Abstract
Plasma-activated water (PAW) is receiving increased attention as a booster of seed germination and seedling vigor, and some studies have described use of PAW to manage crop pathogens. Here, we examined physicochemical properties of two PAWs (referred to as PAW 6.0 and 9.4 min with atmospheric plasma jet) and assessed “their indirect effects” (applied as supplementary irrigation) on host suitability of tomato plants (Solanum lycopersicum L.) to two-spotted spider mites (Tetranychus urticae Koch). Exposure of water to cold plasma significantly lowered pH and increased concentrations of H2O2, NO2−, and NO3−. Supplementary PAW irrigations elicited significant increases in leaf composition of several elements (N, P, K S, Ca, and Mg), leaf reflectance, plant size, and trichome densities (except non-glandular trichomes on the adaxial surface). Preference bioassays revealed significant avoidance of settling and reduced oviposition by two-spotted spider mites on leaf discs from PAW-irrigated plants compared to those from untreated control plants. Performance bioassays showed a significant decrease in two-spotted spider mite populations on PAW-irrigated plants. Results presented in this study provide comprehensive support to the hypothesis that indirect effects of supplementary PAW irrigation significantly reduce host plant suitability to two-spotted spider mites. PAW 6.0 may be slightly better than PAW 9.4, and this difference in performance is discussed in this study. Applications of PAW as supplementary irrigation are likely highly compatible with other IPM tactics and should be considered an innovative and sustainable component in twenty-first-century pest management.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Key message
-
Cold plasma treatment alters water properties, increasing H2O2, NO2−, and NO3− concentrations.
-
Plasma activated water (PAW) irrigation boosts leaf composition, reflectance, plant size, and trichome densities.
-
PAW-irrigated plants reduce settling and oviposition avoidance of two-spotted spider mites.
-
PAW-irrigated plants reduce two-spotted spider mite populations.
-
PAW should be considered an innovative and sustainable component in 21st century pest management.
Introduction
Ensuring sustainable crop productivity and global food security is one of the defining challenges of the new millennium (Farooq et al. 2019). Moreover, it is crucial to examine and promote technologies that can boost food production without causing adverse risks to surrounding ecosystems and urban environments. In this context, there has been growing interest in use of “cold plasma technologies” in crop production (Xu et al. 2023; Guo et al. 2021; Yepez et al. 2022).
Cold plasma, also known as non-thermal plasma, is typically generated by applying a discharge (thermal, electromagnetic, or electric fields, microwave, and radio frequencies) to gases (air, Ar, O2, He, or N2) at low or atmospheric pressure (Guo et al. 2021). Cold plasma treatments produce a dynamic mixture of ions, new electrons, excited molecules, and free radicals (Guo et al. 2021; Thirumdas et al. 2015; Pankaj and Keener 2017). When applied to water, active compounds, like reactive oxygen and nitrogen species (RONS) (H2O2, NO2−, or NO3−), are generated (Gao et al. 2022; Han et al. 2023). Reactive species generated during the plasma activation process increase water salinity, electrical conductivity, and acidity (Chew et al. 2023). PAW has been shown to enhance seed germination, root and shoot growth, photosynthesis, nutrient availability, floral regulation, and yield (Fan et al. 2020; Vichiansan et al. 2023; Priatama et al. 2023; Sivachandiran and Khacef 2017; Škarpa et al. 2020; Guragain et al. 2021). It is also a disinfectant with antimicrobial properties against bacteria (Perez et al. 2019; Lee et al. 2023), fungi (Ambrico et al. 2020; Ju et al. 2023), and viruses (Filipić et al. 2019; Hanbal et al. 2018). Additionally, PAW has been demonstrated to possess significant insecticidal activity against citrus mealybug Planococcus citri (Risso) (Hemiptera: Pseudococcidae) (Ten Bosch et al. 2017).
The above-mentioned studies highlight the potential of PAW and its direct effects on pests (pathogens and insects). Less research attention has been given to the potential of PAW as a booster of plant defense mechanisms (Laurita et al. 2021; Zambon et al. 2020; Adhikari et al. 2019). This topic is here referred to as the “indirect effects” of PAW—how treatments (either foliar applications or via means of supplemental irrigation) may elicit significant reduction in host plant suitability to herbivorous pests. As part of resistance mechanisms against arthropod pests, plant trichomes and their chemical compounds play a significant role in crop protection (Simmons and Gurr 2005; Savi et al. 2022). Tomato (Solanum lycopersicum L.) plants, for instance, possess seven types of trichomes, which are categorized as non-glandular (types II, III, and V) and glandular (types I, IV, VI, and VII) (Simmons and Gurr 2005). Non-glandular trichomes can serve as physical barriers to micro-arthropods on the leaf surface while glandular trichomes act as physical and deterrent barriers to small herbivores, through their high viscosity of allelochemical secretions such as acyl sugars, methyl-ketones, and sesquiterpenes (Rakha et al. 2017; Savi 2019; Yang et al. 2023). Trichomes, when present in high densities, can suppress pest movement, or negatively impact pest survival, development, and reproduction (Savi 2019; Savi et al. 2022; Salazar-Mendoza et al. 2023). Furthermore, nutritional composition of plants may be positively correlated with suitability to herbivorous pests (Chaboussou 2004; Joern et al. 2012). If so, it may be possible to indirectly manage and suppress pest populations by manipulating element composition of crops (Altieri and Nicholls 2003; Huber et al. 2012). However, to our knowledge, it has not yet been explored to what extent PAW may be used to alter plant element composition or boost defensive traits, such as trichome density, in ways that would reduce suitability of crops to herbivorous pests.
Use of trichome density and element composition as response variables in studies of indirect plant responses to PAW, and other treatments, requires destructive sampling of crop leaves. Additionally, these response variables require processing of leaf materials and are hampered by their general “representativeness.” Meaning, trichome density can only feasibly be counted on small pieces of leaves from individual plants, so it is possible that within-leaf variation and vertical variation among leaves within canopies affect counts. Regarding element composition, costs of analyses may hamper inclusion of individual samples from each plant from all combinations of treatments and replications, so there may be statistical constraints due to few observations (degrees of freedom). Furthermore, selection of leaves to be included in each sample may also pose some concerns about subjectivity and representativeness of leaf materials being analyzed. As an attempt to address such challenges and to explore possible complementarity and highly innovative technologies, optical sensing of plants may be considered. As reviewed elsewhere (Li et al. 2017; Nansen 2016), when lighting conditions, projection angle, distance between camera and objects are quasi-constant over time and space, then reflectance (or transmission) features acquired from plant leaves may be used to quantify differences in qualitative traits among objected subjected in different categories (i.e., subjected to different treatments). Furthermore, suitability of a given host plant may be associated with its element composition, which is detectable based on features in leaf profiles (Nansen et al. 2021). Accordingly, leaf reflectance profiling may be proposed as an approach to, non-destructively, detect and diagnose effects of PAW treatments and provide valuable and quantitative insight into crop plant responses to PAW, other growth-promoting treatments.
