Abstract
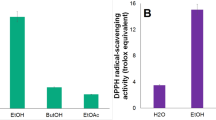
The present study assessed the phytochemicals and biological activities of Rosa persica root extracts. The phytoconstituents of R. persica root were quantified by gas chromatography-mass spectrometry (GC-MS) and ultra-performance liquid chromatography-electrospray ionization-photodiode array detection-mass spectrometry (UPLC-ESI-PDA-MSn). Concentrations of total phenolic content (TPC), total flavonoid content (TFC), total proanthocyanidin content (TPAC), and antioxidant capacity were also measured in aqueous and ethanol (EtOH) extracts. Their bioactivities in terms of antiviral (against two cell lines), antimicrobial (against eight bacteria strains), antifungal (against two fungal strains), and cytotoxic (against eight cell lines) effects were assessed. The maximum effects were found in EtOH extract of R. persica root. The main components of the R. persica essential oil (RPEO) were methyl eugenol (73.1%) and geranyl acetone (10.3%). UPLC–ESI-PDA–MSn analysis showed that gallic acid (GA) was found in the highest concentration in the EtOH extract (461.80 ± 90.3 μg/g). In the antiviral assay, the half maximal inhibitory concentrations (IC50) of EtOH extract for Coxsackievirus B3 (CV-B3) and Coxsackievirus B4 (CV-B4) were 29.26 ± 0.5 and 24.70 ± 0.2 µg/mL, respectively. The cytotoxic concentrations (CC50) calculated for EtOH extract against both cell lines were significantly lower than aqueous extract (89.13 ± 0.3 and 75.03 ± 0.1 µg/mL, respectively). The most sensitive bacteria strain in both extracts was Staphylococcus aureus. Activity in fungal strains, Candida albicans was more sensitive than Aspergillus niger. The current cytotoxicity results between the two extracts of R. persica showed that both had the highest and lowest cytotoxic activity against human glioblastoma (U-87-MG) and human breast cancer (MDA-MB-231) cell lines, respectively.

Similar content being viewed by others
References
Alcântara MA et al (2019) Effect of the solvent composition on the profile of phenolic compounds extracted from chia seeds. Food Chem 275:489–496
Ambigaipalan P, de Camargo AC, Shahidi F (2017) Identification of phenolic antioxidants and bioactives of pomegranate seeds following juice extraction using HPLC-DAD-ESI-MSn. Food Chem 221:1883–1894
Amini M, Rineh MK, Yazdani M (2016) Study of the chemical compositions of Iranian rose flower essence oil (Rosa persica). Bulg Chem Commun 48:27–29
Azadmanesh R, Tatari M, Asgharzade A, Taghizadeh SF, Shakeri A (2021) GC/MS profiling and biological traits of eucalyptus globulus L. essential oil exposed to solid lipid nanoparticle (SLN). J Essent Oil Bear Plants 24:863–878
Aziman N, Abdullah N, Noor ZM, Kamarudin WSSW, Zulkifli KS (2014) Phytochemical profiles and antimicrobial activity of aromatic Malaysian herb extracts against food-borne pathogenic and food spoilage microorganisms. J Food Science 79:M583–M592
Basaki T et al (2009) Assessing Rosa persica genetic diversity using amplified fragment length polymorphisms analysis. Sci Hortic 120:538–543
Boso S, Gago P, Santiago JL, Álvarez-Acero I, Martínez MC (2019) Concentration of flavanols in red and white winemaking wastes (grape skins, seeds and bunch stems), musts, and final wines. Erwerbs-Obstbau 61:75–84
Chakraborty N, Chandra S, Acharya K (2017) Biochemical basis of improvement of defense in tomato plant against Fusarium wilt by CaCl2. Physiol Mol Biol Plants 23:581–596
Čulenová M et al (2019) Multiple In vitro biological effects of phenolic compounds from Morus alba root bark. J Ethnopharmacol 248:112296
Esmaeilzadeh Kenari R, Mohsenzadeh F, Amiri ZR (2014) Antioxidant activity and total phenolic compounds of Dezful sesame cake extracts obtained by classical and ultrasound-assisted extraction methods. Food Sci Nutr 2:426–435
Farhadi F, Iranshahi M, Taghizadeh SF, Asili J (2020) Volatile sulfur compounds: The possible metabolite pattern to identify the sources and types of asafoetida by headspace GC/MS analysis. Ind Crops Prod 155:112827
Fattahi A et al (2021) UPLC–PDA-ESI–QTOF–MS/MS and GC-MS analysis of Iranian Dracocephalum moldavica L. Food Sci Nutr 9(8):4278–4286
Ghavi ABA, Mazandarani M (2017) Aut ecology and phytochemical survey of ziziphora clinopodioides lam. With ethnopharmacology and floristic spectrum of medicinal plants in bovanlou region (northern khorasan province)
