Abstract
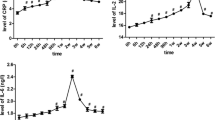
To investigate the relationship between apical periodontitis [AP] severity and inflammatory markers [interleukin (IL)-12, tumor necrosis factor-alpha (TNF-α), and Mid-Regional Pro Adrenomedullin (MR-proADM)] in patients with AP. A total of 162 subjects were divided into three categories: AP group (n = 80), periodontitis (P) group (n = 42), healthy control group (n = 40). The scoring of disease severity in 80 AP patients without any periodontal disease, using dental radiographs, was based on “The Abscess Score” (AS), as those having at least 1 tooth with AP and severity of PAI 3–4 were classified as AS 1 (mild); those with only1 tooth and severity of PAI 5 as AS 2 (moderate) and those having > 2 tooth with severity of PAI 5 as AS 3 (severe). Blood samples were collected from all of the patients. Enzyme-linked immunosorbent assay was used to evaluate the samples. The MR-pro ADM levels of both the AP and P groups were considerably higher than the control group (p < 0.01). The IL-12 levels of the AP group were higher than the P and control groups (p < 0.05). TNF-α levels of the P group were significantly higher than both the AP and control groups (p < 0.01). MR-pro ADM levels of both the AP and Periodontitis groups were higher than the control group. TNF-α was a biomarker of periodontitis, while IL-12 was a biomarker of apical periodontitis.




Similar content being viewed by others
Data Availability
The datasets analyzed during the current study are available from the corresponding author on reasonable request.
References
Silva TA, Garlet GP, Lara VS, Martins W Jr, Silva JS, Cunha FQ. Differential expression of chemokines and chemokine receptors in inflammatory periapical diseases. Oral Microbiol Immunol. 2005;20:310–6.
Wilson M, Reddi K, Henderson B. Cytokine-inducing components of periodontopathogenic bacteria. J Periodontal Res. 1996;31:393–407.
Abbott PV, Yu C. A clinical classification of the status of the pulp and the root canal system. Aust Dent J. 2007;52:17–31.
Tibúrcio-Machado CS, Michelon C, Zanatta FB, Gomes MS, Marin JA, Bier CA. The global prevalence of apical periodontitis: a systemic review and meta-analysis. Int Endod J. 2021;54:712–35.
Jakovljevic A, Nikolic N, Jacimovic J, Pavlovic O, Milicic B, Beljic-Ivanovic K, et al. Prevalence of apical periodontitis and conventional nonsurgical root canal treatment in general adult population: An updated systematic review and meta-analysis of cross-sectional studies published between 2012 and 2020. J Endod. 2020;46:1371–86.
Georgiou AC, Crielaard W, Armenis I, de Vries R, van der Waal SV. Apical periodontitis is associated with elevated concentrations of inflammatory mediators in peripheral blood: A systematic review and meta-analysis. J Endod. 2019;45:1279–95.
Gomes MS, Blattner TC, Sant’Ana Filho M, et al. Can apical periodontitis modify systemic levels of inflammatory markers? A systematic review and meta-analysis. J Endod. 2013;39:1205–17.
Caplan DJ. Epidemiologic issues in studies of association between apical periodontitis and systemic health. Endod Topics. 2004;8:5–35.
Huang GT, Do M, Wingard M, Park JS, Chugal N. Effect of interleukin-6 deficiency on the formation of periapical lesions after pulp exposure in mice. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2001;92:83–8.
Garrido M, Cardenas AM, Astorga J, et al. Elevated systemic inflammatory burden and cardiovascular risk in young adults with endodontic apical lesions. J Endod. 2019;45:111–5.
Hinson JP, Kapas S, Smith DM. Adrenomedullin, a multifunctional regulatory peptide. Endocr Rev. 2000;21:138–67.
Hussain QA, McKay IJ, Gonzales-Marin C, Allaker RP. Detection of adrenomedullin and nitric oxide in different forms of periodontal disease. J Periodontal Res. 2016;51:16–25.
