Abstract
A chronosequence approach was applied to evaluate the effects of silver birch succession on soil organic matter (SOM). The studies covered five plots (arable field [AF], fallow [FA] and three birch stands aged 8, 33 and 40 years), and all located in the same complex of Brunic Arenosols in central Poland. The O and A horizons of the soils were sampled and analysed using the standard procedures in soil science. It was found that the birch succession was accompanied by dynamic changes in the stock and quality of the SOM. The transition from AF to FA soils did not significantly change the content or pool of SOM in the A horizon; however, it did modify its fractional composition and internal structure. This was reflected by an increase in the humic acids content and a decrease in the fulvic acid, as well as in the values of the elemental and 13C-NMR analyses. Rapid losses in the SOM content and stock in the A horizon were recorded after entering the silver birch. The soils under the young birches were characterised by lower contents of low-molecular humus fractions, which are more susceptible to microbial degradation, than the FA soils. In addition, the birch succession contributed to a lower internal oxidation of the humic acids and an increase in the content of aliphatic structures in their molecules. The next stages of succession were characterised by gradually increasing SOM contents and stocks, accompanied by the increasing internal oxidation and maturity of the humic acids.
Similar content being viewed by others
Introduction
The growing costs of production, the decreasing quality and productivity of soils due to intensive agricultural use, and/or their contamination with various substances constitutes major reasons for the abandonment of agricultural areas in many regions, including Europe (Kurganova et al. 2014). Afforestation has commonly been used to protect abandoned soils against further negative changes (Kilian 1998). Moreover, the practice can also be applied for carbon sequestration (Paul et al. 2002; Miripanah et al. 2019) and to increase the potential to provide various ecosystem services (Pèrez-Silos et al. 2021). The spontaneous development of vegetation can also be observed on non-afforested areas. This is a multi-stage process that follows a typical pattern of species replacement. Annual plants are typical of early colonisation, and these being replaced by perennial species in the next stages (Corbet 1995; Wilcox 1998). The process of succession is controlled by various ecological mechanisms that are involved in three alterative models––facilitation, tolerance and inhibition (Connell and Slatyer 1977). The species composition of vegetation during this process depends on a complex of environmental factors, particularly the soil nutrient status and pH, the water regime, climate and the relief (Kilian 1998). Some species, such as silver birch, grey alder and common aspen, are characterised by a high succession potential due to their tolerance of a broad array of environmental conditions, their few site requirements, rapid growth and large seed fall (Špulák et al. 2010).
Significant changes have been recorded in soils during ecological succession (Mielnik et al. 2021). Root growth typically increases soil porosity, water filtration and water holding capacity, whereas the bulk density decreases (Osman 2013; Yan et al. 2016). Following this, the topsoil becomes better aerated (Wei et al. 2013). During the succession, there is usually an increase in the total organic carbon (TOC) content and sometimes also certain biogenic elements (Matamala et al. 2008; Kalinina et al. 2011; Spohn et al. 2016). However, the effect on the soil nutrients, soil organic matter (SOM) and indicators of the soil’s ecochemical state (pH, sorption and buffering properties) strongly depends on the plant species (Augusto et al. 2002). The uptake of elements by roots, their accumulation in the biomass and their return to the soil via throughfall, stemflow and litterfall are considered to be major mechanisms that influence the soil cover during the succession process (Nilsson et al. 1999; Parzych et al. 2010; Jonczak and Mackiewicz 2012).
Silver birch (Betula pendula Roth) is widely distributed in forests of the Northern Hemisphere and has a large succession potential (Oikonomakis et al. 2000). It is often the main tree species on abandoned land undergoing natural regeneration (Špulák et al. 2010; Zasada et al. 2014; Franiel and Kompała-Bąba 2021). Therefore, it has a large potential to influence the soil. Jonczak et al. (2020) reviewed the current knowledge on the effects of birches on certain soil components, characteristics and processes. The authors concluded that several aspects related to soil–birch interactions are still poorly understood and require further exploration. Generally, aspects related to forest management and biomass productivity are more frequently represented in the literature than those related to soil–birch ecological feedback processes. The effect of birch on SOM quality and quantity is among the least-explored topics. Several studies have focussed on SOM stocks and their major characteristics. Rosenvald et al. (2011) reported increasing SOM contents in soils with stand age. This observation confirmed the findings of an earlier study performed in Latvia by Daugaviete et al. (2003). An increase in SOM stocks after the introduction of birch has also been reported by Holubík et al. (2014) for the Czech Republic. There are also comparative studies available that cover birch and other tree species stands. For example, Fernández-Núñez et al. (2010) reported a greater SOM accumulation in the soil under birch than under pine. Conversely, Kirby and Potvin (2007) and Ma et al. (2015) reported lower SOM contents under birch stands than under conifers. Jonczak (2013) found lower SOM stocks in post-arable soils afforested with silver birch when compared to arable field, meadow and fallow soils and black alder stands. Data on the impact of birch on SOM quality are scarce. However, some positive effects have been reported by Gawęda et al. (2018). Smolander and Kitunen (2002) recorded higher dissolved organic carbon (DOC) contents in the soils of birch stands when compared to other tree species. This observation can be explained by the positive effect that birches have on soil microorganisms (Priha and Smolander 1997, 1999; Saetre et al. 1999), and the higher degradability of DOC in the litter layer of birch stands compared to other tree species (Kiikkilä et al. 2006). The fractional composition of SOM and the internal structure of humic substances have not been studied in any detail in birch stands. To summarise, the available knowledge on the effects of birch on SOM quantity and quality is insufficient and inconsistent.
Considering the importance of the issue, we initialised a broad study focussed on silver birch–soil interactions in post-arable stands. The study covered a wide spectrum of issues, with a particular emphasis on SOM. In this study, we evaluated the quantitative and qualitative changes of SOM in the chronosequence of a silver birch succession on poor quality, and sandy (Brunic Arenosol) soils that had been abandoned after agricultural production. The study was based on five plots, constituting a typical sequence of spontaneous vegetation development, including arable field and fallow land and three birch stands aged 8, 33 and 40 years.
Materials and methods
Study area
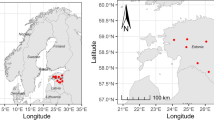
The study was conducted in central Poland (51.872 N, 20.297 E), representing a cold climatic zone, with a warm summer, humid and continental climate, according to the Köppen–Geiger classification (Peel et al. 2007). The climate is characterised by favourable solar conditions against the background territory of Poland. The mean annual temperature was 8.3 °C, and the mean annual sum of precipitation was 538.3 mm for Skierniewice station during the period 1951–2019. The warmest month was July, with a mean monthly air temperature of 18.6 °C, whilst the coldest was January, with a mean air temperature of −2.3 °C. The annual distribution of precipitation was characterised by a large bias between the warm (347.8 mm) and cold (190.5 mm) half-year. July was the month with the highest sum of precipitation, (81.7 mm) and January had the lowest (26.3 mm).
The study covered a sequence of five stands, located in the same complex of Brunic Arenosols developed from fluvioglacial sands, including arable field (AF), fallow (FA) and 8 year-old (YS), 33 year-old (MS) and 40-year-old (OS) successions of silver birch. Tree age was taken from Forest Database developed by National Forests. The birch stands differed in terms of their basic characteristics (Table 1). The highest density and lowest diameters (i.e. diameter at breast height and DBH) were typical of the YS. Tree density decreased, and DBH increased with stand age. The admixtures of black cherry (Prunus serotina Ehrh.), common aspen (Populus tremula L.) and grey alder (Alnus incana (L.) Moench) that were observed are typical of the spontaneous development of vegetation.