As illustrated in Fig. 1, we examined the hypotheses that: (1) trichome density and element composition of tomato plants are significantly affected by supplementary PAW irrigation, (2) PAW irrigations elicit significant changes to leaf reflectance profiles, (3) based on preference bioassays, arthropod herbivores will show avoidance of PAW-irrigated plants, and (4) based on performance bioassays, arthropod pest populations will grow significantly slower on PAW-irrigated plants. To address these hypotheses, we used two-spotted spider mites, Tetranychus urticae Koch (Acari Tetranychidae) as a model herbivore. Two-spotted spider mite is an important herbivore that poses considerable damage to many plants including tomato due to their short life cycle and high reproductive capacity (Tiftikçi et al. 2020; Savi et al. 2021). Control of this important pest is widely based on application of synthetic miticides, which may lead to environmental contamination and evolution of resistance (Attia et al. 2013). Resistance development of this herbivore to 96 active ingredients has been reported (APRD, http://www.pesticideresistance.org/). Due to these issues, alternative control measures have been sought globally, including the use of predatory mites, such as Phytoseiulus persimilis Athias-Henriot (Acari: Phytoseiidae) (McMurtry 2013). However, implementation of biological control programs has not been widely adopted by commercial tomato growers due to inconsistent performance and need for extensive pest population monitoring (Weinblum et al. 2021). Therefore, it has been recommended that growers use a combination of soft miticides, and biological control methods, or mite-resistant plant genotypes (Tiftikçi et al. 2020, 2022; Lucini et al. 2015; Savi et al. 2022). Although these practices are somewhat effective, they may not always provide sufficient control over two-spotted spider mite populations, particularly under high-pressure conditions. Hence, additional management tactics are needed to complement these practices and enhance their effectiveness. Applications of supplementary irrigation with PAW are likely highly compatible with other IPM tactics and should be considered an innovative and sustainable component in twenty-first-century pest management.
Material and methods
Two-spotted spider mites
To establish two-spotted spider mite colony used in this study, some individuals were collected from soybean (Glycine max L.) at the University of California, UC Davis, USA. These mites were then reared on tomato plants (variety Micro-tom) for several generations in a greenhouse maintained at the following conditions: 25.2 ± 1 °C, 77 ± 10% RH, and a L:D 12:12 photoperiod.
Production and characterization of PAW
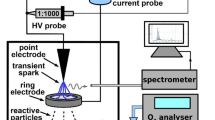
We evaluated two PAWs, in which UC Davis tap water (control) was treated for 6.0 min (PAW 6.0) and 9.4 min (PAW 9.4); PAW 6.0 and PAW 9.4 were specifically chosen for this study based on their observed higher responsiveness to plant growth in preliminary studies involving a broader range of PAWs. PAWs were generated using the Openair™ Plasma System from Plasmatreat USA, Inc., which included TS CD50 treatment station, FG5001 plasma generator equipped with 1KVA digital processor and 10,145 nozzle, CD50 plasma disinfection jet, EPU2 hydrogen peroxide evaporator system, DVE10 pressure supply control unit, HTR11 plasma high-voltage transformer, PTU2 high-voltage cabinet for two transformers and accessories, FLC air/O2 and flow control. We operated the system under the following parameters: voltage (302 V), air pressure (38–42 mBar), frequency (23 kHz), and current (12.6A). Each treatment consisted of 1 L of water in a 2.5-L Pyrex glass beaker (diameter 131 mm) with a stir bar maintained on a hot magnetic stirrer device operating at 600 rpm for continuous agitation during plasma treatment. To prevent water spillage during plasma treatment, a corrosion-resistant 316 stainless steel sheet with a central 10-cm-diameter opening, fitted for the plasma nozzle inlet, was placed atop the beaker. A consistent distance of 1 cm was maintained between the water surface and plasma nozzle inside the beaker. To ensure consistent performance throughout the treatment process, an air compressor was regularly monitored and maintained at 85 psi. 24 h after treatment, we measured the physicochemical properties of PAW 6.0, PAW 9.4, and control. Levels of pH and electrical conductivity (EC; indicative of active ions in water) were monitored using pH/EC meter (OHAUS-AB331M-F) while oxidation–reduction potential (ORP; ability of solution to gain or lose electrons) was estimated through ORP meter (Hanna HI2202-01). Concentrations of reactive species, such as nitrate (NO3−) and nitrite (NO2−) as well as hydrogen peroxide (H2O2), were obtained using a benchtop multiparameter photometer (HI83399-01). Physicochemical parameters measurements were collected based on a completely randomized design with eight replicates (different times that the PAW was prepared) per treatment. PAW samples were stored in glass containers in a refrigerator (5 °C) for a maximum of four days before application to tomato plants.
Plant growing conditions
Tomato plants variety BQ273 were grown in the greenhouse facilities at UC Davis. Initially, seeds were sown in tray cells filled with a homogeneous mixture of pumice, sphagnum peat moss, sand, redwood sawdust, and dolomite, at a ratio of 5.23 kg per m3 of soil mix, which had been sterilized in an autoclave at 121 °C for 1 h. Trays were then placed in a greenhouse, and automated sprinkling irrigation using a Mix Rite injector (Model 2502) provided water and fertilizer four times a day (at 7 am, 10 am, 2 pm, and 5 pm). The fertilizer was prepared using UC Davis modified Hoagland's solution with the following element composition (values in ppm): N = 150, P = 50, K = 200, Ca = 175, Mg = 55, S = 120, Fe = 2.5, Cu = 0.02, B = 0.5, Mn = 0.5, Mo = 0.01, and Zn = 0.05. Four weeks after sowing, seedlings were individually transplanted to 1.5-L pots 80% filled with a homogeneous mixture of soil, sand, and tanned bovine manure (1:1:1) autoclaved at 121 °C for 1 h. Thirty pots were randomly divided into three treatment groups (PAW 6.0, PAW 9.4, and control). Pots were maintained in a greenhouse under the following conditions: 25.2 ± 1 °C, 77 ± 10% RH, and a L:D 12:12 photoperiod. A 35 ml water and fertilizer solution (prepared at rates previously mentioned) was supplied twice a day (at 7 am and 6 pm) to the pots through automated drip irrigation. Additionally, 50 ml of PAW 6.0, PAW 9.4, or tap water was applied once a day, between 11 am and 12 noon, to individual plants until the end of the experiments. Thus, PAW treatments represented 42% of total water supplied to each tomato plant.
Element composition and trichome densities
After two weeks of treatments, leaf samples were collected from the median third of the plant canopy for analysis of elemental composition (N, P, K, Ca, Mg, S, Fe, Cu, B, Mn, Zn and Na). Each sample consisted of 0.5 g of dried leaflet material obtained from two plants, for a total of four samples per treatment. Element composition analyses were performed by University of California Davis analytical laboratory, and standard methods for analyses can be found at https://anlab.ucdavis.edu. Regarding trichome assessment (Fig. 1), five leaflets excised from the median third of the canopy of each plant amounting to 50 leaflets per treatment were examined. All glandular and non-glandular trichomes were counted in an area of 1 mm2 on the abaxial and adaxial sides close to the middle portion of the main vein. We used 10× magnification microscope (SZ61; Olympus SZ61 Trinocular Stereo Zoom Microscope, 8x-40x, SZ2-LGB Illuminator Camera), equipped with a digital camera (Olympus DP27) connected to a computer software, Cell Sens Entry.