Yildirim NC, Yurekli M. The effect of adrenomedullin and cold stress on interleukin-6 levels in some rat tissues. Clin Exp Immunol. 2010;161:171–5.
Hussain QA, McKay IJ, Gonzales-Marin C, Allaker RP. Regulation of adrenomedullin and nitric oxide production by periodontal bacteria. J Periodontal Res. 2015;50:650–7.
Jansson L. Relationship between apical periodontitis and marginal bone loss at individual level from a general population. Int Dent J. 2015;65:71–6.
Ørstavik D, Kerekes K, Eriksen HM. The periapical index: a scoring system for radiographic assessment of apical periodontitis. Dent Traumatol. 1986;2:20–34.
Tonetti MS, Greenwell H, Kornman KS. Staging and grading of periodontitis: Framework and proposal of a new classification and case definition. J Periodontol. 2018;89:S159–72.
Suchetha A, Garg A, Lakshmi P, Bhat D, Sapna N, Apoorva SM. Adrenomedullin, periodontitis, diabetes-unraveling the equivocal relationship: A clinicobiochemical cross-sectional study. Contemp Clin Dent. 2013;4:454–9.
Allaker RP, Kapas S. Adrenomedullin and mucosal defence: interaction between host and microorganism. Regul Pept. 2003;112:147–52.
Hosokawa I, Hosokawa Y, Ozaki K, Nakae H, Matsuo T. Adrenomedullin suppresses tumour necrosis factor alpha-induced CXC chemokine ligand 10 production by human gingival fibroblasts. Clin Exp Immunol. 2008;152:568–75.
Kapas S, Bansal A, Bhargava V, et al. Adrenomedullin expression in pathogen-challenged oral epithelial cells. Peptides. 2001;22:1485–9.
Lundy FT, O’Hare MM, McKibben BM, Fulton CR, Briggs JE, Linden GJ. Radioimmunoassay quantification of adrenomedullin in human gingival crevicular fluid. Arch Oral Biol. 2006;51:334–8.
Zhang CZ, Cheng XQ, Li JY, et al. Saliva in the diagnosis of diseases. Int J Oral Sci. 2016;8:133–7.
Elsasser TH, Kahl S. Adrenomedullin has multiple roles in disease stress: development and remission of the inflammatory response. Microsc Res Tech. 2002;57:120–9.
Horiuchi T, Mitoma H, Harashima S, Tsukamoto H, Shimoda T. Transmembrane TNF-alpha: structure, function and interaction with anti-TNF agents. Rheumatology. 2010;49:1215–28.
Wang H, Czura C, Chargaff E. Tumor necrosis factor. In: Thomson AW, Lotze MT, editors. The cytokine handbook. Amsterdam: Elsevier; 2003.
Nair SP, Meghji S, Wilson M, Reddi K, White P, Henderson B. Bacterially induced bone destruction: mechanisms and misconceptions. Infect Immun. 1996;64:2371–80.
Jakovljevic A, Knezevic A, Karalic D, Soldatovic I, Popovic B, Milasin J, Andric M. Pro-inflammatory cytokine levels in human apical periodontitis: correlation with clinical and histological findings. Aust Endod J. 2015;41:72–7.
Lee SM, Yang H, Tartar DM, Gao B, Luo X, Ye SQ, et al. Prevention and treatment of diabetes with resveratrol in a non-obese mouse model of type 1 diabetes. Diabetologia. 2011;54:1136–46.
Gazivoda D, Dzopalic T, Bozic B, Tatomirovic Z, Brkic Z, Colic M. Production of pro-inflammatory and immunoregulatory cytokines by inflammatory cells from periapical lesions in culture. J Oral Pathol Med. 2009;38:605–11.
Prso IB, Kocjan W, Simić H, Brumini G, et al. Tumor necrosis factor-alpha and interleukin 6 in human periapical lesions. Mediators Inflamm. 2007;2007:38210.
Steinman RM. Linking innate to adaptive immunity through dendritic cells. Novartis Found Symp. 2006;279:101–9.