Soil sampling and analysis
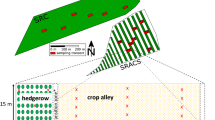
The study was performed in 2019. The soils were sampled from 20 × 25 m plots. One soil profile was described from each plot to provide a basic characterisation of the soils. The soils were described using the Food and Agriculture Organisation (FAO) of the United Nations criteria (FAO 2006) and classified according to the World Reference Base (WRB) system of the International Union of Soil Sciences Working Group (WRB 2015). Then, one disturbed and two undisturbed (100 cm3) samples were taken from horizons. The bulk density and total porosity were determined from undisturbed samples using the gravimetric method. The disturbed samples were air dried and sieved through a 2.0 mm sieve to remove the coarse fraction. Measurements taken on the earth fraction included the particle size distribution by mixed sieve and pipette methods, the pH, evaluated potentiometrically in a suspension of water and 1 mol dm−3 potassium chloride (KCl), and the TOC content and total nitrogen (N) content by dry combustion (Vario MacroCube, Elementar, Germany).
In addition, the O and A horizons were sampled from 20 regularly distributed sub-plots per plot. The O horizon samples were taken as 20 cm diameter cores, dried at 65 °C, weighed and milled into powder. One disturbed and one undisturbed sample of the A horizon were taken from each sub-plot and prepared for analysis using the same procedures as for the samples taken from the soil profiles. The thickness of the A horizon was measured in each location. Laboratory analyses included measuring the bulk density, total porosity, pH, TOC and N contents using the same methods as described above. The SOM content was determined by loss on ignition at 550 °C. The SOM fractional composition was analysed using the Schnitzer procedure (Gonet 1999). Sequential extraction included the determination of fractions extracted during sample decalcification (Cdec), fulvic acids (FAs), humic acids (HAs) and humins (HUs). The contents of all the fractions are given as the percentage of TOC (%TOC) in this paper. The HAs were extracted using the Schnitzer procedure (Gonet 1999), purified using a soft hydrofluoric (HF) and hydrochloric (HCl) acid solution (6 ml 40% HF + 5 ml 38% HCl + 990 ml deionised water), washed with deionised water and freeze-dried. Their irradiation absorbances at wavelengths of 465 and 665 nm were determined in 0.01% alkaline (0.1 mol dm−3 sodium hydroxide [NaOH]) solutions using a Jenway 6105 UV–VIS spectrophotometer, and the E4/6 quotients were calculated based on these data. The elemental composition (C, N and hydrogen [H]) of the freeze-dried HAs was determined by dry combustion (Vario MacroCube, Elementar, Germany). The oxygen (O) content was calculated as 100%–%C–%N–%H. The contents of all the elements are given in atomic percentages relative to the ash-free mass. The ratio of internal oxidation (ω) of the HAs was calculated based on their elemental compositions, following Żdanow (1965). A 13C nuclear magnetic resonance (NMR) analysis, with cross-polarisation and magic-angle spinning (CPMAS), was performed using a Bruker Avance III 400 MHz spectrometer. The chemical shift regions were referred to alkyl C (0–45 ppm), O-alkyl C (45–110 ppm), aryl C (110–160 ppm) and carboxylic C (160–220 ppm), following Wilson (1987).
Statistical analysis
The statistical analyses included the calculation of significance of differences between the means using the one-way analysis of variance (ANOVA), and homogeneous groups were distinguished with the use of Tukey test for significance level equal 0.05. Moreover, principal component analysis (PCA) and cluster analysis were used to investigate the relationship between the variables characterising soils and the multidimensional differentiation of the studied stands. Ward's method and the square of the Euclidean distance were used in the cluster analysis. All analyses were conducted using Statistica 13 software (Dell 2022).
Results
Basic characteristics of the soils
The soils of the studied plots showed features typical of Brunic Arenosols, including the sequence of horizons (A–Bw–C) and the texture––loamy sand or sandy loam in the A horizons (Fig. 1) and sand or loamy sand in the deeper horizons. The thickness of the A horizons varied from 22.8 ± 3.3 cm in the OS to 30.5 ± 2.2 cm in the FA soils (Table 2). The bulk density ranged from 1.26 ± 0.11 g cm−3 in the FA to 1.46 ± 0.08 g cm−3 in the YS soils, whereas total porosity ranged from 43.4% in the YS to 51.1% in the FA soils. The pH was comparable in all the A horizons, changing from 4.4 ± 0.1 to 4.8 ± 0.5 for pH-H2O and from 3.8 ± 0.2 to 4.0 ± 0.1 for pH-KCl. Higher pH values were noted in the O horizons (5.3 ± 0.2–5.8 ± 0.3 for pH-H2O and 5.2 ± 0.2–5.7 ± 0.4 for pH-KCl). The soils were generally poor in TOC (7.3 ± 1.2–11.9 ± 2.0 g kg−1) and N (0.57 ± 0.07–0.97 ± 0.13 g kg−1) in the A horizons. The TOC: N ratio ranged from 11.6 ± 0.6:1 in the AF soils to 15.6 ± 1.6:1 in the MS soils. In the O horizons, it ranged from 31.5 ± 6.3:1 to 35.3 ± 4.6:1. The differences between the plots of TOC and N contents and the TOC:N ratios were statistically significant in the majority of cases (Table 2).
SOM content and fractional composition
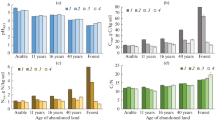
The SOM content in A horizons ranged from 15.68 ± 1.92 to 22.90 ± 2.47 g kg−1, being the lowest in the YS and the highest in the AF soils (Table 3). Generally, the SOM content was comparable in the AF, FA and OS plots. In the birch stands, there was a positive correlation with stand age. When compared to the SOM stock, the highest values were noted in the AF and FA soils, whereas the lowest were in the birch stands. Differences between the SOM in the birch stand soils were low and usually statistically insignificant, with 89.4–91.3% of the SOM in the forest soils being allocated to the A horizons and 8.7–10.6% occurring in the litter layers.
The humin fraction (HUs) predominated amongst the SOM components in all the studied soils (Table 3). In the O horizons, it constituted 76.9 ± 1.8–79.6 ± 2.2% of the TOC, whereas in the A horizons, and this was 63.1 ± 3.5–76.4 ± 2.9% of the TOC. The HUs contents in the A horizons increased from the AF to the MS and then, decreased with succession age. Differences between the stands were statistically significant in majority of cases. The FAs occurred in amounts of 9.3 ± 1.2% TOC to 10.6 ± 1.5% TOC in the O horizons and 13.7 ± 1.8% TOC to 26.0 ± 4.0% TOC in the A horizons. In the A horizons, the FAs contributed most to the OS and AF soils and much less to the soils of the remaining stands. As for the previous fraction, differences between the stands in terms of FAs content were usually statistically significant. The HAs constituted 8.7 ± 1.2–11.8 ± 1.1% TOC of the O horizons and 7.7 ± 1.2–13.5 ± 1.3% TOC of the A horizons. Significantly, lower HA contents were noted in the forest soils than the AF and FA soils. The Cdec fraction occurred in its lowest amounts in both the O and A horizons, mostly constituting around 2% TOC. Only in the A horizons of the OS soils did it occur in higher amounts (Table 3).