Hyperspectral imaging
After two weeks of supplementary PAW irrigation, hyperspectral reflectance data were collected from all tomato plants (Fig. 1). We utilized a push-broom hyperspectral camera (PIKA L; www.resonon.com) mounted on a custom-built robotic rail system positioned approximately 1.5 m above the plant canopy, achieving a spatial resolution of approximately 9 pixels mm−2 (Nguyen et al. 2020; Nansen et al. 2021). The hyperspectral camera acquires data in 150 spectral bands ranging from 380 to 1015 nm, with a spectral resolution of 4.2 nm. Light source consisted of 12 and 15 W, 12 V halogen light bulbs placed on either side of the lens. White calibration was performed using a piece of white Teflon; both dark and white calibrations were carried out immediately before each imaging event to obtain relative reflectance. Hyperspectral imaging data were collected around 2 pm inside a dark room, maintained at 20–23 °C and 50–75% RH. Number of green pixels (after radiometric filtering to omit background pixels) per tomato plant was determined and used as an indicator of plant size.
Preference bioassays
Attractiveness and oviposition preferences of two-spotted spider mites were assessed through two-choice bioassays. The experimental unit involved placing two 30-mm leaf discs (each from a different treatment), with their abaxial surface facing up and made from leaves excised from the middle third of the canopy of 35-day-old tomato plants (irrigated with treatments for two weeks), inside a Petri dish (10 cm in diameter and 1.5 cm high). The Petri dish contained a 1-cm-thick foam mat lined with hydrophilic cotton soaked in deionized water (Fig. 1). Leaf discs were positioned in close proximity and connected by a plastic piece to allow the movement of mites from one to the other. Ten 6-day-old adult females were introduced into the center of the plastic piece. The experimental units were maintained under controlled conditions in a growth chamber with the following settings: 25.2 ± 1 °C, 77 ± 10% RH, and a L:D 12:12 photoperiod. The numbers of two-spotted spider mite females and their respective eggs on each leaf disc were recorded after 24 h. The experimental design was fully randomized, with 20 replicates for each of the three paired combinations (control–PAW 6.0, control–PAW 9.4, and PAW 6.0–PAW 9.4).
Performance bioassays
After 20 days of treatments, all tomato plants were infested with two-spotted spider mites (Fig. 1), placing 15 adult females on each of the second and fourth leaves (counted from the bottom). Population growth of two-spotted spider mites was monitored on all leaves on each plant at weeks 1, 2, and 3, by counting the numbers of mobile stages (larvae, nymphs–pooling protonymphs and deutonymphs–and adults) with a magnifying glass 10x. Upon completion of the last evaluation, all leaves of each plant were collected to count the number of eggs on each of them, under a stereomicroscope.
Data analyses
All data processing, analyses, and classifications were performed in R v3.6.1 (The R Foundation for Statistical Computing, Vienna, Austria). Leaf compositions of N, K, Ca, Mg, Zn and Mn followed Shapiro assumptions of normality (library ‘dplyr’) and were therefore subjected to Tukey HSD analysis of variance. Conversely, leaf compositions of P, S, B, Fe, and Cu and physicochemical properties did not meet homoscedasticity and normality assumptions, even after transformations; accordingly, these variables were analyzed based on nonparametric Kruskal–Wallis’ comparison of averages of treatments (library ‘AgroR’). Similarly, average trichome densities in response to treatments did not meet homoscedasticity and normality assumptions and were therefore analyzed using a Generalized Linear Model (GLM) with negative binomial distribution (library ‘AgroR’). In two-choice and performance bioassays, counts of adult female spider mites and eggs were analyzed using GLM with binomial error distribution. In case of significant differences between treatments, multiple comparisons for GLM models were performed using “emmeans” function in the “multcomp” library and Šidák-adjusted confidence intervals. Furthermore, principal component analysis (library ‘devtools’) was performed with element composition, trichome densities, and bioassay data as explanatory variables.
Tukey analysis of variance was used to examine average numbers of pixels per tomato plant among the three treatments. To compare leaf reflectance among treatments, random subsamples of 1000 green pixels were drawn from each tomato plant (total of 30,000 pixels). Leaf reflectance among treatments was classified based on support vector machine (svm) (library ‘e1071’) with radial kernel function and no specific hyperparameters (i.e., cost or gamma). Kappa values were generated for the assessment of classification performance and interpreted as follows: 0 = poor, 0.01–0.20 = slight, 0.21–0.40 = fair, 0.41–0.60 = moderate, 0.61–0.80 = substantial, and 0.81–1.00 = almost perfect (Landis and Koch 1977). In addition, we included tenfold cross-validation (Fauvel et al. 2015; Huang et al. 2019; Jin et al. 2009). In k-fold cross-validation, training data sets are divided into 'k' equal portions, which in this study was set at 10 [so 1500 observations (pixels) in each portion]. The classification model is trained on 'k − 1' of these portions, while the remaining portion is used for validation. This process is repeated 'k' times, with each fold serving as validation, and results from 'k' tests are averaged to produce a single estimation of model performance.
Results
Physicochemical properties of the water
pH of PAW 6.0 and PAW 9.4 elicited significant reductions (χ2 = 23.26, df = 2, p < 0.001) compared to the control (Fig. 2a). Electrical conductivity (EC) increased significantly (χ2 = 23.14, df = 2, p < 0.001) with significantly higher values for PAW 9.4 intermediate for that PAW 6.0 and lower in control. ORP followed the same trend (χ2 = 23.16, df = 2, p < 0.001), being also significantly higher for PAW 9.4 (563.2), intermediate for PAW 6.0, and lower for control (Fig. 2b). Concentration of H2O2 in both PAW 6.0 and PAW 9.4 increased significantly (χ2 = 12.3, df = 2, p = 0.002), compared to control (0.41). Concentrations of NO2−(χ2 = 23.67, df = 2, p < 0.001) and NO3−(χ2 = 23.15, df = 2, p < 0.001) increased substantially, with higher values for PAW 9.4, intermediate for that of PAW 6.0 and lower for control (Fig. 2c).
a pH, conductivity, b oxidation–reduction potential (ORP), hydrogen peroxide concentration (H2O2), c concentrations of nitrate (NO2−) and nitrite (NO3−) in untreated tap water (control) and 6.0 (PAW 6.0) and 9.4 min (PAW 9.4) plasma-activated water. Lines followed by different letters are significantly different (Kruskal–Wallis's test, p < 0.05)
Element composition and trichome densities
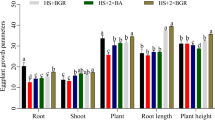
Compared to control, both PAW 6.0 and PAW 9.4 irrigations significantly increased the accumulation of N, P, K S, Ca, and Mg in tomato leaves, but decreased Zn and Fe content (Table 1). While PAW 6.0 increased B accumulation, PAW 9.4 treatment reduced it compared to control. No significant differences were observed between control and both PAW 6.0 and PAW 9.4 irrigations for Mn and Cu.
PAW-irrigated plants showed a significant increase in the numbers of non-glandular and glandular trichomes compared to control plants. This increase was observed when considering the abaxial and adaxial leaf surfaces together or separately, except for non-glandular trichomes on the adaxial surface (Table 2).
