc Surg. 1995;110(6):1732–44.
Carpenter EW, Lindenauer SM. Immunosuppression in arterial and venous allografts. Arch Surg. 1973;106(1):77–8.
Mingoli A, Edwards JD, Feldhaus RJ, Hunter WJ 3rd, Naspetti R, Cavallari N, et al. Fresh vein allograft survival in dogs after cyclosporine treatment. J Surg Res. 1996;62(1):95–102.
Perloff LJ, Reckard CR, Rowlands DT Jr, Barker CF. The venous homograft: an immunological question. Surgery. 1972;72(6):961–70.
Vermassen F, Degrieck N, De Kock L, Goubeau J, Van Landuyt K, Noens L, et al. Immunosuppressive treatment of venous allografts. Eur J Vasc Surg. 1991;5(6):669–75.
Ricotta JJ, Collins GJ Jr, Rich NM, Reynolds DG. Failure of immunosuppression to prolong venous allograft survival. Arch Surg. 1980;115(1):99–101.
Bandlien KO, Toledo-Pereyra LH, MacKenzie GH, Choudhury SP, Cortez JA. Immunosuppression with cyclosporine. A new approach to improve patency of venous allografts. Arch Surg. 1983;118(7):829–33.
Zoja C, Furci L, Ghilardi F, Zilio P, Benigni A, Remuzzi G. Cyclosporin-induced endothelial cell injury. Lab Invest. 1986;55(4):455–62.
Khanna A, Kapur S, Sharma V, Li B, Suthanthiran M. In vivo hyperexpression of transforming growth factor-beta1 in mice: stimulation by cyclosporine. Transplantation. 1997;63(7):1037–9.
Miller VM, Bergman RT, Gloviczki P, Brockbank KG. Cryopreserved venous allografts: effects of immunosuppression and antiplatelet therapy on patency and function. J Vasc Surg. 1993;18(2):216–26.
Vincenti F, Jensik SC, Filo RS, Miller J, Pirsch J. A long-term comparison of tacrolimus (FK506) and cyclosporine in kidney transplantation: evidence for improved allograft survival at five years. Transplantation. 2002;73(5):775–82.
Conflict of interest
The authors declare that there is no actual or potential conflicts of interest in relation to this article.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Varga, M., Matia, I., Lodererova, A. et al. The minimal dose of FK506 is sufficient to facilitate the arterialisation of venous allografts in rats. Eur Surg 45, 8–14 (2013). https://doi.org/10.1007/s10353-012-0179-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10353-012-0179-3