Elemental composition of the HAs
The major component of the HAs was H followed by C (Table 4). The H content fluctuated around 40 atomic % and showed relatively low variability amongst the stands and soil horizons. The C showed higher variability, with its content in the O horizons ranging from 31.91 to 33.65 atomic %, whereas in the A horizons, and it was 28.60 to 33.64 atomic %. The lowest C contents were recorded in the AF and OS soils, whereas the highest were in the FA soils. Considerable differences in the N content were found between the O and A horizons, and these values being 1.25–1.37 and 2.06–2.46 atomic %, respectively. The HAs richest in N were from the A horizons of the YS soils, whereas the poorest were from the O horizons of the MS soils. The O content in the HAs from the litter layer increased with stand age. In the A horizons, O showed the general tendency AF > FA > YS < MS < OS with homogenous two groups FA, YS, MS and AF, FA, OS. The similar tendency was noted for the ω developed by Żdanov (1965), where two homogenous groups were determined AF, FA, YS, MS and AF, FA, OS (Table 4).
HAs UV–visible spectroscopy and 13C-NMR
The E4/6 quotient is commonly used as an indicator of soil humus quality and maturity. In this studied soils, it varied significantly in the O (7.1 ± 0.3–8.0 ± 0.3) and A (4.5 ± 0.2–4.9 ± 0.3) horizons (Table 5). The tendency observed is typical. The 13C-NMR analysis indicated a predominance of aliphatic (alkyl, O-alkyl) over aromatic and carbonyl structures in all the studied soils (Table 6). The aliphatic structures comprised 57.1–67.9%, aromatic 21.3–26.3% and carbonyl 8.4–18.4% C. Some differences in the internal structure of the HAs were noted amongst the stands. Typically, the most clearest differences were recorded between the O and A horizons. The greatest variability was noted in the spectral regions 0–45, 90–110, 160–190 and 190–220 ppm. The HAs extracted from the O horizons were generally richer in aliphatic and poorer in carboxylic structures than those from the A horizons. There were also some clear tendencies when comparing the HAs from the A horizons amongst the stands. The transformation from AF to FA and then, YS to MS soils showed increasing tendencies in the 0–45 and 190–200 ppm spectral regions and decreasing tendencies in the 45–60, 60–90 and 90–110 ppm regions.
The characteristics of the studied stands based on selected variables
Fig. 2 shows the results of PCA analysis. PCA was carried out for all studied stands in the O and A horizons for the following variables: fractional composition (contents of Cdec, HAs, FAs and HUs expressed as a % of TOC), the E4/6 quotient of HAs, elemental (C, H, N and O) composition of HAs and the content of basic structures (Calip, Carom and Ccarb) based on 13C-NMR analysis. The first two components provide 72.92% information about the variability of the data set under consideration. The differences between stands in relation to the horizontal axis (PC1) are the most important. This means that the O and A horizons introduced the greatest differentiation of stands. Calip, E4/6 and HUs were strongly positively correlated, and all the O horizons (YS–O, MS–O and OS–O) had high values of these variables. They were strongly negatively correlated with variables FA, HAs component O and N and Ccarb which were very high in the OS–A stand as well as in the rest of A horizons of stands. The HAs component C was strongly negatively correlated with Cdec and FAs, which were very high in the OS–A stand. The FA–A stand had a very high value of HAs, and low values of HAs component H, thus, was significantly different from other A horizons.
Based on the same variables for the stands in the O and A horizons, a cluster analysis was performed (Fig. 3). Three stand groups were distinguished as a result of the analysis. The stands in the O horizons were grouped showing similarities between them, as were the stands in the A horizons. In the O horizons, YS–O and MS–O were the most similar. In the A horizons, AF–A and OS–A were the most similar as were YS–A and MS–A.
Cluster analysis (Ward method, Euclidean distance) carried out for all studied stands in the O and A horizons for the following variables: fractional composition Cdec, HAs, FAs and HUs expressed as a % of TOC, the E4/6 quotient, HAs components, i.e. C, H, N, O and contents of Calip., Carom., Ccarb. based on 13C-NMR analysis
Discussion
The changes recorded in the soils during succession must be considered in the context of a broad understanding of the initial state of the soils, their ability to buffer external factors, the species composition of invading vegetation and the quantitative and qualitative transformation of the soil microbiome. These factors strongly influence soil processes and the cycling of matter and energy and are also reflected in the characteristics of SOM. Abandoned arable soils are often poor in SOM. Several studies have shown that conventional agriculture causes a strong depletion of SOM stocks and a deterioration of its quality (Compton and Bonne 2000; Murty et al. 2002), followed by negative changes in soil aggregation (Polláková et al. 2018; Šimanský et al. 2019) and other soil characteristics. Finally, the soil fertility and productivity decrease below the levels profitable for agricultural production. This problem concerns mainly sandy soils, which are naturally poor in nutrients, usually acidified and characterised by low cation-exchange, water holding and buffering capacities (Okołowicz et al. 2003; Jonczak and Sztabkowski 2021). Negative changes in arable soils can be reduced by applying suitable fertilisation, in particular organic (Abiven et al. 2009; Šimanský et al. 2019), and using certain agrotechnical practices, such as no-till, strip-till, ridge-till and mulch-till systems. The positive effects of reduced tillage on soil quality have been reported by several authors (e.g. Gadermaier et al. 2012; Devine et al. 2014; Blanco-Canqui and Ruis 2018; Kobierski et al. 2020).
The rehabilitation of degraded soils is a priority of sustainable management policies. This process is partially controlled in afforested areas. Soil amelioration can essentially contribute to maintaining the sustainability of forest ecosystems (Kilian 1998). However, many abandoned soils undergo natural regeneration. The development of annual vegetation, including grasses and herbs, usually constitutes the first step in that process. The positive effects of such vegetation on SOM have been well reported in the literature. Leinweber et al. (1993) observed a rapidly growing SOM content and changes in its structure, in the initial stages of succession. Reuter (1991) found that the decomposition of grass residues led to a significant accumulation of SOM, and the steepest increase being between the second and seventh years. Positive effects have also been observed where arable fields have been transformed into pastures (Römkens et al. 1999; Pulleman et al. 2000; Guo and Gifford 2002). Grass vegetation effectively contributes to the development of a stable soil structure (Pulleman et al. 2005) and the physical protection of SOM in aggregates (Six et al. 1998; Chevallier et al. 2004). Moreover, it effectively prevents SOM mineralisation by limiting soil heating (Li et al. 2016). The most common positive effects of grass vegetation on SOM were partially confirmed in the present study. The SOM and TOC contents in the AF and FA soils did not differ statistically (Tables 2, 3). The same tendency was observed in the SOM stocks (Table 3). Some differences were observed in the SOM fractional compositions. The FA soils were characterised by a higher contribution of HAs and HUs and a lower contribution of FAs (Table 3). This observation is in line with the findings of Ukalska-Jaruga et al. (2019). Based on our results, it can be stated that grass vegetation contributed to the greater stability of humic substances. This was also confirmed by the results of the elemental analysis of the HAs; a decrease in H content with increasing C and N content was noted (Table 4). The H:C atomic ratios in the HAs, which fluctuated around 1.2–1.4, correspond to aromatic structures coupled to an aliphatic chain containing up to 10 C atoms (Gonet 1989). The 13C-NMR analysis did not reveal any clear differences between the AF and FA soils (Table 6). Generally, in both these soils, aliphatic predominated over aromatic structures, which is typical of poorly advanced humification (Dębska 2004). However, it should be noted that the CPMAS 13C-NMR spectra were semiquantitative (Kinchesh et al. 1995; Mathers et al. 2000), and the C detected using this technique represents only part of the total pool of C in the sample (Mao et al. 2000).