Hyperspectral imaging
Average size of tomato plants (based on green pixels) varied significantly among all three treatments, with PAW 6.0 plant being the largest (F = 42.36, df = 2, 27, p < 0.001) (Fig. 3a and b). Thus, it appeared that especially PAW 6.0 positively affected overall plant growth. Based on k = 10 validation, 30,000 pixels (10,000 from each treatment) were classified with 61% accuracy. In addition, the svm classification was associated with a kappa value = 0.57, which is considered “moderate” accuracy (Landis and Koch 1977). In the interpretation of the confusion matrix of classified pixels (Table 3), control pixels were classified with only 9% mis-classification, suggesting a marked difference between control plants and plants receiving supplementary PAW irrigation. The same analysis also showed that only 10–14% of pixels from PAW 6.0 and PAW 9.4 were mis-classified as control pixels. Thus, classification of hyperspectral imaging data provided strong support for the claim that this technology can be used to assess differences between PAW-irrigated plants. The difference between treatments was greater in spectral bands around 445 nm and between 700 and 1105 nm wavelength, as indicated by the separation of the three lines in Fig. 4a, and the height of the PAW lines in relation to the fixed control line (Fig. 4b).
Representative images of plants grown under different treatments a control (untreated tap water), PAW 6.0, and PAW 9.4. The two PAWs corresponded to plasma activation of water for 6.0 (PAW 6.0) and 9.4 min (PAW 9.4). Average (± SE) number of pixels per tomato plant from the three treatment, which is a relative indicator of canopy size
a Leaf reflectance data from plant grown under grown under different treatments control (untreated tap water), PAW 6.0, and PAW 9.4. The two PAWs corresponded to plasma activation of water for 6.0 (PAW 6.0) and 9.4 min (PAW 9.4). b Relative difference of PAW treatments compared to control (PAW/Control) with red line denoting no difference = 1
Preference bioassay
Out of the 10 females tested on each leaf disc (n = 20) in each two-choice test, 83.5%, 84.5%, and 95.5% chose one of the two options: control vs. PAW 6.0, control vs. PAW 9.4, and PAW 6.0 vs. PAW 9.4, respectively (Fig. 5a). The number of settled mites was significantly lower on leaf discs from both PAW 6.0 (χ2 = 18.38, df = 1, p < 0.001)- and PAW 9.4 (χ2 = 14.602, df = 1, p < 0.001)-irrigated plants compared to those of the control. No significant difference was observed for the number of settled mites in test involving leaf discs from PAW 6.0- and PAW 9.4-irrigated plants (χ2 = 0.09, df = 1, p = 0.75). Similarly, there was no significant difference in the per capita daily oviposition rate between leaf discs in tests involving PAW 6.0 vs. PAW 9.4 (χ2 = 0.24, df = 1, p = 0.62), or control vs PAW 9.4 (χ2 = 1.64, df = 1, p = 0.19), but it was significantly lower on PAW 6.0 leaf discs in test involving PAW 6.0 vs control (χ2 = 10.32, df = 1, p = 0.001) (Fig. 5b).
Preference bioassays results. Average (± SE) percentages of females of two-spotted spider mites (a) and per capita daily oviposition rate (b) in leaf discs from both PAW-irrigated and untreated tomato plants through dual-choice tests 24 h after release in dual-choice leaf disc assay (initial number of females = 10; number of replicates = 20), contrasting control versus PAW 6.0, control versus PAW 9.4, and PAW 6.0 versus PAW 9.4. Asterisk (*) indicates significant preference for one of the options (GLM with two side binomial tests: p < 0.05; df = 1)
Performance bioassay
Significant effects of PAW-irrigated plants or control (χ2 = 138.821, df = 2; p < 0.001), time (χ2 = 171.69, df = 3; p < 0.001) and their interaction (χ2 = 36.38, df = 6; p < 0.001) were observed on population growth of mobile two-spotted spider mites (Fig. 6a). One week after infestation, PAW 6.0- or PAW 9.4-irrigated plants showed a significant delay in population growth of mobile spider mites compared to control. Population growth on PAW 6.0, PAW 9.4, or control treatments 2 weeks after infestation followed the same trend reported for the previous week. After three weeks of the infestation, a similar pattern was observed for the number of mobile mites, also lower on tomato plants grown under PAW 6.0, and PAW 9.4 compared with control. In the destructive measurement evaluation, number of eggs was significantly (χ2 = 29.79, df = 2; p < 0.001) lower on PAW 6.0- and PAW 9.4-irrigated plants than in the control plants (Fig. 6b).
Performance results. Average number (± SE) of a mobile two-spotted spider mites (TSSM) at weeks 0, 1, 2, and 3 after mite infestation and b eggs recorded in destructive measurements (3-week after infestation) on plants growth under control or PAW treatments (initial females infested/ plant = 30, number of replicates = 10)
Principal component analysis
Principal component analysis was deployed to element composition, trichome, and bioassay data to elucidate trends (Fig. 7). Except for B and PAW 6.0 (which were highly correlated with PCA 2), all variables were positively or negatively correlated with PCA 1. That is, Cu, Fe, Zn, Mn, and all two-spotted spider mite parameters from preference and performance bioassays (preference by adults, oviposition, and population density) were positioned along the positive side of PCA 1 and highest in control. Conversely, Mg, N, P, K, S, and trichome densities were positioned along the negative side of PCA 1 and highest in PAW 6.0 and PAW 9.4. As PCA 1 explained more than seven times (88.04%) the variance explained by PCA 2 (11.96%), this principal component analysis suggested that most of the variance was caused by the difference between control, PAW 6.0, and PAW 9.4.
Principal component analysis (PCA) of element composition (Cu, Fe, K, Mg, Mn, S, and Zn), trichome densities (Gland T—glandular trichomes, and Non-gland. T—non-glandular trichomes), and bioassays data (Set avoid—avoidance of settling by two-spotted spider mites, Ovip avoid—avoidance of oviposition by two-spotted spider mites, Ep. Grow—egg population growth, MMPop Grow—mobile spider mite population growth) from tomato grown under control, PAW 6.0, and PAW 9.4 treatments
Discussion
In this study, we examined physicochemical properties of PAW 6.0 and PAW 9.4, and their indirect effects on the suitability of tomato plants as host of two-spotted spider mites. Our results showed significant differences in physicochemical parameters between PAW 6.0 and PAW 9.4 compared to control. We demonstrated that supplementary PAW irrigation elicited significant increase in N, P, K S, Ca, and Mg leaf retention, plant size (based on green pixels), leaf reflectance profiles, and trichome densities (except non-glandular trichomes on the adaxial surface). Additionally, preference bioassays revealed significant avoidance of two-spotted spider mites, as evidenced by reduced settling and oviposition on leaf discs from PAW-irrigated plants compared to control plants. Performance bioassays over three weeks showed significantly lower two-spotted spider mite populations on PAW-irrigated plants. Thus, results corroborated our study hypothesis that indirect effects of PAW might reduce host plant suitability against arthropod pest attack. These results provide novel insights into the versatile potential of PAW as a tool to manipulate plant resistance as part of IPM strategies.
PAW physicochemical properties
Most of physicochemical properties data from PAW 6.0 and PAW 9.4 in our study are comparable to those obtained by other studies using atmospheric plasma jet technology. For instance, as reported by Ma et al. (2015), Shen et al. (2016), Sivachandiran and Khacef (2017), Liao et al. (2018), and Guragain et al. (2023), pH of tap water decreased from 6.0–7.19 to 2.3–4.69 after 5–30 min of plasma treatment, as observed in the present study. Previous studies have shown that water acidification is the result of formation and accumulation of active ions in the solution, specifically nitrite (NO2–), nitrate (NO3–) ions, and H2O2 (Dunand et al. 2007; Ma et al. 2015). It was determined in those studies that NO2–, NO3–, and H2O2, initially absent in untreated water, increased, respectively, to the ranges of 0.49–0.86, 1.49–75.0, and 2.15–70.0 mg/L, after 5–20 min of plasma treatment (Sivachandiran and Khacef 2017; Liao et al. 2018; Stoleru et al. 2020; Than et al. 2022; Guragain et al. 2021), which also aligns with our findings. However, values of NO2–, NO3–, and H2O2 obtained in our studies after plasma treatment are much higher than found in above-mentioned studies. These differences may be attributed to the plasma reactor design, treatment time, voltage, and feed gas used for plasma generation (Ma et al. 2015; Zhou et al. 2018).