Hunter CA. New IL-12-family members: IL-23 and IL-27, cytokines with divergent functions. Nat Rev. 2005;5:521–31.
Pope RM, Shahrara S. Possible roles of IL-12-family cytokines in rheumatoid arthritis. Nat Rev Rheumatol. 2013;9:252–6.
Bright JJ, Musuro BF, Du C, Sriram S. Expression of IL-12 in CNS and lymphoid organs of mice with experimental allergic encephalitis. J Neuroimmunol. 1998;82:22–30.
Abbas AK, Lichtman AH, Pillai S. Cellular and molecular immunology, Philadelphia. 7th ed. Amsterdam: Elsevier/Saunders; 2012.
Zhang C, Zhang J, Niu J, Zhou Z, Zhang J, Tian Z. Interleukin-12 improves cytotoxicity of natural killer cells via upregulated expression of NKG2D. Hum Immunol. 2008;69:490–500.
Queiroz-Junior CM, Silva MJ, Corrêa JD, et al. A controversial role for IL-12 in immune response and bone resorption at apical periodontal sites. Clin Dev Immunol. 2010;2010:327417.
Ridao-Sacie C, Segura-Egea JJ, Fernández-Palacín A, Bullón-Fernández P, Ríos-Santos JV. Radiological assessment of periapical status using the periapical index: comparison of periapical radiography and digital panoramic radiography. Int Endod J. 2007;40:433–40.
Petersen J, Glaßl EM, Nasseri P, et al. The association of chronic apical periodontitis and endodontic therapy with atherosclerosis. Clin Oral Investig. 2014;18:1813–23.
Vidal F, Fontes TV, Marques TVF, Gonçalves LS. Association between apical periodontitis lesions and plasmatic levels of C-reactive protein, interleukin 6 and fibrinogen in hypertensive patients. Int Endod J. 2016;49:1107–15.
Virtanen E, Nurmi T, Söder PÖ, Airila-Månsson S, Söder B, Meurman JH. Apical periodontitis associates with cardiovascular diseases: a cross-sectional study from Sweden. BMC Oral Health. 2017;17:107.
Mattila KJ, Nieminen MS, Valtonen VV, et al. Association between dental health and acute myocardial infarction. Brit Med J. 1989;298:779–81.
Ribas D, Fernández-Carranco MC, Hajji N, Bobadilla P, Monteseirín J. Eosinophil cationic protein and histamine production by neutrophils from periodontitis patients. J Periodontol. 2018;89:228–34.
Noh MK, Jung M, Kim SH, et al. Assessment of IL-6, IL-8 and TNF-α levels in the gingival tissue of patients with periodontitis. Exp Ther Med. 2013;6:847–51.
Ross JH, Hardy DC, Schuyler CA, Slate EH, Mize TW, Huang Y. Expression of periodontal interleukin-6 protein is increased across patients with neither periodontal disease nor diabetes, patients with periodontal disease alone and patients with both diseases. J Periodontal Res. 2010;45:688–94.
Kita T, Kitamura K, Hashida S, Morishita K, Eto T. Plasma adrenomedullin is closely correlated with pulse wave velocity in middle-aged and elderly patients. Hipertens Res. 2003;26:887–93.
Acknowledgements
All authors contributed equally to all study design, data collection, statistical analysis, data interpretation, manuscript preparation, and literature search. We would want to thank Dr. Batuhan Akca and Dr. Hasan Alp Topcu for their support during the English editing of our article.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have stated explicitly that there are no conflicts of interest in connection with this article.
Ethical approval
This research has been conducted in full accordance with ethical principles, including the World Medical Association Declaration of Helsinki (version 2008). All interventions were undertaken with the understanding and written consent of each participant and according to the above-mentioned principles. The study has been independently reviewed and approved by an ethical board.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ersahan, S., Ozcelik, F., Sirin, D.A. et al. Is adrenomedullin upregulation due to apical periodontitis independent of periodontal disease?. Odontology 111, 708–718 (2023). https://doi.org/10.1007/s10266-022-00767-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10266-022-00767-9