The transformation of SOM during the latter stages of succession strongly depends on the species composition of the invading trees (Marcos et al. 2010). There are several mechanisms involved in causing the effects of forest vegetation on soils, as reviewed by Augusto et al. (2002). Litterfall production and its microbiochemical transformation are one of the most important amongst these, especially in the context of SOM. Litterfall is a key link in the biogeochemical cycling of matter and energy (Nordén 1994; Astel et al. 2009; Krishna and Mohan 2017), providing an important pool of soil nutrients (Ukonmaanaho et al. 2008; Straková et al. 2010) and the precursors to humic substances (Taylor et al. 1989; Stevenson 1994; Cotrufo et al. 2015). Generally, trees with higher litter quality exert a more positive influence on soil biodiversity (van Calster et al. 2008) and the intensity of biochemical processes (Kooijman and Martinez-Hernandez 2009). The issue of litterfall production and its chemistry and decomposition in birch stands has not been sufficiently explored until now, however. Studies by Aussenac et al. (1972) and Tripathi et al. (2006) have shown that litterfall production in birch stands is no different from that in forests dominated by many deciduous tree species (Shen et al. 2019). Generally, silver birch litterfall is moderately abundant in nutrients (Berg and Staaf 1987; Perala and Alm 1990; Johansson 1995; Brandtberg et al. 2004; Carnol and Bazgir 2013), with leaves, as the major component, decomposing rapidly (in 1–2 years) under temperate climatic conditions (Mikola 1985; Tripathi et al. 2006; Hordecki and Jagodziński 2019) and more slowly under colder climatic conditions (Huttunen et al. 2009). Hence, mull-type humus is typical of birch stands. This was confirmed in the studied chronosequence. The litter horizons were poorly developed and relatively rich in N, which was also reflected in the low C:N ratios (Table 2). This relatively high N abundance can be explained by the post-arable nature of the stand. Pools of SOM in the O horizon were lower than in the A horizon (Table 3). The qualitative characteristics of the SOM were typical of litter layers, including the low contribution of humified components with high residual (non-humified) fraction contents (Table 3). Moreover, the HAs were rich in H and C, but poor in N (Table 4). However, the 13C-NMR results showed an increasing aromaticity of the HAs with stand age (Table 6). Alkyl and O-alkyl C predominated in all the studied samples. This is typical of most forest soils, particularly in their litter horizons (Ussiri and Johnson 2003; Rumpel et al. 2005). The observed tendencies cannot be unambiguously explained by birch age due to the admixture of other species. In addition, the characteristics of the litter horizons need to be carefully interpreted, especially in mull-type humus, due to the single sampling. This approach does not reflect dynamic changes in the SOM that can be observed over time as an effect of decomposition and the seasonal variability of the weather conditions, amongst other factors.
The process of litterfall decomposition involves the following sequence of changes: (1) the preferential degradation of structures, such as cellulose and hemicellulose, by the microbial community, resulting in a decrease in the O-alkyl C content; (2) the decomposition of aromatic C and (3) the accumulation of alkyl C. Dissolved organic matter (DOM), as a product of that process, is transported towards deeper soil horizons by leaching. The intensity of its production and transport is strongly affected by a complex of abiotic and biotic factors, which have been the focus of numerous studies. For example, Aber et al. (1993) and Currie et al. (1996) both highlighted the importance of tree species composition, Clark et al. (2005) soil pH and Møller et al. (1999) soil microbial activity. In the mineral horizons, DOM is involved in various soil structural components and processes (Chen and Avnimelech 1986). The secondary synthesis of its low-molecular compounds is of key importance in humus formation (Stevenson 1994) and development of the A horizon. Due to its location at the top of the soil profile, the characteristics of that horizon cause DOM to be highly dynamic and susceptible to impacts from external factors, particularly land-use type and vegetation.
Previous studies have demonstrated the various effects of tree species composition on SOM content and quality. Most authors have reported increasing SOM contents and pools during the transition from arable field or grassland to forest land use (e.g. Gonet et al. 2009; Oktaba and Kusińska 2012; Rytter and Rytter 2020). This is logical because the SOM content in soils is directly affected by the input of plant-derived C (Wang et al. 2017), and this is usually greater in forest ecosystems. However, in young tree stands, litterfall production is often low. Moreover, accordingly to Oades (1988), there is a greater intensity of mineralisation in forests; thus, their SOM contents are lower than those of grassland soils. Li et al. (2016) reported that the effects of afforestation on soil C and N can be strongly modified by lithology. The impact of birch trees is not fully understood in this context, although previous studies have highlighted relatively positive effects (Daugaviete et al. 2003; Rosenvald et al. 2011; Holubík et al. 2014). Vladychenskii et al. (2007) also found much higher SOM contents under birches when compared to arable soils, although the pools not differ much. Moreover, the authors noted significant differences in the distribution of humus in the microprofiles under birch, oak and pine forests. The positive effects of birches on SOM reported by previous authors were not confirmed by the present study, with our findings clearly indicating a rapid loss of that component following silver birch succession (Table 3). This tendency can be explained by increased dehydrogenase activity and accelerated mineralisation (Gawęda et al. 2019) in the young birch stands compared to the older stands. This hypothesis is also supported by the fractional composition of the SOM. The soils under the young birches were characterised by a lower content of low-molecular humus fractions (Cdec, FAs) (Table 3), and these are more susceptible to microbial degradation. Moreover, in the YS, there was typically a lower internal oxidation of HAs, and the highest content of aliphatic structures with the lowest aromatic content (Table 6). The accelerated mineralisation of low-molecular SOM fractions may also be partially associated with the high nutritional demands of young birches (Miller 1984). Generally, it can be concluded that the transition from FA to a YS of silver birch contributes to negative changes in SOM characteristics.
During the latter stages of succession, some evidence of SOM regeneration was observed. The SOM content and stock gradually increased, until ultimately reaching values typical of AF and FA soils at the OS stage (Table 3). This is in line with the findings of previous studies by Smal and Olszewska (2008) and Tanner et al. (2014). However, considering the admixtures in the studied stands, it was not possible to precisely estimate the role of silver birch in that process. The occurrence of alder in the OS stand could be of particular importance. Podwika et al. (2018) reported the strongest positive effects of alder on soil quality compared to several other species. Alder litterfall is rich in nutrients (Jonczak et al. 2016) and rapidly decomposes (Jonczak et al. 2015). The increasing tendency in SOM content in the studied silver birch chronosequence was mainly associated with increasing contents of FAs, whereas the HAs remained relatively stable (Table 3). The E4/6 ratios of the HAs did not changed significantly (Table 5); however, a large increase in the internal oxidation of the HAs was noted (Table 4), accompanied by considerable changes in their molecular structures (Table 6).