Properties of PAW-irrigated tomato plants
Physicochemical properties of PAW are recognized for their substantial impact on plant properties (Guo et al. 2021), and alterations in leaf reflectance profiles may indicate such changes. In our study, we observed a significant increase in relative leaf reflectance at 445 and 700–1015 nm, which correspond to the blue and near-infrared spectrum, respectively. These specific spectral bands are known for their strong absorption properties by chlorophyll and leaf pigmentation (Sims and Gamon 2002). This suggests that plants irrigated with water whose physicochemical properties have been altered by plasma discharge may improve the uptake and utilization of essential nutrients necessary for efficient photosynthetic processes in plants (Kučerová et al. 2021). Additionally, previous studies have indicated that changes in plant leaf reflectance within specific wavelength bands, such as 775–850 nm and 910–960 nm, can be indicative of nitrogen levels in plants (Sishodia et al. 2020; Rubo and Zinkernagel 2022; Rubio-Delgado et al. 2021). Given that PAW contains reactive nitrogen species (RNS), changes in leaf reflectance due to PAW treatments may be attributed to nitrogen assimilation. This finding is supported by our element composition analysis, which showed an enhancement in leaf nitrogen content in response to PAW treatments. Moreover, our results demonstrated an improvement in the levels of other essential elements such as P, S, Ca, and Mg. These positive effects of supplementary PAW irrigation on nutrient uptake and maintenance of a balanced nutrient profile in plants can be attributed to the beneficial role of hydrogen peroxide (H2O2), a reactive oxygen species found in PAW, which interacts with RNS. This aligns with the findings of Nurnaeimah et al. (2020), who observed that exogenous application of 8-mM H2O2 not only significantly improved nitrogen mineral uptake, but also the uptake of other elements including arsenic, iron, calcium, and potassium. Wang et al. (2023) also reported that PAW supplementation enhanced accumulation of both macro-elements (Ca, P, Na, K, S, and Mg) and microelements (Mn, Fe, and Zn) in fresh wheat (Triticum aestivum L.). A truly innovative and important aspect of this study is how we were able to associate leaf reflectance features with element composition, and our results were corroborated by the existing body of literature. Non-glandular and glandular trichomes densities have been extensively documented as the primary mechanism by which plants defend themselves against arthropod pests (Savi et al. 2022; de Lima Filho et al. 2022; Lucini et al. 2015; Koller et al. 2007). In our study, we observed an increase in the density of both non-glandular and glandular trichomes on PAW-irrigated plants. This is the first report showing that the plants irrigated with PAW resulted in a significant increase in both non-glandular and glandular trichomes density on tomato leaves. These findings are promising and could potentially enhance plant resistance, thus contributing to efforts to reduce reliance on synthetic pesticides and promote sustainable pest management practices.
Paw-irrigated plants responses to two-spotted spider mites
Leaf discs from both 6- and 9.4-min PAW-irrigated plants revealed a significant avoidance of settling by two-spotted spider mites indicating a potential deterrent effect of PAW supplementation. Additionally, significant avoidance of oviposition by two-spotted spider mites was observed on leaf discs from 6-min PAW-irrigated plants suggesting that plants irrigated with a specific PAW regime may provide more robust deterrent effect against arthropod pests. On the other hand, development and reproduction of two-spotted spider mites were substantially delayed on both 6- and 9.4-min PAW-irrigated plants as evidenced by their lower population growth on those plants. These findings support the evidence that PAW is potentially useful as a tool to enhance antixenosis and antibiosis resistance mechanisms in plants against arthropod pests (Savi et al. 2021, 2022), with PAW 6.0 appearing to provide better protection to tomato plants than PAW 9.4. The observed deterrent effects of PAW on two-spotted spider mites in our study may be attributed to the increased trichome densities, particularly glandular trichomes resulting from PAW irrigation. These trichomes produce a range of secondary metabolites including methyl-ketones (notably 2-tridecanone), sesquiterpene, zingiberene, and acyl sugars, which are known have toxic, repellent, and/or anti-nutritive effects on spider mites (Savi et al. 2021, 2022; Lucini et al. 2015; Oliveira et al. 2020; Rioja et al. 2017). However, our study did not assess the differences in secondary metabolites on PAW-irrigated tomato plants in the present study. Further research is required to identify the specific compounds associated with the trichomes found in PAW-irrigated plants that provide enhanced defense against two-spotted spider mite attacks.
Additionally, exogenous application of H2O2 has been shown to enhance common bean (Phaseolus vulgaris L.) plant resistance against two-spotted spider mites (Alakhdar and Shoala 2021). H2O2 acts as a messenger molecule, triggering the activation of OXI1 kinases, induction of callose production, triggering of MAPK cascades, and promotion of the transcription of defense-related genes (Neill et al. 2002; Rentel et al. 2004). Therefore, the presence of H2O2 in PAW supplemented to the plants may have also contributed to the significant deterrent effect on two-spotted spider mites.
Previous studies have also reported that H2O2 in PAW stimulates the production of phytohormones, such as salicylic acid and jasmonic acid, which are essential for activating plant immune responses (Laurita et al. 2021; Adhikari et al. 2019). Additionally, PAW has been reported to increase the activity of antioxidant enzymes such as superoxide dismutase, peroxidase, and catalase (Ramazzina et al. 2022; Kučerová et al. 2019, 2021), which might neutralize harmful reactive oxygen species produced during pest attacks. However, at excessive concentration, H2O2 can induce oxidative stress in plants, potentially leading to suboptimal responses to abiotic and biotic stresses (Alakhdar and Shoala 2021). This could explain the low deterrent effect on two-spotted spider mite oviposition observed on plants irrigated with PAW 9.4, which contains a high level of H2O2. Therefore, PAW regime must carefully consider H2O2 levels and other properties to avoid inducing stress and compromising plant resilience. Moreover, other factors, such as the nutritional balance resulting from PAW irrigation in our study, might have contributed to the resistance of tomato plants against two-spotted spider mites, although consensus on the relationship between plant nutritional state and defense mechanisms is still elusive (Singh and Sood 2017; Chaboussou 2004; Altieri and Nicholls 2003). It is essential to consider environmental factors such as temperature, humidity, soil conditions, and plant genotype which might affect PAW-irrigated plants and two-spotted spider mite interactions.
This study provides for the first time evidence that PAW could serve be a valuable tool for sustainable pest management. In addition to its established roles in seed germination, plant growth, yield enhancement, and plant pathogen control, PAW was found to improve plant nutrient uptake, increase leaf reflectance, and increase trichome densities (a key plant defense mechanism against arthropod pests). As a result, it reduced the preference and performance of two-spotted spider mites. These findings are encouraging and suggest the need for further investigation to determine the practical use of PAW in pest management. This would involve assessing its effectiveness on different plant crops, varieties, and pest species, as well as examining potential synergistic effects with other pest management strategies. It is also important to evaluate the benefits of repeated administration of PAW throughout the crop cycle under various environmental conditions and to consider the quality of the plant products and any possible side effects. This comprehensive assessment will provide valuable insights into the broader implications and limitations of PAW for sustainable pest management practices.