Gore M, Desai N (2014) Characterization of phytochemicals and evaluation of anti-cancer potential of Blumea eriantha DC. Physiol Mol Biol Plants 20:475–486
Handoussa H et al (2013) Anti-inflammatory and cytotoxic activities of dietary phenolics isolated from Corchorus olitorius and Vitis vinifera. J Funct Foods 5:1204–1216
Jassbi A, Zamanizadehnajarib S, Tahara S (2003) Polyphenolic antioxidant constituents of Rosa persica. J Chem Soc Pak 25:323–327
Kamble V, Attar U, Umdale S, Nimbalkar M, Ghane S, Gaikwad N (2020) Phytochemical analysis, antioxidant activities and optimized extraction of embelin from different genotypes of Embelia ribes Burm f.: a woody medicinal climber from Western Ghats of India. Physiol Mol Biol Plants 26:1855–1865
Koczka N, Stefanovits-Bányai É, Ombódi A (2018) Total polyphenol content and antioxidant capacity of rosehips of some rosa species. Medicines 5:84
Korkmaz M, Dogan NY (2018) Analysis of genetic relationships between wild roses (Rosa L. Spp.) growing in Turkey. Erwerbs-Obstbau 60:305–310
Kumar S, Pandey AK (2013) Chemistry and biological activities of flavonoids: an overview. Sci World J 2013:1–16
Li Y, Guo C, Yang J, Wei J, Xu J, Cheng S (2006) Evaluation of antioxidant properties of pomegranate peel extract in comparison with pomegranate pulp extract. Food Chem 96:254–260
Liu Y, Lu B, Peng J (2011) Hepatoprotective activity of the total flavonoids from Rosa laevigata Michx fruit in mice treated by paracetamol. Food Chem 125:719–725
Llorent-Martínez E et al (2017) Evaluation of antioxidant potential, enzyme inhibition activity and phenolic profile of Lathyrus cicera and Lathyrus digitatus: Potential sources of bioactive compounds for the food industry. Food Chem Toxicol 107:609–619
Majiya H, Galstyan A (2020) Dye extract of calyces of Hibiscus sabdariffa has photodynamic antibacterial activity: A prospect for sunlight-driven fresh produce sanitation. Food Sci Nutr 8:3200–3211
Mavi A, Terzi Z, Özgen U, Yildirim A, Coşkun M (2004) Antioxidant properties of some medicinal plants: Prangos ferulacea (Apiaceae), Sedum sempervivoides (Crassulaceae), malva neglecta (malvaceae), Cruciata taurica (Rubiaceae), Rosa pimpinellifolia (Rosaceae), Galium verum subsp. verum (Rubiaceae), urtica dioica (urticaceae). Biol Pharm Bull 27:702–705
Messi AN et al (2016) Phenolic compounds from the roots of Ochna schweinfurthiana and their antioxidant and antiplasmodial activities. Phytochem Lett 17:119–125
Moradkhani S, Rezaei-Dehghanzadeh T, Nili-Ahmadabadi A (2020) Rosa persica hydroalcoholic extract improves cadmium-hepatotoxicity by modulating oxidative damage and tumor necrosis factor-alpha status. Environ Sci Pollut Res Int 27:31259–31268. https://doi.org/10.1007/s11356-020-09450-4
Naik PM, Al-Khayri JM (2020) Influence of culture parameters on phenolics, flavonoids and antioxidant activity in cell culture extracts of date palm (Phoenix dactylifera L.). Erwerbs-Obstbau 62:181–188
Rocchetti G, Senizza B, Zengin G, Senkardes I, Sadeer NB, Mahomoodally MF, Lucini L (2019) Metabolomics-based profiling with chemometric approach to delineate the bio-pharmaceutical properties of fruit extracts from Ligustrum vulgare L. Ind Crops Prod 140:111635
Sadat-Hosseini M, Farajpour M, Boroomand N, Solaimani-Sardou F (2017) Ethnopharmacological studies of indigenous medicinal plants in the south of Kerman, Iran. J Ethnopharmacol 199:194–204
Sadraei H, Asghari G, Emami S (2013) Inhibitory effect of Rosa damascena Mill flower essential oil, geraniol and citronellol on rat ileum contraction. Res Pharma Sci 8:17
Sadraei H, Asghari G, Jalali F (2016) Assessment of hydroalcoholic and hexane extracts of Rosa persica Mich. flower on rat ileum spasm. Res Pharma Sci 11:160
Sarikurkcu C, Ozer MS, Tlili N (2019) LC–ESI–MS/MS characterization of phytochemical and enzyme inhibitory effects of different solvent extract of Symphytum anatolicum. Ind Crops Prod 140:111666
Shakeri A et al (2019) LC-ESI/LTQOrbitrap/MS/MS and GC–MS profiling of Stachys parviflora L. and evaluation of its biological activities. J Pharm Biomed Anal 168:209–216
Shiota S, Shimizu M, Sugiyama J, Morita Y, Mizushima T, Tsuchiya T (2004) Mechanisms of action of corilagin and tellimagrandin I that remarkably potentiate the activity of beta-lactams against methicillin-resistant Staphylococcus aureus. Microbiol Immunol 48:67–73. https://doi.org/10.1111/j.1348-0421.2004.tb03489.x