Conclusions
Our findings confirm the clear inter-relationship between land-use type, vegetation and SOM characteristics. The spontaneous development of vegetation was accompanied by dynamic changes in the SOM stocks and quality in the abandoned sandy soils. There was little difference, in the transition from AF to FA soil, in the SOM stocks in the A horizons, although the SOM fractional composition and internal structure were modified. Increasing contents of HAs with decreasing contents of FAs might be considered to be an indicator of SOM stabilisation under FA vegetation. This was also supported by the results of the elemental and 13C-NMR analyses of the HAs. This observation highlights the positive effects of grass vegetation on SOM quality and supports the observations of several other authors. In its initial stage, the silver birch succession was accompanied by a rapid loss in SOM in the A horizon. This was probably because of accelerated mineralisation and the high nutritional demands of that tree species. This hypothesis was supported by the qualitative changes in, and fractional composition of, the SOM. The soils under young birches were characterised by a lower content of low-molecular humus fractions, which are more susceptible to microbial degradation, when compared to the FA soils. The birch succession contributed to a lower internal oxidation of HAs and an increase in the content of aliphatic structures in their molecules. Consequently, it can generally be stated that the initial stage of silver birch succession caused negative changes in the SOM characteristics. However, during the following stages of succession, some evidence of regeneration was observed. The contents of SOM gradually increased, mainly due to an increase in FAs. A considerable increase in the internal oxidation of the HAs, accompanied by structural changes in their molecules, as evidenced by 13C-NMR analysis, confirms the stabilisation of those substances. To summarise, our study has provided strong evidence for negative rather than positive changes in SOM caused by silver birch succession on sandy soils. More utilitarial conclusion of this study is that afforestation of post-agricultural sandy soils with that tree does not contribute to sustainable management of its resources. The results of the study provide grounds for considering the relevance of this practice. However, further studies are highly recommended to confirm or reject our hypothesis based on a wider number of cases, covering a broader spectrum of environmental/soil conditions.
References
Aber JD, Magill A, Boone R, Melillo JM, Steudler P, Bowden RD (1993) Plant and soil responses to three years of chronic nitrogen additions at the Harvard Forest, Petersham, MA. Ecol Appl 3:156–166. https://doi.org/10.2307/1941798
Abiven S, Menasseri S, Chenu C (2009) The effects of organic inputs over time on soil aggregate stability-a literature analysis. Soil Biol Biochem 41:1–12. https://doi.org/10.1016/j.soilbio.2008.09.015
Astel A, Parzych A, Trojanowski J (2009) Comparision of litterfall and nutrient return in a Vaccinio uliginosi-Betuletum pubescentis and a Empetro nigri-Pinetum forest stands in northern Poland. Forest Ecol Manage 257:2331–2341. https://doi.org/10.1016/j.foreco.2009.03.026
Augusto L, Ranger J, Binkley D, Rothe A (2002) Impact of several common tree species of European temperate forests on soil fertility. Ann for Sci 59:233–253. https://doi.org/10.1051/forest:2002020
Aussenac G, Bonneau M, Le Tacon F (1972) Restitution des éléments minéraux au sol par l’intermédiaire de la litière et des précipitations dans quatre peuplements forestiers de l’est de la France. Oecol Plant 7:1–21
Berg B, Staaf H (1987) Release of nutrients from decomposing white birch and Scots pine needle litter. Pedobiologia 30(1):55–63
Blanco-Canqui H, Ruis SJ (2018) No-tillage and soil physical environment. Geoderma 326:164–200. https://doi.org/10.1016/j.geoderma.2018.03.011
Brandtberg PO, Bengtsson J, Lundkvist H (2004) Distributions of the capacity to take up nutrients by Betula spp. and Picea abies in mixed stands. Forest Ecol Manag 198:193–208. https://doi.org/10.1016/j.foreco.2004.04.012
Carnol M, Bazgir M (2013) Nutrient return to the forest floor through litter and throughfall under 7 forest species after conversion from Norway spruce. Forest Ecol Manag 309:66–75. https://doi.org/10.1016/j.foreco.2013.04.008
Chen Y, Avnimelech Y (eds) (1986) The role of organic matter in modern agriculture. Martinus Nijhoff, Dordrecht, The Netherlands
Chevallier T, Blanchart E, Albrecht A, Feller C (2004) The physical protection of soil organic carbon in aggregates: a mechanism of carbon storage in a Vertisol under pasture and market gardening (Martinique, West Indies). Agr Ecosys Environ 103:375–387. https://doi.org/10.1016/j.agee.2003.12.009
Clark JM, Chapman PJ, Adamson JK, Lane SN (2005) Influence of drought-induced acidification on the mobility of dissolved organic carbon in peat soils. Glob Change Biol 11:791–809. https://doi.org/10.1111/j.1365-2486.2005.00937.x
Compton JE, Boone RD (2000) Long-term impacts of agriculture on soil carbon and nitrogen in New England forests. Ecology 81(8):2314–2330. https://doi.org/10.2307/177117
Connell JH, Slatyer RO (1977) Mechanisms of succession in natural communities and their role in community stability and organization. Am Nat 111(982):1119–1144
Corbet SA (1995) Insects, plants and succession: advantages of long term set-aside. Agr Ecosys Environ 53:201–217. https://doi.org/10.1016/0167-8809(94)00581-X
Cotrufo MF, Soong JL, Horton AJ, Campbell EE, Haddix ML, Wall DH, Parton WJ (2015) Formation of soil organic matter via biochemical and physical pathways of litter mass loss. Nat Geosci 8:776–779. https://doi.org/10.1038/ngeo2520
Currie WS, Aber JD, McDowell WH, Boone RD, Magill AH (1996) Vertical transport of dissolved organic C and N under long-term N amendments in pine and hardwood forests. Biogeochemistry 35:471–505. https://doi.org/10.1007/BF02183037
Daugaviete M, Krŭmina M, Kǎpopstos V, Lazdinš A (2003) Farmland afforestation: the plantations of birch Betula pendula Roth. on different soils. Baltic for 9(1):9–21
Dębska B (2004) Właściwości substancji humusowych gleby nawożonej gnojowicą. Rozprawy nr 110. Wydawnictwo Uczelniane ATR, Bydgoszcz
Dell Inc. (2022) Dell statistica data analysis software system. Version 13. 2016. Available online: software.dell.com accessed on 30 Jun 2022
Devine S, Markewitz D, Hendrix P, Coleman D (2014) Soil aggregates and associated organic matter under conventional tillage, no-tillage, and forest succession after three decades. PLoS ONE 9(1):e84988. https://doi.org/10.1371/journal.pone.0084988
FAO (2006) Guidelines for soil description. FAO, Rome
Fernández-Núñez E, Rigueiro-Rodrígueza A, Mosquera-Losadaa MR (2010) Carbon allocation dynamics one decade after afforestation with Pinus radiata D. Don and Betula alba L. under two stand densities in NW Spain. Ecol Eng 36:876–890. https://doi.org/10.1016/j.ecoleng.2010.03.007
Franiel I, Kompała-Bąba A (2021) Reproduction strategies of the silver birch (Betula pendula Roth) at post-industrial sites. Sci Rep 11:11969. https://doi.org/10.1038/s41598-021-91383-0
Gadermaier F, Berner A, Fließbach A, Friedel JK, Mäder P (2012) Impact of reduced tillage on soil organic carbon and nutrient budgets under organic farming. Renew Agr Food Syst 27:1–13. https://doi.org/10.1017/S1742170510000554
Gawęda T, Błońska E, Małek S (2019) Soil organic carbon accumulation in post-agricultural soils under the influence birch stands. Sustainability 11:4300. https://doi.org/10.3390/su11164300
Gawęda T, Błońska E, Małek S, Bijak S, Zasada M (2018) Zastosowanie ITGL w ocenie gleb porolnych z naturalnym odnowieniem brzozy. Sylwan 162(5):396–402
Gonet SS (1989) Właściwości kwasów huminowych gleb o zróżnicowanym nawożeniu. Rozprawy 33. Bydgoszcz, ATR.