References
Adhikari B, Adhikari M, Ghimire B, Park G, Choi EH (2019) Cold atmospheric plasma-activated water irrigation induces defense hormone and gene expression in tomato seedlings. Sci Rep. https://doi.org/10.1038/s41598-019-52646-z
Alakhdar HH, Shoala T (2021) Exogenous application of hydrogen peroxide in different resistant bean cultivars of Phaseolus vulgaris to Tetranychus urticae (Acari: Tetranychidae). Arthropod-Plant Interact 15(3):439–445. https://doi.org/10.1007/s11829-021-09829-1
Altieri MA, Nicholls CI (2003) Soil fertility management and insect pests: harmonizing soil and plant health in agroecosystems. Soil Tillage Res 72(2):203–211. https://doi.org/10.1016/S0167-1987(03)00089-8
Ambrico PF, Šimek M, Rotolo C, Morano M, Minafra A, Ambrico M, Pollastro S, Gerin D, Faretra F, de Miccolis Angelini RM (2020) Surface dielectric barrier discharge plasma: a suitable measure against fungal plant pathogens. Sci Rep. https://doi.org/10.1038/s41598-020-60461-0
Attia S, Grissa KL, Lognay G, Bitume E, Hance T, Mailleux AC (2013) A review of the major biological approaches to control the worldwide pest Tetranychus urticae (Acari: Tetranychidae) with special reference to natural pesticides. J Pest Sci 86(3):361–386. https://doi.org/10.1007/s10340-013-0503-0
Chaboussou F (2004) Healthy crops: a new agricultural revolution. Jon Carpenter for the Gaia Foundation, Charlbury
Chew NSL, Ooi CW, Yeo LY, Tan MK (2023) Hybrid atmospheric pressure plasma generation and DC electrospray aerosolization of plasma-activated water for surface pathogen disinfection. Plasma Process Polym 20(3):2200128. https://doi.org/10.1002/ppap.202200128
de Lima Filho RB, Resende JTV, de Oliveira JRF, Nardi C, Silva PR, Rech C, Oliveira LVB, Ventura MU, Ribeiro Silva ALB (2022) Relationship between acylsugars and leaf trichomes: mediators of pest resistance in tomato. Insects 13(8):738. https://doi.org/10.3390/insects13080738
Dunand C, Crèvecoeur M, Penel C (2007) Distribution of superoxide and hydrogen peroxide in Arabidopsis root and their influence on root development: possible interaction with peroxidases. New Phytol 174(2):332–341. https://doi.org/10.1111/j.1469-8137.2007.01995.x
Fan L, Liu X, Ma Y, Xiang Q (2020) Effects of plasma-activated water treatment on seed germination and growth of mung bean sprouts. J Taibah Univ Sci 14(1):823–830. https://doi.org/10.1080/16583655.2020.1778326
Farooq M, Rehman A, Pisante M (2019) Sustainable agriculture and food security. Springer, Berlin, pp 3–24. https://doi.org/10.1007/978-3-030-23169-9_1
Fauvel M, Dechesne C, Zullo A, Ferraty F (2015) Fast forward feature selection of hyperspectral images for classification with gaussian mixture models. IEEE J Select Topics Appl Earth Observ Remote Sens 8(6):2824–2831. https://doi.org/10.1109/JSTARS.2015.2441771
Filipić A, Primc G, Zaplotnik R, Mehle N, Gutierrez-Aguirre I, Ravnikar M, Mozetič M, Žel J, Dobnik D (2019) Cold atmospheric plasma as a novel method for inactivation of Potato Virus Y in water samples. Food Environ Virol 11(3):220–228. https://doi.org/10.1007/s12560-019-09388-y
Gao Y, Li M, Sun C, Zhang X (2022) Microbubble-enhanced water activation by cold plasma. Chem Eng J 446:137318. https://doi.org/10.1016/j.cej.2022.137318
Guo D, Liu H, Zhou L, Xie J, He C (2021) Plasma-activated water production and its application in agriculture. J Sci Food Agric 101(12):4891–4899. https://doi.org/10.1002/jsfa.11258
Guragain RP, Baniya HB, Pradhan SP, Pandey BP, Subedi DP (2021) Influence of plasma-activated water (PAW) on the germination of radish, fenugreek, and pea seeds. AIP Adv 11(12):125304. https://doi.org/10.1063/5.0070800
Guragain RP, Baniya HB, Shrestha B, Guragain DP, Subedi DP (2023) Improvements in germination and growth of sprouts irrigated using plasma activated water (PAW). Water 15(4):744. https://doi.org/10.3390/w15040744
Han Q-Y, Wen X, Gao J-Y, Zhong C-S, Ni Y-Y (2023) Application of plasma-activated water in the food industry: a review of recent research developments. Food Chem 405:134797. https://doi.org/10.1016/j.foodchem.2022.134797
Hanbal SE, Takashima K, Miyashita S, Ando S, Ito K, Elsharkawy MM, Kaneko T, Takahashi H (2018) Atmospheric-pressure plasma irradiation can disrupt tobacco mosaic virus particles and RNAs to inactivate their infectivity. Arch Virol 163(10):2835–2840. https://doi.org/10.1007/s00705-018-3909-4
Huang L, Ding W, Liu W, Zhao J, Huang W, Xu C, Zhang D, Liang D (2019) Identification of wheat powdery mildew using in-situ hyperspectral data and linear regression and support vector machines. J Plant Pathol 101(4):1035–1045. https://doi.org/10.1007/s42161-019-00334-2
Huber D, Römheld V, Weinmann M (2012) Chapter 10 - Relationship between nutrition, plant diseases and pests. In: Marschner P (ed) Marschner’s Mineral Nutrition of Higher Plants, 3rd edn. Academic Press, San Diego, pp 283–298. https://doi.org/10.1016/B978-0-12-384905-2.00010-8
Jin J, Tang L, Hruska Z, Yao H (2009) Classification of toxigenic and atoxigenic strains of Aspergillus flavus with hyperspectral imaging. Comput Electron Agric 69(2):158–164
Joern A, Provin T, Behmer ST (2012) Not just the usual suspects: Insect herbivore populations and communities are associated with multiple plant nutrients. Ecology 93(5):1002–1015. https://doi.org/10.1890/11-1142.1
Ju S, Li B, Yang X, Wu J, Pei X, Zhuang J, Ma R, Jiao Z, Zhou R, Cullen PJ (2023) Plasma-activated water for inactivation of Fusarium graminearum spores: an in-vitro wheat study. LWT 183:114815. https://doi.org/10.1016/j.lwt.2023.114815
Koller M, Knapp M, Schausberger P (2007) Direct and indirect adverse effects of tomato on the predatory mite Neoseiulus californicus feeding on the spider mite Tetranychus evansi. Entomol Exp Appl 125(3):297–305. https://doi.org/10.1111/j.1570-7458.2007.00625.x
Kučerová K, Henselová M, Slováková Ľ, Hensel K (2019) Effects of plasma activated water on wheat: germination, growth parameters, photosynthetic pigments, soluble protein content, and antioxidant enzymes activity. Plasma Process Polym 16(3):1800131. https://doi.org/10.1002/ppap.201800131
Kučerová K, Henselová M, Slováková Ľ, Bačovčinová M, Hensel K (2021) Effect of plasma activated water, hydrogen peroxide, and nitrates on lettuce growth and its physiological parameters. App Sci 11(5):1985. https://doi.org/10.3390/app11051985