Singh C, Upadhyay R, Tiwari KN (2022) Comparative analysis of the seasonal influence on polyphenolic content, antioxidant capacity, identification of bioactive constituents and hepatoprotective biomarkers by in silico docking analysis in Premna integrifolia L. Physiol Mol Biol Plants 28(1):223–249
Taghizadeh SF, Rezaee R, Davarynejad G, Karimi G, Nemati SH, Asili J (2018) Phenolic profile and antioxidant activity of Pistacia vera var. Sarakhs hull and kernel extracts: the influence of different solvents. J Food Meas Charact 12:2138–2144
Taghizadeh SF, Rezaee R, Badiebostan H, Giesy JP, Karimi G (2019a) Occurrence of mycotoxins in rice consumed by Iranians: a probabilistic assessment of risk to health. Food Addit Contam Part A 37(2):342–354
Taghizadeh SF, Rezaee R, Mehmandoust M, Madarshahi FS, Tsatsakis A, Karimi G (2019b) Coronatine elicitation alters chemical composition and biological properties of cumin seed essential oil. Microb Pathog 130:253–258
Taghizadeh SF, Rezaee R, Mehmandoust M, Badibostan H, Karimi G (2020) Assessment of in vitro bioactivities of Pis v 1 (2S albumin) and Pis v 2.0101 (11S globulin) proteins derived from pistachio (Pistacia vera L.). J Food Meas Charact 14:1054–1063
Taghizadeh SF, Azizi M, Asili J, Madarshahi FS, Rakhshandeh H, Fujii Y (2021a) Therapeutic peptides of Mucuna pruriens L.: Anti-genotoxic molecules against human hepatocellular carcinoma and hepatitis C virus. Food Sci Nutr 9(6):2908–2914
Taghizadeh SF, Azizi M, Rezaee R, Madarshahi FS, Mehmandoust M, Karimi G, Asili J (2021b) Cytotoxic activity of cis-(E)-and trans-(Z)-spiroethers isolated from various Arnebia species. S Afr J Bot 142:114–123
Tahirović A, Bašić N (2017) Determination of phenolic content and antioxidant activity of Rosa canina L. fruits in different extraction systems. Work Fac For Univ Sarajevo 1:47–59
Tao X et al (2016) Total flavonoids from Rosa laevigata Michx fruit ameliorates hepatic ischemia/reperfusion injury through inhibition of oxidative stress and inflammation in rats. Nutrients 8:418
Trandafir I, Cosmulescu S (2020) Total phenolic content, antioxidant capacity and individual phenolic compounds of defatted kernel from different cultivars of walnut. Erwerbs-Obstbau 62:309–314
Wang H, Helliwell K (2001) Determination of flavonols in green and black tea leaves and green tea infusions by high-performance liquid chromatography. Food Res Int 34:223–227
Xu X, Guo S, Hao X, Ma H, Bai Y, Huang Y (2018) Improving antioxidant and antiproliferative activities of colla corii asini hydrolysates using ginkgo biloba extracts. Food Sci Nutr 6:765–772
Yasa N, Masoumi F, Rouhani RS, Haji AA (2009) Chemical composition and antioxidant activity of the extract and essential oil of Rosa damascena from Iran, population of Guilan
Yi O, Jovel EM, Towers GH, Wahbe TR, Cho D (2007) Antioxidant and antimicrobial activities of native Rosa sp. from British Columbia, Canada. Int J Food Sci Nutr 58:178–189. https://doi.org/10.1080/09637480601121318
Yu J, Ahmedna M, Goktepe I (2005) Effects of processing methods and extraction solvents on concentration and antioxidant activity of peanut skin phenolics. Food Chem 90:199–206
Zhang S et al (2013) Protection of the flavonoid fraction from Rosa laevigata Michx fruit against carbon tetrachloride-induced acute liver injury in mice. Food Chem Toxicol 55:60–69
Zor M, Şengül M, Topdaş EF, Yılmaz B (2022) Physicochemical properties, antioxidant activities, and chemical compositions of extracts obtained from Rosa pimpinellifolia L. flesh and seeds using different methods and solvents. Erwerbs-Obstbau. https://doi.org/10.1007/s10341-022-00676-9
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
A. Koohestanian, M. Tatari, M. Samadi Kazemi, A. Asgharzade, and S.F. Taghizadeh declare that they have no competing interests.
Rights and permissions
Springer Nature oder sein Lizenzgeber hält die ausschließlichen Nutzungsrechte an diesem Artikel kraft eines Verlagsvertrags mit dem/den Autor*in(nen) oder anderen Rechteinhaber*in(nen); die Selbstarchivierung der akzeptierten Manuskriptversion dieses Artikels durch Autor*in(nen) unterliegt ausschließlich den Bedingungen dieses Verlagsvertrags und dem geltenden Recht.
About this article
Cite this article
Koohestanian, A., Tatari, M., Samadi Kazemi, M. et al. Phytochemicals, Antioxidant Activity, and Biological Activities of Rosa persica Root. Erwerbs-Obstbau 65, 1127–1138 (2023). https://doi.org/10.1007/s10341-022-00740-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10341-022-00740-4