Gonet SS (1999) Ekstrakcja z gleb i oczyszczanie kwasów huminowych metodami Shnitzera. In: Dziadowiec H, Gonet S (eds) Przewodnik metodyczny do badań materii organicznej gleb, Prace Komisji Naukowych Polskiego Towarzystwa Gleboznawczego nr 120. Komisja Chemii Gleb, Zespół Materii Organicznej Gleb II/16, Warszawa, pp 42–44
Gonet SS, Dębska B, Dziamski A, Banach-Szott M, Zaujec A, Szombathova N (2009) Properties of organic matter in Haplic Luvisol under arable, meadow and forest management. Pol J Soil Sci 42(2):139–148
Guo LB, Gifford RM (2002) Soil carbon stocks and land use change: a meta analysis. Global Change Biol 8:345–360. https://doi.org/10.1046/j.1354-1013.2002.00486.x
Holubík O, Podrázský V, Vopravil J, Khel T, Remeš J (2014) Effect of agricultural lands afforestation and tree species composition on the soil reaction, total organic carbon and nitrogen content in the uppermost mineral soil profile. Soil Water Res 9:192–200. https://doi.org/10.17221/104/2013-SWR
Hordecki P, Jagodziński AM (2019) Site type effect on litter decomposition rates: a three-year comparison of decomposition process between spoil heap and forest sites. Forests 10(4):353. https://doi.org/10.3390/f10040353
Huttunen L, Aphalo PJ, Lehto T, Niemela P, Kuokkanen K, Kelloma S (2009) Effects of elevated temperature, elevated CO2 and fertilization on quality and subsequent decomposition of silver birch leaf litter. Soil Biol Biochem 41:2414–2421. https://doi.org/10.1016/j.soilbio.2009.08.014
IUSS Working Group WRB (2015) World Reference Base for Soil Resources 2014, update 2015. International soil classification system for naming soils and creating legends for soil maps. World Soil Resources Reports No. 106. FAO, Rome
Johansson MB (1995) The chemical composition of needle and leaf litter from Scots pine, Norway spruce and white birch in Scandinavian forests. Forestry 68:49–62. https://doi.org/10.1093/forestry/68.1.49
Jonczak J (2013) Soil organic matter properties in Stagnic Luvisols under different land use types. Acta Agrophys 20(4):565–576
Jonczak J, Jankiewicz U, Kondras M, Kruczkowska B, Oktaba L, Oktaba J, Olejniczak I, Pawłowicz E, Polláková N, Raab T, Regulska E, Słowińska S, Sut-Lohmann M (2020) The influence of birch trees (Betula spp.) on soil environment – a review. Forest Ecol Manage 477:1–13. https://doi.org/10.1016/j.foreco.2020.118486
Jonczak J, Mackiewicz K (2012) Influence of pine and spruce admixture in European beech stand on some properties of organic and humic horizons of Dystric Arenosols and the intensity of biological turnover. Pol J Soil Sci 45(2):221–233
Jonczak J, Olejniczak M, Parzych A, Sobisz Z (2016) Dynamics, structure and chemistry of litterfall in headwater riparian forest on the area of Middle Pomerania. J Elementol 21(2):381–392. https://doi.org/10.5601/jelem.2015.20.3.938
Jonczak J, Parzych A, Sobisz Z (2015) Decomposition of four tree species leaf litters in headwater riparian forest. Baltic for 21(1):133–143
Jonczak J, Sztabkowski K (2021) Spatial variability of Brunic Arenosols and associated soils along the slope of the Słupia River valley (Middle Pomerania, northern Poland). Soil Sci Ann 72(4):143892. https://doi.org/10.37501/soilsa/143892
Kalinina O, Krause SE, Goryachkin SV, Karavaeva NA, Lyuri DI, Giani L (2011) Self-restoration of post-agrogenic Chernozems of Russia: soil development, carbon stocks and dynamics of carbon pools. Geoderma 162:196–206. https://doi.org/10.1016/j.geoderma.2011.02.005
Kiikkilä O, Kitunen V, Smolander A (2006) Dissolved soil organic matter from surface organic horizons under birch and conifers: degradation in relation to chemical characteristics. Soil Biol Biochem 38:737–746. https://doi.org/10.1016/j.soilbio.2005.06.024
Kilian W (1998) Forest site degradation—temporary deviation from the natural site potential. Ecol Eng 10:5–18. https://doi.org/10.1016/S0925-8574(97)10021-0
Kinchesh P, Powlson DS, Randall EW (1995) 13C NMR studies of organic matter in whole soils: I. Quantitation possibilities. Eur J Soil Sci 46:125–138. https://doi.org/10.1111/j.1365-2389.1995.tb01819.x
Kirby KR, Potvin C (2007) Variation in carbon storage among tree species: implications for the management of a small-scale carbon sink project. For Ecol Manag 246(2–3):208–221. https://doi.org/10.1016/j.foreco.2007.03.072
Kobierski M, Lemanowicz J, Wojewódzki P, Kondratowicz-Maciejewska K (2020) The effect of organic and conventional farming systems with different tillage on soil properties and enzymatic activity. Agronomy 10(11):1809. https://doi.org/10.3390/agronomy10111809
Kooijman AM, Martinez-Hernandez GB (2009) Effects of litter quality and parent material on organic matter characteristics and N-dynamics in Luxembourg beech and hornbeam forests. For Ecol Manag 257:1732–1739. https://doi.org/10.1016/j.foreco.2009.01.030
Krishna MP, Mohan M (2017) Litter decomposition in forest ecosystems: a review. Energ Ecol Environ 2(4):236–249. https://doi.org/10.1007/s40974-017-0064-9
Kurganova I, Lopes de Gerenyu V, Six J, Kuzyakov Y (2014) Carbon cost of collective farming collapse in Russia. Glob Chang Biol 20:938–947. https://doi.org/10.1111/gcb.12379
Leinweber P, Friindz R, Reuter JG (1993) A 13C-NMR study on the formation of soil organic matter from grass residues. 2. Pflanzenernahr Bodenk 156:415–420
Li C, Li Q, Zhao L, Ge S, Chen D, Dong Q, Zhao X (2016) Land-use effects on organic and inorganic carbon patterns in the topsoil around Qinghai Lake basin, Qinghai-Tibetan Plateau. CATENA 147:345–355. https://doi.org/10.1016/j.catena.2016.07.040
Ma J, Bu R, Liu M, Chang Y, Qin Q, Hu Y (2015) Ecosystem carbon storage distribution between plant and soil in different forest types in Northeastern China. Ecol Eng 81:353–362. https://doi.org/10.1016/j.ecoleng.2015.04.080
Mao JD, Hu WG, Schmidt-Rohr K, Davies G, Ghabbour EA, Xing B (2000) Quantitative characterization of humic substances by solid-state carbon-13 nuclear magnetic resonance. Soil Sci Soc Am J 64:873–884. https://doi.org/10.2136/sssaj2000.643873x
Marcos E, Calvo L, Marcos JA, Taboada Á, Tárrega R (2010) Tree effects on the chemical topsoil features of oak, beech and pine forests. Eur J Forest Res 129:25. https://doi.org/10.1007/s10342-008-0248-0
Matamala R, Jastrow JD, Miller RM, Garten CT (2008) Temporal changes in C and N stocks of restored prairie: implications for C sequestration strategies. Ecol Appl 18:1470–1488. https://doi.org/10.1890/07-1609.1