Landis JR, Koch GG (1977) The measurement of observer agreement for categorical data. Biometrics 33(1):159–174
Laurita R, Contaldo N, Zambon Y, Bisag A, Canel A, Gherardi M, Laghi G, Bertaccini A, Colombo V (2021) The use of plasma-activated water in viticulture: Induction of resistance and agronomic performance in greenhouse and open field. Plasma Process Polym 18(1):2000206. https://doi.org/10.1002/ppap.202000206
Lee G, Choi S-W, Yoo M, Chang H-J, Lee N (2023) Effects of plasma-activated water treatment on the inactivation of microorganisms present on cherry tomatoes and in used wash solution. Foods 12(13):2461. https://doi.org/10.3390/foods12132461
Li X, Xu H, Feng L, Fu X, Zhang Y, Nansen C (2017) Using proximal remote sensing in non-invasive phenotyping of invertebrates. PLoS ONE 12(5):e0176392. https://doi.org/10.1371/journal.pone.0176392
Liao X, Su Y, Liu D, Chen S, Hu Y, Ye X, Wang J, Ding T (2018) Application of atmospheric cold plasma-activated water (PAW) ice for preservation of shrimps (Metapenaeus ensis). Food Control 94:307–314. https://doi.org/10.1016/j.foodcont.2018.07.026
Lucini T, Faria MV, Rohde C, Resende JTV, De Oliveira JRF (2015) Acylsugar and the role of trichomes in tomato genotypes resistance to Tetranychus urticae. Arthropod-Plant Interact 9(1):45–53. https://doi.org/10.1007/s11829-014-9347-7
Ma R, Wang G, Tian Y, Wang K, Zhang J, Fang J (2015) Non-thermal plasma-activated water inactivation of food-borne pathogen on fresh produce. J Hazard Mater 300:643–651. https://doi.org/10.1016/j.jhazmat.2015.07.061
McMurtry JA, de Moraes GJ, Sourassou NF (2013) Revision of the lifestyles of phytoseiid mites (Acari:Phytoseiidae) and implications for biological control strategies. Syst Appl Acarol 18(4):297–320. https://doi.org/10.11158/saa.18.4.1
Nansen C (2016) The potential and prospects of proximal remote sensing of arthropod pests. Pest Manag Sci 72(4):653–659. https://doi.org/10.1002/ps.4209
Nansen C, Murdock M, Purington R, Marshall S (2021) Early infestations by arthropod pests induce unique changes in plant compositional traits and leaf reflectance. Pest Manag Sci 77(11):5158–5169. https://doi.org/10.1002/ps.6556
Neill SJ, Desikan R, Clarke A, Hurst RD, Hancock JT (2002) Hydrogen peroxide and nitric oxide as signalling molecules in plants. J Exp Bot 53(372):1237–1247. https://doi.org/10.1093/jexbot/53.372.1237
Nguyen HDD, Pan V, Pham C, Valdez R, Doan K, Nansen C (2020) Night-based hyperspectral imaging to study association of horticultural crop leaf reflectance and nutrient status. Comput Electron Agric 173:105458. https://doi.org/10.1016/j.compag.2020.105458
Nurnaeimah N, Mat N, Suryati Mohd K, Badaluddin NA, Yusoff N, Sajili MH, Mahmud K, Mohd Adnan AF, Khandaker MM (2020) The Effects of hydrogen peroxide on plant growth, mineral accumulation, as well as biological and chemical properties of Ficus deltoidea. Agronomy 10(4):599. https://doi.org/10.3390/agronomy10040599
Oliveira JRFd, Resende JTVd, Filho RBdL, Roberto SR, Silva PRd, Rech C, Nardi C (2020) Tomato breeding for sustainable crop systems: high levels of Zingiberene providing resistance to multiple arthropods. Horticulturae 6(2):34
Pankaj SK, Keener KM (2017) Cold plasma: background, applications and current trends. Curr Opin Food Sci 16:49–52. https://doi.org/10.1016/j.cofs.2017.07.008
Perez SM, Biondi E, Laurita R, Proto M, Sarti F, Gherardi M, Bertaccini A, Colombo V (2019) Plasma activated water as resistance inducer against bacterial leaf spot of tomato. PLoS ONE 14(5):e0217788. https://doi.org/10.1371/journal.pone.0217788
Priatama RA, Beak HK, Song I, Park S-J, Lee YK (2023) Long-term plasma-activated-water irrigation improves fruit yield in tomato. Res Square Platform LLC. https://doi.org/10.21203/rs.3.rs-3066150/v1
Rakha M, Bouba N, Ramasamy S, Regnard J-L, Hanson P (2017) Evaluation of wild tomato accessions (Solanum spp.) for resistance to two-spotted spider mite (Tetranychus urticae Koch) based on trichome type and acylsugar content. Genet Resour Crop Evol 64(5):1011–1022. https://doi.org/10.1007/s10722-016-0421-0
Ramazzina I, Lolli V, Lacey K, Tappi S, Rocculi P, Rinaldi M (2022) Fresh-Cut eruca sativa treated with plasma activated water (PAW): evaluation of antioxidant capacity, polyphenolic profile and redox status in CaCO2 cells. Nutrients. https://doi.org/10.3390/nu14245337
Rentel MC, Lecourieux D, Ouaked F, Usher SL, Petersen L, Okamoto H, Knight H, Peck SC, Grierson CS, Hirt H, Knight MR (2004) OXI1 kinase is necessary for oxidative burst-mediated signalling in Arabidopsis. Nature 427(6977):858–861. https://doi.org/10.1038/nature02353
Rioja C, Zhurov V, Bruinsma K, Grbic M, Grbic V (2017) Plant-herbivore interactions: a case of an extreme generalist, the two-spotted spider mite Tetranychus urticae. Mol Plant Microbe Interact 30(12):935–945
Rubio-Delgado J, Pérez CJ, Vega-Rodríguez MA (2021) Predicting leaf nitrogen content in olive trees using hyperspectral data for precision agriculture. Precis Agric 22(1):1–21. https://doi.org/10.1007/s11119-020-09727-1
Rubo S, Zinkernagel J (2022) Exploring hyperspectral reflectance indices for the estimation of water and nitrogen status of spinach. Biosyst Eng 214:58–71. https://doi.org/10.1016/j.biosystemseng.2021.12.008
Salazar-Mendoza P, Magalhães DM, Lourenção AL, Bento JMS (2023) Differential defensive and nutritional traits among cultivated tomato and its wild relatives shape their interactions with a specialist herbivore. Planta. https://doi.org/10.1007/s00425-023-04108-0
Savi PJ, de Moraes GJ, Boiça-Junior AL, Melville CC, Carvalho RF, Lourenção AL, Andrade AL, Junior DJ (2019) Impact of leaflet trichomes on settlement and oviposition of Tetranychus evansi (Acari: Tetranychidae) in African and South American tomatoes. Syst Appl Acarol 24(12):2559–2576. https://doi.org/10.11158/saa.24.12.19.short
Savi PJ, Gonsaga RF, de Matos STS, Braz LT, de Moraes GJ, Andrade DJ (2021) Performance of Tetranychus urticae (Acari: Tetranychidae) on three hop cultivars (Humulus lupulus). Exp Appl Acarol 84(4):733–753. https://doi.org/10.1007/s10493-021-00643-1