Mathers NJ, Mao XA, Xu ZH, Saffigna PG, Berners-Price SJ, Perera MCS (2000) Recent advances in application of 13C and 15N NMR spectroscopy. Aust J Soil Res 38:769–787. https://doi.org/10.1071/SR99074
Mielnik L, Hewelke E, Weber J, Oktaba L, Jonczak J, Podlasiński M (2021) Changes in the soil hydrophobicity and structure of humic substances in sandy soil taken out of cultivation. Agr Ecosys Environ 319:107554. https://doi.org/10.1016/j.agee.2021.107554
Mikola P (1985) The effect of tree species on the biological properties of forest soil. National Swedish Environmental Protection Board, 3017, pp 1–29
Miller HG (1984) Nutrient cycles in birchwoods. Proc Roy Soc Edinb 853:83–96. https://doi.org/10.1017/S0269727000003912
Miripanah Z, Tavakoli M, Rostaminya M, Naderi M (2019) Carbon sequestration via afforestation as a sustainable action to mitigate climate change in Western Iran. A Un Nat Sust Dev J 43(3):194–202. https://doi.org/10.1111/1477-8947.12185
Møller J, Miller M, Kjøller A (1999) Fungal-bacterial interaction on beech leaves: influence on decomposition and dissolved organic carbon quality. Soil Biol Biochem 31:367–374. https://doi.org/10.1016/S0038-0717(98)00138-2
Murty D, Kirschbaum MUF, Mcmurtrie RE, Mcgilvray H (2002) Does conversion of forest to agricultural land change soil carbon and nitrogen? A review of the literature. Global Change Biol 8:105–123. https://doi.org/10.1046/j.1354-1013.2001.00459.x
Nilsson MCh, Wardle DA, Dahlberg A (1999) Effects of plant litter species composition and diversity on the boreal forest plant-soil system. Oikos 86:16–26
Nordén U (1994) Leaf litterfall concentrations and fluxes of elements in deciduous tree species. Scand J for Res 9(1–4):9–16. https://doi.org/10.1080/02827589409382807
Oades JM (1988) The retention of organic matter in soils. Biogeochemistry 5:35–70
Oikonomakis NG, Ganatsas P (2020) Secondary forest succession in Silver birch (Betula pendula Roth) and Scots pine (Pinus sylvestris L.) southern limits in Europe, in a site of Natura 2000 network – an ecogeographical approach. For Syst 29(2):e010. https://doi.org/10.5424/fs/2020292-15680
Okołowicz M, Czępińska-Kamińska D, Janowska E, Konecka-Betley K (2003) Rozmieszczenie fosforu w glebach rezerwatu biosfery “Puszcza Kampinoska.” Roczniki Gleboznawcze Soil Sci Ann 54(3):39–48
Oktaba L, Kusińska A (2012) Soil organic matter in afforested post-agricultural soils. Pol J Soil Sci 45(1):39–47
Osman KT (2013) Soils principles. Springer, London, Properties and Management. https://doi.org/10.1007/978-94-007-5663-2
Parzych A, Sobisz Z, Trojanowski J (2010) Variability of nitrogen and phosphorus concentration and the net primary production Vaccinium vitis-idaea L. and Vaccinium myrtillus L. in chosen woodland ecosystems of the Słowiński National Park. Arch Environ Prot 36(2):91–104
Paul KI, Polglase PJ, Nyakuengama JG, Khanna PK (2002) Change in soil carbon following afforestation. For Ecol Manag 168(1–3):241–257. https://doi.org/10.1016/S0378-1127(01)00740-X
Peel MC, Finlayson BL, McMahon TA (2007) Updated world map of the Köppen−Geiger climate classification. Hydrol Earth Syst Sci 11(5):1633–1644. https://doi.org/10.1127/0941-2948/2006/0130
Perala DA, Alm AA (1990) Reproductive ecology of birch: a review. For Ecol Manag 32:1–38. https://doi.org/10.1016/0378-1127(90)90104-J
Pèrez-Silos I, Alvarez-Martinez JM, Barquìn J (2021) Large-scale afforestation for ecosystem service provisioning: learning from the past to improve the future. Landsc Ecol 36:3329–3343. https://doi.org/10.1007/s10980-021-01306-7
Podwika M, Solek-Podwika K, Ciarkowska K (2018) Changes in the properties of grassland soils as a result of afforestation. iForest 11:600–608. https://doi.org/10.3832/ifor2556-011
Polláková N, Šimanský V, Kravka M (2018) The influence of soil organic matter fractions on aggregates stabilization in agricultural and forest soils of selected Slovak and Czech hilly lands. J Soils Sediments 18:2790–2800. https://doi.org/10.1007/s11368-017-1842-x
Priha O, Smolander A (1997) Microbial biomass and activity in soil under Pinus sylvestris, Picea abies and Betula pendula at originally similar field afforestation sites. Biol Fertil Soils 24:45–51. https://doi.org/10.1007/BF01420219
Priha O, Smolander A (1999) Nitrogen transformations in soil under Pinus sylvestris, Picea abies and Betula pendula at two forest sites. Soil Biol Biochem 31:965–977. https://doi.org/10.1016/S0038-0717(99)00006-1
Pulleman MM, Bouma J, van Essen EA, Meijles EW (2000) Soil organic matter content as a function of different land use history. Soil Sci Soc Am J 64:689–693. https://doi.org/10.2136/sssaj2000.642689x
Pulleman MM, Six J, Van Breemen N, Jongmans AG (2005) Soil organic matter distribution and microaggregate characteristics as affected by agricultural management and earthworm activity. Eur J Soil Sci 56:453–467. https://doi.org/10.1111/j.1365-2389.2004.00696.x
Reuter G (1991) 35 Jahre Rostocker Dauerversuche. I. Entwicklung der Humusgehalte. 11. Entwicklung chemischer Bodeneigenschaften. Arch Acker- u Pflanzenb Bodenkd 35:357–374
Römkens PFAM, van der Plicht J, Hassink J (1999) Soil organic matter dynamics after the conversion of arable land to pasture. Biol Fertil Soils 28:277–284. https://doi.org/10.1007/s003740050494
Rosenvald K, Kuznetsova T, Ostonen I, Truu M, Truu J, Uri V, Lohmus K (2011) Rhizosphere effect and fine-root morphological adaptations in a chronosequence of silver birch stands on reclaimed oil shale post-mining areas. Ecol Eng 37:1027–1034. https://doi.org/10.1016/j.ecoleng.2010.05.011
Rumpel C, Seraphin A, Dignac MF, Michaelis W, Eusterhues K, Kögel-Knabner I (2005) Effect of base hydrolysis on the chemical composition of organic matter of an acid forest soil. Org Geochem 36:239–249. https://doi.org/10.1016/j.orggeochem.2004.08.001
Rytter R-M, Rytter L (2020) Changes in soil chemistry in an afforestation experiment with five tree species. Plant Soil 456:425–437. https://doi.org/10.1007/s11104-020-04726-7
Saetre P, Brandtberg PO, Lundkvist H, Bengtsson J (1999) Soil organisms and carbon, nitrogen and phosphorus mineralization in Norway spruce and mixed Norway spruce-birch stands. Biol Fert Soils 28:382–388. https://doi.org/10.1007/s003740050508
Shen G, Chen D, Wu Y, Liu L, Liu Ch (2019) Spatial patterns and estimates of global forest litterfall. Ecosphere 10(2):e02587. https://doi.org/10.1002/ecs2.2587
Šimanský V, Juriga M, Jonczak J, Uzarowicz Ł, Stępień W (2019) How relationships between soil organic matter parameters and soil structure characteristics are affected by the long-term fertilization of a sandy soil. Geoderma 342:75–84. https://doi.org/10.1016/j.geoderma.2019.02.020