Savi PJ, de Moraes GJ, Carvalho RF, Andrade DJ (2022) Bottom-up effects of breeding tomato genotypes on behavioural responses and performance of Tetranychus evansi population. J Pest Sci 95(3):1287–1301. https://doi.org/10.1007/s10340-021-01437-5
Shen J, Tian Y, Li Y, Ma R, Zhang Q, Zhang J, Fang J (2016) Bactericidal effects against S. aureus and physicochemical properties of plasma activated water stored at different temperatures. Sci Rep 6(1):28505. https://doi.org/10.1038/srep28505
Simmons AT, Gurr GM (2005) Trichomes of Lycopersicon species and their hybrids: effects on pests and natural enemies. Agric for Entomol 7(4):265–276. https://doi.org/10.1111/j.1461-9555.2005.00271.x
Sims DA, Gamon JA (2002) Relationships between leaf pigment content and spectral reflectance across a wide range of species, leaf structures and developmental stages. Remote Sens Environ 81(2):337–354. https://doi.org/10.1016/S0034-4257(02)00010-X
Singh V, Sood AK (2017) Plant Nutrition: a tool for the management of hemipteran insect-pests-a review. Agric Rev. https://doi.org/10.18805/ag.r-1637
Sishodia RP, Ray RL, Singh SK (2020) Applications of remote sensing in precision agriculture: a review. Remote Sens 12(19):3136. https://doi.org/10.3390/rs12193136
Sivachandiran L, Khacef A (2017) Enhanced seed germination and plant growth by atmospheric pressure cold air plasma: combined effect of seed and water treatment. RSC Adv 7(4):1822–1832. https://doi.org/10.1039/c6ra24762h
Škarpa P, Klofáč D, Krčma F, Šimečková J, Kozáková Z (2020) Effect of plasma activated water foliar application on selected growth parameters of maize (Zea mays L.). Water 12(12):3545. https://doi.org/10.3390/w12123545
Stoleru V, Burlica R, Mihalache G, Dirlau D, Padureanu S, Teliban G-C, Astanei D, Cojocaru A, Beniuga O, Patras A (2020) Plant growth promotion effect of plasma activated water on Lactuca sativa L. cultivated in two different volumes of substrate. Sci Rep. https://doi.org/10.1038/s41598-020-77355-w
Ten Bosch L, Köhler R, Ortmann R, Wieneke S, Viöl W (2017) Insecticidal effects of plasma treated water. Int J Environ Res Public Health 14(12):1460. https://doi.org/10.3390/ijerph14121460
Than HAQ, Pham TH, Nguyen DKV, Pham TH, Khacef A (2022) Non-thermal plasma activated water for increasing germination and plant growth of Lactuca sativa L. Plasma Chem Plasma Process 42(1):73–89. https://doi.org/10.1007/s11090-021-10210-6
Thirumdas R, Sarangapani C, Annapure US (2015) Cold Plasma: a novel non-thermal technology for food processing. Food Biophys 10(1):1–11. https://doi.org/10.1007/s11483-014-9382-z
Tiftikçi P, Kök Ş, Kasap İ (2020) Biological control of twospotted spider mites [Tetranychus urticae Koch (Acari: Tetranychidae)] using Phytoseiulus persimilis Athias-Henriot (Acari: Phytoseidae) at different ratios of release on field-grown tomatos. Biol Control 151:104404. https://doi.org/10.1016/j.biocontrol.2020.104404
Tiftikçi P, Kök Ş, Kasap İ (2022) The effect of host plant on the biological control efficacy of the predatory mite, Phytoseiulus persimilis Athias-Henriot against two-spotted spidermites, Tetranychus urticae koch on field-grown vegetables. Crop Prot 158:106012. https://doi.org/10.1016/j.cropro.2022.106012
Vichiansan N, Chatmaniwat K, Sungkorn M, Leksakul K, Chaopaisarn P, Boonyawan D (2023) Effect of plasma-activated water generated using plasma jet on tomato (Solanum lycopersicum L. var. cerasiforme) seedling growth. J Plant Growth Regul 42(2):935–945. https://doi.org/10.1007/s00344-022-10603-7
Wang J, Cheng J-H, Sun D-W (2023) Enhancement of wheat seed germination, seedling growth and nutritional properties of wheat plantlet juice by plasma activated water. J Plant Growth Regul 42(3):2006–2022. https://doi.org/10.1007/s00344-022-10677-3
Weinblum N, Cna’Ani A, Yaakov B, Sadeh A, Avraham L, Opatovsky I, Tzin V (2021) Tomato cultivars resistant or susceptible to spider mites differ in their biosynthesis and metabolic profile of the monoterpenoid pathway. Front Plant Sci. https://doi.org/10.3389/fpls.2021.630155
Xu H, Liu C, Huang Q (2023) Enhance the inactivation of fungi by the sequential use of cold atmospheric plasma and plasma-activated water: synergistic effect and mechanism study. Chem Eng J 452:139596. https://doi.org/10.1016/j.cej.2022.139596
Yang F, Zhang X, Shen H, Xue H, Tian T, Zhang Q, Hu J, Tong H, Zhang Y, Su Q (2023) Flavonoid-producing tomato plants have a direct negative effect on the zoophytophagous biological control agent Orius sauteri. Insect Sci 30(1):173–184. https://doi.org/10.1111/1744-7917.13085
Yepez X, Illera AE, Baykara H, Keener K (2022) Recent advances and potential applications of atmospheric pressure cold plasma technology for sustainable food processing. Foods 11(13):1833. https://doi.org/10.3390/foods11131833
Zambon Y, Contaldo N, Laurita R, Várallyay E, Canel A, Gherardi M, Colombo V, Bertaccini A (2020) Plasma activated water triggers plant defence responses. Sci Rep. https://doi.org/10.1038/s41598-020-76247-3
Zhou R, Zhou R, Prasad K, Fang Z, Speight R, Bazaka K, Ostrikov K (2018) Cold atmospheric plasma activated water as a prospective disinfectant: the crucial role of peroxynitrite. Green Chem 20(23):5276–5284. https://doi.org/10.1039/C8GC02800A
Funding
This work was supported by CDFA (California Department of Food and Agriculture) Grant No. 21-0732-001-SF and USDA (United States Department of Agriculture)–NIFA (National Institute of Food and Agriculture) Grant No. 2023-51106-40946.
Author information
Authors and Affiliations
Contributions
Conceptualization was performed by PJS; methodology, investigation, and analysis by PJS, AM, HK, YZ, and CN; writing draft by PJS, GJM, and CN. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Consent to publication
All authors contributed critically to the drafts and gave final approval for publication.
Additional information
Communicated by Cesar Rodriguez-Saona.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Savi, P.J., Mantri, A., Khodaverdi, H. et al. Indirect effects of plasma-activated water irrigation on Tetranychus urticae populations. J Pest Sci (2024). https://doi.org/10.1007/s10340-024-01791-0
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10340-024-01791-0