Six J, Elliott ET, Paustian K, Doran JW (1998) Aggregation and soil organic matter accumulation in cultivated and native grassland soils. Soil Sci Soc Am J 62:1367–1377. https://doi.org/10.2136/sssaj1998.03615995006200050032x
Smal H, Olszewska M (2008) The effect afforestation with Scots pine (Pinus silvestris L.) of sandy post-arable soils on their selected properties. II. Reaction, carbon, nitrogen and phosphorus. Plant Soil 305:171–187. https://doi.org/10.1007/s11104-008-9538-z
Smolander A, Kitunen V (2002) Soil microbial activities and characteristics of dissolved organic C and N in relation to tree species. Soil Biol Biochem 34:651–660. https://doi.org/10.1016/S0038-0717(01)00227-9
Spohn M, Novák TJ, Incze J, Giani L (2016) Dynamics of soil carbon, nitrogen, and phosphorus in calcareous soils after land-use abandonment – a chronosequence study. Plant Soil 401:185–196. https://doi.org/10.1007/s11104-015-2513-6
Špulák O, Souček J, Bartoš J, Kacálek D (2010) Potential of young stands with birch dominance established by succession on abandoned agricultural land. For Res Rep 55(3):165–170
Stevenson FJ (1994) Humus chemistry. Wiley, Chichester
Straková P, Anttila J, Spetz P, Kitunen V, Tapanila T, Laiho R (2010) Litter quality and its response to water level drawdown in boreal peatlands at plant species and community level. Plant Soil 335:501–520. https://doi.org/10.1007/s11104-010-0447-6
Tanner LH, Smith DL, Curry J, Twist J (2014) Effect of land use change on carbon content and CO2 flux of cloud forest soils, Santa Elena, Costa Rica. Open J Soil Sci 4:64–71. https://doi.org/10.4236/ojss.2014.42009
Taylor BR, Parkinson D, Parsons WJF (1989) Nitrogen and lignin content as predictors of litter decay rates: a microcosm test. Ecology 70:97–104. https://doi.org/10.2307/1938416
Tripathi SK, Sumida A, Shibata H, Ono K, Uemura S, Kodama Y, Hara T (2006) Leaf litterfall and decomposition of different above- and belowground parts of birch (Betula ermanii) trees and dwarf bamboo (Sasa kurilensis) shrubs in a young secondary forest in Northern Japan. Biol Fertil Soils 43:237–246. https://doi.org/10.1007/s00374-006-0100-y
Ukalska-Jaruga A, Klimkowicz-Pawlas A, Smreczak B (2019) Characterization of organic matter fractions in the top layer of soils under different land uses in Central-Eastern Europe. Soil Use Manage 35:595–606. https://doi.org/10.1111/sum.12514
Ukonmaanaho L, Merilä P, Nöjd P, Nieminen TM (2008) Litterfall production and nutrient return to the forest floor in Scots pine and Norway spruce stands in Finland. Boreal Environ Res 13(SB):67–91
Ussiri DAN, Johnson CE (2003) Characterization of organic matter in a northern hardwood forest soil by 13C NMR spectroscopy and chemical methods. Geoderma 111:123–149. https://doi.org/10.1016/S0016-7061(02)00257-4
van Calster H, Baeten L, Verheyen K, de Keersmaeker L, Dekeyser S, Rogister JE, Hermy M (2008) Diverging effects of overstorey conversion scenarios on the understorey vegetation in a former coppice-with-standards forest. For Ecol Manag 256:519–528. https://doi.org/10.1016/j.foreco.2008.04.042
Vladychenskii AS, Shcheglov KA, Manakhov DV (2007) Humus content and distribution in the profile of dark gray forest soils under different plants. Mos Univ Soil Sci Bull 62(1):22–28. https://doi.org/10.3103/S0147687407010048
Wang Z, Liu S, Huang C, Liu Y, Bu Z (2017) Impact of land use change on profile distributions of organic carbon fractions in peat and mineral soils in Northeast China. CATENA 152:1–8. https://doi.org/10.1016/j.catena.2016.12.022
Wei X, Li X, Jia X, Shao M (2013) Accumulation of soil organic carbon in aggregates after afforestation on abandoned farmland. Biol Fert Soils 49:637–646. https://doi.org/10.1007/s00374-012-0754-6
Wilcox A (1998) Early plant succession on former arable land. Agr Ecosys Environ 69:143–157. https://doi.org/10.1016/S0167-8809(98)00104-2
Wilson MA (1987) NMR-Techniques and application in geochemistry and soil chemistry. Pergamon Press, Oxford
Yan DF, Wang DC, Yang XT (2016) Effects of surface root system on soil infiltration at different vegetation types in Danjiangkou Reservoir area. Sci Soil Water Conserv 14:35–44. https://doi.org/10.16843/j.sswc.2016.03.005
Zasada M, Bijak S, Bronisz K, Bronisz A, Gawęda T (2014) Biomass dynamics in young silver birch stands on post-agricultural lands in central Poland. Drewno 57(192):29–39. https://doi.org/10.12841/wood.1644-3985.S07.02
Żdanow JA (1965) Sriedniaja stepen okislenija ugleroda i nezamenimost aminokislot. Biochimija 30(6):1257–1259
Acknowledgements
The research was supported by the National Science Centre, Poland, project no. 2018/31/B/NZ9/01994, title “Transformation of post-arable soils due to afforestation with birch”
Funding
The research was supported by the National Science Centre, Poland, project no. 2018/31/B/NZ9/01994, title “Transformation of post-arable soils due to afforestation with birch”.
Author information
Authors and Affiliations
Contributions
JJ—planning and general management of the studies, isolation and purification of humic acids, 13C-NMR and elemental analysis of humic acids, manuscript concept and writing. LO—field studies, humus fractionation, manuscript writing. EP—determination of basic soil properties. AC—field studies, manuscript writing. ER—field studies, manuscript writing. SS—characterisation of the study area, manuscript writing. IO—field studies, manuscript writing. JO—collecting data on stands, field studies. BK—laboratory analysis, manuscript writing. MK—field studies, sample preparation for further analysis. UJ—field studies. EW-G—statistical analysis.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper. The authors declare the following financial interests/personal relationships which may be considered as potential competing interests: Jerzy Jonczak reports financial support was provided by National Science Centre Poland.
Additional information
Communicated by Agustin Merino.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Jonczak, J., Oktaba, L., Pawłowicz, E. et al. Soil organic matter transformation influenced by silver birch (Betula pendula Roth) succession on abandoned from agricultural production sandy soil. Eur J Forest Res 142, 367–379 (2023). https://doi.org/10.1007/s10342-022-01527-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10342-022-01527-8